Quantum Physics Basics
Have you ever wondered what really happens inside the tiniest building blocks of our universe? Quantum physics basics unlock the secrets of atoms and particles that are too small to see, yet they shape everything around you.
This isn’t just complicated science — it’s a fascinating world where particles can be in two places at once and instantly affect each other across distances. By understanding these simple ideas, you’ll see how quantum physics influences the technology you use every day and changes how we think about reality itself.
Ready to explore the mind-bending basics of quantum physics? Let’s dive in and make the invisible, visible to you.
Credit: www.gmanetwork.com
Quanta And Discreteness
Quantum physics reveals that energy and matter are not continuous. Instead, they come in small, separate units called quanta. This idea changed how scientists view the microscopic world. It explains many strange behaviors that classical physics could not.
Quanta show us that nature works in steps, not in smooth waves. This concept helps us understand light, atoms, and all matter at a deep level. The study of quanta is the foundation of quantum mechanics.
Energy Packets
Energy does not flow in a steady stream. It travels in tiny packets called photons. Each photon carries a fixed amount of energy. This amount depends on the light's frequency. Higher frequency light has more energetic photons.
This idea solved puzzles about light emission and absorption. It showed why atoms emit light only in certain colors. These colors match the energy differences between atom states. This discovery was key to building quantum theory.
Matter In Quanta
Matter also shows quantum behavior. Particles like electrons exist in discrete energy states. They cannot have any energy value but only specific levels. Electrons jump between these levels by absorbing or emitting energy packets.
This discreteness explains the structure of atoms and molecules. It also leads to the strange idea that particles can act like waves. Understanding matter in quanta changed physics and chemistry forever.
Wave-particle Duality
Wave-particle duality is a key idea in quantum physics. It shows how tiny things like electrons and light can act like both waves and particles. This idea helps us understand strange behaviors in the quantum world. Scientists use wave-particle duality to explain many experiments and natural phenomena.
Particle Behavior
Particles are small bits of matter with specific locations. They can collide, bounce, and move like tiny balls. In quantum physics, particles sometimes behave like solid objects. For example, electrons can hit a screen at certain points, showing their particle nature. This behavior helps us predict where particles might be found.
Wave Characteristics
Waves spread out and can interfere with each other. They have properties like wavelength and frequency. In quantum physics, particles also show wave-like behavior. Electrons can create patterns of light and dark bands, called interference patterns. These patterns prove that particles can act like waves at the same time.
Superposition Principle
The superposition principle is a fundamental concept in quantum physics. It states that a quantum system can exist in multiple states at the same time. This idea challenges our everyday experience where things are usually in one state only. In quantum physics, particles like electrons or photons do not have a single position or energy. Instead, they are described by a combination of all possible states.
This combination is called a superposition. It means the particle holds many possibilities simultaneously. The superposition principle is key to understanding how quantum computers and other technologies might work. It shows the strange and fascinating nature of the quantum world.
Multiple States
Quantum particles do not have definite properties until measured. Instead, they exist in many states at once. For example, an electron can be in different places or have different spins simultaneously. This is very different from classical physics where objects have one clear state.
The superposition of states allows quantum systems to carry more information. Each state contributes to the overall behavior of the particle. Scientists describe this using a mathematical tool called a wave function. The wave function holds all the possible states and their probabilities.
Measurement Effects
Measuring a quantum system changes its state. Before measurement, the particle is in superposition. After measurement, it appears in only one state. This process is called wave function collapse.
The act of measuring forces the system to choose a definite state. This effect is unique to quantum physics. It means the observer plays an active role in determining the outcome. Measurement changes the reality of the quantum system.
Credit: www.energy.gov
Uncertainty Principle
The Uncertainty Principle is a fundamental idea in quantum physics. It tells us there are limits to what we can know about tiny particles. This principle was introduced by Werner Heisenberg in 1927. It changed how scientists think about measurement and observation in the quantum world.
The principle states that we cannot precisely know both the position and momentum of a particle at the same time. The more exactly we know one, the less exactly we know the other. This is not due to technical errors but a natural limit of the quantum world.
Position And Momentum Limits
In quantum physics, position means where a particle is. Momentum is how fast and in which direction it moves. The Uncertainty Principle says we cannot measure both with perfect accuracy. If we measure position very precisely, momentum becomes uncertain. If we measure momentum accurately, position becomes uncertain.
This limit exists because particles behave like waves. Waves spread out in space and time, making exact measurement impossible. The more we focus on one property, the more the other spreads out. This is a key difference from classical physics, where such measurements can be exact.
Implications For Measurement
The Uncertainty Principle affects how scientists observe and measure quantum systems. It means we cannot predict exact outcomes, only probabilities. Measurement changes the system being measured. Observing a particle’s position affects its momentum and vice versa.
This principle also means the universe at small scales is inherently uncertain. It challenges the idea of a perfectly predictable world. Scientists use this idea to develop new technologies like quantum computing and cryptography. Understanding these limits is crucial for progress in quantum research.
Quantum Entanglement
Quantum entanglement is one of the most fascinating ideas in quantum physics. It describes a unique connection between particles that links their properties. These particles behave as if they are connected, even when separated by large distances. This strange link challenges our usual understanding of space and time.
Particle Connections
Entangled particles share a special bond. Their states depend on each other. If you measure one particle, you instantly know the state of the other. This happens regardless of the distance between them. The connection is so strong that the particles act like a single system. Scientists use this idea to explore new technologies like quantum computing.
Non-local Effects
Quantum entanglement shows non-local effects. This means an action on one particle affects the other immediately. It seems to break the rule that nothing can travel faster than light. These effects puzzled even the greatest physicists. Experiments confirmed that entanglement is real and not just a theory. This discovery opens new questions about how the universe works.
Quantum Spin
Quantum spin is a fundamental property of particles in quantum physics. It is a type of angular momentum that does not have a direct classical counterpart. Spin helps explain many behaviors of particles like electrons and photons. It plays a key role in quantum mechanics and quantum computing.
Understanding quantum spin opens the door to grasping more complex quantum phenomena. It is not about particles spinning like tiny tops. Instead, spin is an intrinsic feature that affects how particles interact and combine.
Intrinsic Angular Momentum
Quantum spin is called intrinsic angular momentum. This means it is built into the particle itself. Unlike classical angular momentum, which depends on motion, spin exists even if the particle is at rest.
Particles have fixed spin values, such as 1/2 or 1. These values determine how particles behave in magnetic fields and how they follow quantum rules. Spin is quantized, which means it can only take certain values.
Spin States
Spin states describe the orientation of a particle's spin. For example, an electron can have "spin up" or "spin down" states. These states are important in quantum mechanics and help explain phenomena like magnetism.
Spin states can combine in complex ways. This leads to effects like entanglement and superposition. Measuring a spin state changes the system, showing the strange nature of the quantum world.
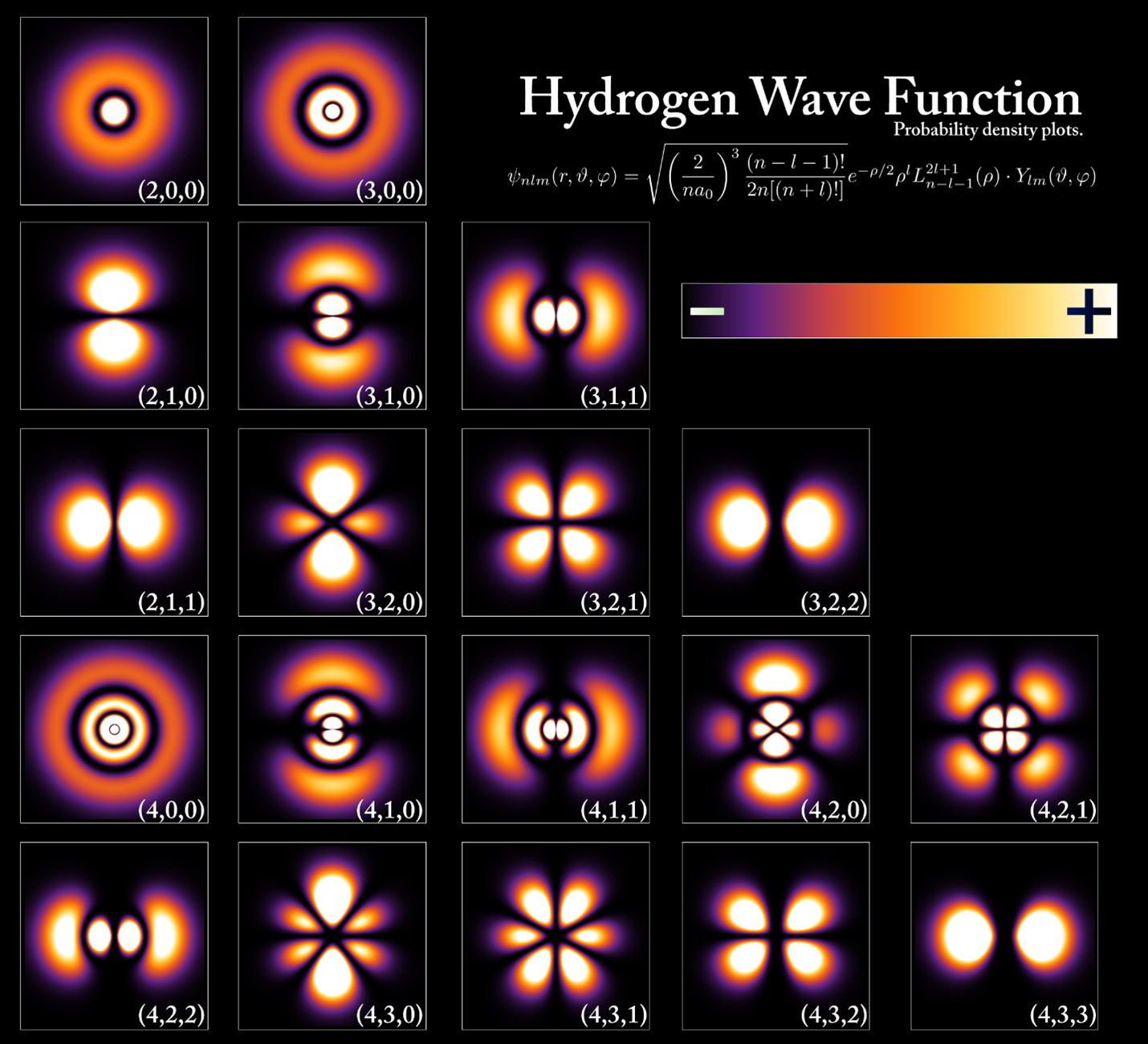
Wave Function Basics
The wave function is a core idea in quantum physics. It describes the state of a particle or system. This function is not just a simple wave but holds all possible information about the particle.
The wave function helps predict where a particle might be or how it behaves. It does this through mathematical rules and probabilities. Understanding the wave function basics is key to learning how quantum physics works.
Probability Amplitudes
The wave function gives a probability amplitude, a complex number. This number tells us the chance of finding a particle in a certain place. Squaring its size (magnitude) gives the actual probability.
Probability amplitudes can add up or cancel out, causing interference patterns. This effect explains many strange quantum behaviors. It differs from classical ideas where probabilities just add up.
Normalization
Normalization makes sure the total probability of finding the particle is one. This means the particle must be somewhere in space. Normalizing the wave function adjusts its scale to meet this rule.
Without normalization, probability results would be meaningless or incorrect. It ensures the wave function is physically valid. Normalization is a simple but crucial step in quantum calculations.
Schrödinger Equation
The Schrödinger Equation is a key idea in quantum physics. It helps describe how tiny particles like electrons behave. This equation shows how a particle's state changes over time and space. Scientists use it to predict the future position and energy of particles. The Schrödinger Equation forms the foundation of quantum mechanics, guiding our understanding of the atomic world.
Time-independent Equation
The time-independent Schrödinger Equation applies when a system's energy does not change over time. It helps find the allowed energy levels of particles. This form is simpler and often used to study atoms and molecules. The equation looks like a wave equation, where the wave function tells us the particle’s probability. Solving it gives important information about stable states and energies.
Potential Functions
Potential functions describe the forces acting on a particle in the Schrödinger Equation. They define areas where particles can move or get trapped. Different potentials create different energy landscapes for particles. For example, a potential well can hold a particle inside a small region. Understanding these potentials helps explain chemical bonds and the behavior of electrons in materials.
Quantum Harmonic Oscillator
The Quantum Harmonic Oscillator is a fundamental concept in quantum physics. It models particles in a potential well shaped like a parabola. This model helps scientists understand vibrations in molecules and atoms at the quantum level.
Unlike classical oscillators, the quantum version shows discrete energy states. It plays a key role in explaining many physical phenomena and forms the basis for more complex quantum systems.
Energy Levels
The energy levels of a quantum harmonic oscillator are quantized. This means the particle can only have specific energy values, not a continuous range. These energy levels are evenly spaced, unlike many other quantum systems.
Each level is labeled by a quantum number, starting from zero. The lowest level is called the ground state. Higher levels are called excited states. The difference between levels equals a fixed amount related to the oscillator’s frequency.
Power Series Solutions
Solving the quantum harmonic oscillator involves a mathematical method called power series. The Schrödinger equation for this system is transformed into a form solved by expanding the wave function in a series.
This approach finds solutions that are physically acceptable and normalizable. The power series method also reveals the shape of wave functions for each energy level. These solutions explain the probability of finding the particle at different positions.
Free Particles And Wave Packets
Free particles in quantum physics move without any forces acting on them. Their behavior is different from classical particles. Instead of having a fixed position, they are described by wave functions. These waves spread out over space, showing the particle’s probability in different places. Wave packets combine many waves to form a localized particle. This helps explain how particles can behave like waves and particles at the same time.
Stationary States
Stationary states are special wave functions with fixed energy. They do not change their shape over time. Instead, they only gain a phase factor that depends on time. These states are important in quantum physics because they represent stable conditions. For a free particle, stationary states are usually waves that extend infinitely. They show a definite momentum but no fixed position.
Boundary Conditions
Boundary conditions tell us how wave functions behave at the edges of a region. They ensure the wave function is smooth and continuous. For example, in a box, the wave must be zero at the walls. These conditions limit the allowed energies and shapes of the wave functions. Even free particles can have boundary conditions if they move in limited spaces. This idea helps explain many quantum phenomena.
Operators And Observables
In quantum physics, operators and observables play a key role. They help us understand physical quantities like position, momentum, and energy. Operators act on wave functions to extract information about these quantities. Observables are physical properties that can be measured. Each observable corresponds to a specific operator. This link between operators and observables forms the foundation of quantum mechanics.
Hermitian Operators
Hermitian operators are very important in quantum physics. They represent measurable quantities. Their main feature is that their eigenvalues are always real numbers. This matches the fact that measurement results are real values. Hermitian operators also have orthogonal eigenfunctions. This property ensures clear and distinct measurement outcomes. Without Hermitian operators, quantum measurements would not make physical sense.
Eigenvalues And Eigenfunctions
Eigenvalues and eigenfunctions are central concepts in quantum physics. An eigenvalue is the measured value of an observable. The eigenfunction is the state of the system associated with that value. When an operator acts on its eigenfunction, it returns the eigenvalue multiplied by the same eigenfunction. This process helps predict measurement results. It also shows the possible states a quantum system can be in.

Credit: www.goodreads.com
Angular Momentum In Quantum Systems
Angular momentum plays a key role in quantum physics. It describes how particles spin and orbit in atoms. Unlike classical physics, quantum angular momentum has unique properties. It does not change smoothly but in fixed steps. This concept helps us understand atomic structure and particle behavior.
Eigenfunctions
Eigenfunctions are special wave functions linked to angular momentum. They represent states with definite angular momentum values. When measured, these states give precise results. The eigenfunctions solve important equations in quantum mechanics. They help predict the outcome of experiments involving spin and rotation.
Quantization
Quantization means angular momentum can only take certain values. It appears in discrete units, not as a continuous range. These values depend on quantum numbers. The smallest unit is called the reduced Planck constant. This restriction leads to unique atomic and molecular properties.
Two-particle Systems
Two-particle systems are a key part of quantum physics. They help us understand how particles interact and behave together at the smallest scales. Studying these systems shows how particles can influence each other even when far apart. This interaction is very different from what we see in everyday life.
In quantum physics, two particles do not just act as separate units. Their states can become linked, creating unique and complex behaviors. These systems form the basis for many quantum technologies and experiments.
Entanglement In Multi-particle Systems
Entanglement occurs when two particles become connected. Their properties depend on each other, no matter the distance. Measuring one particle instantly affects the state of the other. This strange link puzzles scientists but is proven by many experiments.
Entanglement is important for quantum computing and secure communication. It allows particles to share information in ways classical systems cannot. In multi-particle systems, entanglement can involve many particles at once, creating complex networks of connections.
Energy Level Structures
Energy levels in two-particle systems show how particles share energy. These levels depend on the interaction between particles and their surroundings. The structure of energy levels determines the system’s stability and behavior.
Energy levels can split or shift when particles interact. This splitting creates different possible states the system can occupy. Understanding energy level structures helps predict how quantum systems respond to changes.
Quantum Applications
Quantum physics is the science behind many modern technologies. It studies tiny particles smaller than atoms. These particles behave in strange ways that classical physics cannot explain. Understanding quantum physics helps create new tools and devices. These tools improve daily life and technology worldwide.
Quantum applications use the unique properties of particles. These properties include superposition and entanglement. They allow devices to perform better and solve complex problems. Let’s explore two key quantum applications: lasers and transistors, and quantum computing basics.
Lasers And Transistors
Lasers depend on quantum physics to produce light. They work by causing atoms to emit light in a controlled way. This process creates a strong, focused beam of light. Lasers power many devices like barcode scanners, printers, and medical tools.
Transistors are tiny switches used in electronics. Quantum physics explains how electrons move through materials. This knowledge helps build smaller, faster, and more efficient transistors. These transistors are the heart of computers and smartphones.
Quantum Computing Basics
Quantum computing uses quantum bits or qubits. Unlike regular bits, qubits can be both 0 and 1 at the same time. This allows quantum computers to process many calculations simultaneously.
Quantum computers solve problems faster than classical computers for some tasks. They may improve areas like cryptography, materials science, and artificial intelligence. Quantum computing is still developing but shows great promise for the future.
Frequently Asked Questions
What Are The Basic Principles Of Quantum Physics?
Quantum physics studies particles behaving as waves and particles simultaneously. It involves superposition, uncertainty, entanglement, and particle spin principles.
What Is Quantum Physics In Simple Terms?
Quantum physics studies tiny particles like atoms and electrons. It shows they act as waves, exist in multiple states, and connect mysteriously. This science explains how the smallest parts of nature behave, enabling technologies like lasers and computers.
What Are The Basic Fundamentals Of Quantum Physics?
Quantum physics studies particles at atomic and subatomic levels. Key fundamentals include wave-particle duality, superposition, uncertainty principle, entanglement, and spin. These principles explain how particles behave unpredictably and interact in complex ways, forming the basis for modern technologies like lasers and quantum computing.
What Are The 7 Biggest Unanswered Questions In Physics?
The seven biggest unanswered physics questions include dark matter, dark energy, quantum gravity, matter-antimatter imbalance, nature of time, unification of forces, and the measurement problem in quantum mechanics.
Conclusion
Quantum physics reveals a strange and fascinating world. Particles can be waves and exist in many states. We cannot know everything at once, thanks to uncertainty. This science changes how we see reality itself. Understanding basics helps us grasp complex ideas later.
Keep exploring, stay curious, and enjoy the journey.



No comments