Electron Configuration of Xenon
Have you ever wondered what makes xenon such a unique element? Understanding the electron configuration of xenon unlocks the secret to its chemical behavior and stability.
By knowing how its electrons are arranged, you gain insight into why xenon is so inert and how it interacts—or doesn’t—with other elements. You’ll discover a simple, step-by-step explanation of xenon’s electron configuration. Whether you’re a student, a chemistry enthusiast, or just curious, this guide will make complex concepts easy to grasp and help you master the topic quickly.
Ready to dive in and see what lies beneath xenon’s glowing surface? Let’s explore together!
Xenon Basics
Xenon is a noble gas found in group 18 of the periodic table. It is colorless, odorless, and very stable. Scientists use xenon in lighting and medical imaging.
Understanding xenon's electron configuration helps explain its chemical behavior. The arrangement of electrons defines how it interacts with other elements. This section breaks down the key facts about xenon's electrons.
What Is Xenon?
Xenon is a heavy, non-reactive gas. It is rare in the Earth's atmosphere, making up about 0.0000087% by volume. Despite its rarity, xenon has many practical uses.
Atomic Structure Of Xenon
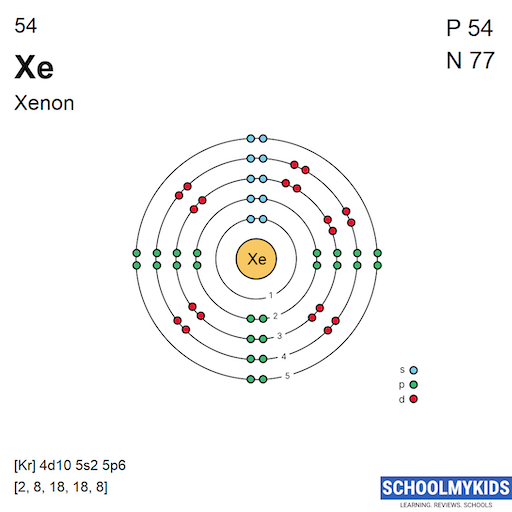
Xenon has 54 protons and 54 electrons. Its atomic number is 54, which means it has 54 electrons orbiting the nucleus. These electrons fill different energy levels or shells.
Why Electron Configuration Matters
The electron configuration shows how electrons are arranged in an atom. This arrangement controls xenon’s chemical properties. It explains why xenon is mostly inert and rarely forms compounds.
Atomic Structure
Xenon’s electron configuration shows how its electrons fill different energy levels. It has a full outer shell, making it stable and unreactive. This pattern explains xenon’s place in the noble gases group.
Xenon is a noble gas with a stable atomic structure. Its atoms have a complete set of electrons in energy levels.
This full electron arrangement makes xenon chemically inert. Understanding this structure helps explain its behavior in reactions.
Electron Shells And Energy Levels
Xenon’s electrons occupy five main shells. These shells surround the nucleus at different distances.
The shells fill in order of increasing energy. Xenon’s outer shell is full, creating stability.
Subshells And Orbitals
Each shell contains subshells labeled s, p, d, and f. These subshells hold electrons in orbitals.
Xenon’s electron configuration includes the s and p subshells filled on outer shells. This arrangement defines its chemical properties.
The electron configuration of xenon is written as 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶.
This notation shows the number of electrons in each subshell. It provides a clear map of electron placement in xenon.
Electron Shells
Xenon’s electron configuration shows how its electrons fill shells around the nucleus. It has five electron shells with a full outer shell. This stable arrangement makes xenon a noble gas with low reactivity.
Xenon is a noble gas with a full set of electron shells. These shells surround the nucleus in layers. Each shell holds a certain number of electrons. The shells fill in a specific order based on energy levels.
Understanding the electron shells helps explain xenon's chemical stability. The complete outer shell makes xenon mostly unreactive. This property is common among noble gases.
First Electron Shell (k-shell)
The first shell of xenon holds 2 electrons. It is the closest shell to the nucleus. These electrons have the lowest energy in xenon. This shell is always full in stable atoms.
Second Electron Shell (l-shell)
The second shell of xenon contains 8 electrons. It fills after the first shell is complete. These electrons have higher energy than the K-shell. The L-shell is also full in xenon.
Third Electron Shell (m-shell)
The third shell holds 18 electrons in xenon. It has more space than the first two shells. This shell includes both s, p, and d orbitals. Xenon's M-shell is completely filled, adding to its stability.
Fourth Electron Shell (n-shell)
Xenon's fourth shell has 8 electrons. This is the outermost shell in xenon. It contains the valence electrons. A full N-shell makes xenon chemically inert in most cases.
Orbital Filling Order
The orbital filling order describes how electrons arrange themselves in an atom's orbitals. This order follows specific rules to minimize energy and maintain stability. For xenon, understanding this order helps explain its chemical properties.
Electrons fill orbitals from lower to higher energy levels. Each orbital can hold a certain number of electrons. The sequence of filling is based on energy levels and sublevels.
Principle Of Aufbau
The Aufbau principle states electrons occupy the lowest energy orbitals first. This principle guides the step-by-step filling process. For xenon, electrons fill orbitals starting from 1s and move upward.
Order Of Sublevel Filling
Sublevels fill in the order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p. Xenon’s electrons follow this pattern precisely. This order reflects increasing energy and electron capacity.
Pauli Exclusion Principle And Hund’s Rule
The Pauli exclusion principle limits two electrons per orbital with opposite spins. Hund’s rule requires electrons to fill orbitals singly first. These rules ensure stable and balanced electron arrangement in xenon.
Xenon Electron Configuration
Xenon is a noble gas with a full set of electrons in its outer shell. Its electron configuration explains its chemical stability. Understanding the arrangement of electrons in xenon helps in studying its properties and reactions.
The electron configuration shows how electrons fill different energy levels and orbitals. Xenon’s position in the periodic table guides this arrangement. This knowledge is important for students and scientists alike.
Xenon Electron Configuration Basics
Xenon has 54 electrons. These electrons occupy energy levels around the nucleus. The configuration follows a specific order based on energy stability. Electrons fill orbitals starting from the lowest energy level.
Full Electron Configuration Of Xenon
The full electron configuration of xenon is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶. This shows how electrons are arranged in shells and subshells. Each number and letter indicate energy level and orbital type.
Xenon’s Electron Configuration And Chemical Properties
Xenon’s filled outer shell makes it very stable. This is why xenon rarely forms compounds. Its electron configuration explains its low reactivity. Understanding this helps predict xenon’s behavior in chemical reactions.
Credit: www.schoolmykids.com
Valence Electrons
Valence electrons are the outermost electrons of an atom. They determine how the atom reacts with other elements. In xenon, these electrons play a key role in its chemical behavior. Understanding xenon's valence electrons helps explain its properties.
What Are Valence Electrons In Xenon?
Xenon has eight valence electrons. These electrons are in the 5p and 5s orbitals. They are found in the outer shell, which is the fifth energy level. The full outer shell makes xenon very stable.
How To Identify Xenon's Valence Electrons
The electron configuration of xenon is [Kr] 4d10 5s2 5p6. The 5s and 5p electrons are its valence electrons. These are the electrons available for bonding or reactions.
Role Of Xenon's Valence Electrons In Chemical Reactions
Xenon rarely reacts because its valence shell is full. It is a noble gas with low chemical reactivity. However, under special conditions, xenon can form compounds using its valence electrons.
Noble Gas Configuration
Noble gas configuration helps simplify how we write electron arrangements. It uses the nearest noble gas to represent inner electrons. This method makes reading electron configurations easier and shorter.
Xenon is a noble gas itself. Its electron configuration shows a full set of shells. This stability is why xenon rarely reacts with other elements.
What Is Noble Gas Configuration?
Noble gas configuration uses a shorthand for electron arrangements. Instead of listing all electrons, we start with a noble gas symbol. Then, we add the extra electrons outside that noble gas core.
For example, instead of writing all electrons for xenon, we use the noble gas before it. This method saves time and space. It also helps focus on the outer electrons that matter most.
Noble Gas Configuration Of Xenon
Xenon's noble gas configuration is based on the previous noble gas, krypton. Krypton represents the first 36 electrons. Then, xenon has electrons filling the next shells.
The full electron configuration for xenon is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶.
In noble gas shorthand, it is written as: [Kr] 4d¹⁰ 5s² 5p⁶.
Why Use Noble Gas Configuration?
This configuration highlights valence electrons clearly. Valence electrons determine chemical properties. The inner electrons are hidden inside the noble gas symbol.
For xenon, this shows a full outer shell of electrons. That explains its lack of chemical reactivity. Noble gas configuration offers a neat way to understand electron setup.

Credit: www.youtube.com
Electron Configuration Notation
Electron configuration notation shows how electrons arrange in an atom. It uses numbers and letters to represent electron locations. This notation helps explain chemical behavior and bonding.
Each part of the notation tells about energy levels and sublevels. The numbers indicate the main energy level. Letters represent sublevels: s, p, d, and f. Superscript numbers show the number of electrons in each sublevel.
Understanding The Basic Structure
The electron configuration starts with the lowest energy level. Electrons fill sublevels in order of increasing energy. This order follows specific rules called the Aufbau principle.
The sequence for filling sublevels goes like this: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on. The notation lists sublevels with their electron counts step by step.
Notation For Xenon's Electron Configuration
Xenon has 54 electrons. Its electron configuration shows how these electrons fill up energy levels and sublevels. The notation is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶
This notation reflects the full shells and stability of the xenon atom.
Reading And Writing Electron Configuration
Start with the lowest level and move up. Write the energy level number, the sublevel letter, and the number of electrons as a superscript. Repeat until all electrons are accounted for.
This method helps visualize electron distribution clearly. It is useful for chemistry studies and predicting element behavior.
Applications In Chemistry
The electron configuration of xenon plays a key role in its chemical behavior and applications. Xenon’s full outer shell makes it mostly unreactive, but under certain conditions, it forms compounds. Understanding its electron arrangement helps chemists predict these reactions.
Xenon’s stable electron configuration allows it to be used in several chemical applications. These include lighting, medicine, and research. The unique properties of xenon arise from its electron setup.
Uses in Noble Gas ChemistryXenon is part of the noble gases group. Its electron configuration shows a complete valence shell. This limits its reactivity but enables the formation of rare compounds.
Chemists study xenon compounds to learn about chemical bonding. These compounds help explore reactions that involve full electron shells.
Xenon in Lighting and LasersXenon’s electron configuration allows it to emit bright light when energized. This makes it useful in lighting applications like flash lamps and arc lamps.
Its unique electron setup also helps produce laser light. Xenon lasers are used in surgery and scientific instruments.
Role in Medical ImagingXenon’s properties derived from its electron configuration make it safe for medical use. It is used as a contrast agent in imaging techniques like MRI.
Its inert nature reduces side effects, making it ideal for patient care. Xenon’s electron arrangement contributes to this safety.
Research in Chemical BondingScientists use xenon to study unusual chemical bonds. Its electron configuration allows the formation of bonds rarely seen in other elements.
This research expands knowledge about chemical reactions and bonding theories. Xenon’s electron setup is essential for these discoveries.
Comparison With Other Elements
Xenon’s electron configuration reveals its place among the noble gases. It has a full outer shell, making it stable and less reactive. Comparing xenon with other elements helps understand its unique properties and behavior.
Examining elements from different groups and periods shows how electron arrangements affect chemical traits. Let’s explore how xenon’s electron configuration differs from some common elements.
Xenon Vs. Argon
Argon has fewer electrons than xenon. Its configuration ends at 3p6, while xenon ends at 5p6. Both have full outer shells. Argon is in period 3; xenon is in period 5. This difference gives xenon a larger atomic size and more electron shells.
Xenon’s larger size affects its ability to form compounds unlike argon, which is mostly inert. The extra shells in xenon allow more complex interactions with other elements.
Xenon Vs. Calcium
Calcium is an alkaline earth metal with a different electron setup. It ends at 4s2, lacking a full p orbital. Calcium easily loses electrons to form positive ions. Xenon, with a full p orbital, rarely loses or gains electrons.
This contrast shows why calcium is reactive and xenon is stable. Calcium’s electrons are more loosely held, while xenon’s electrons are tightly bound, preventing easy reactions.
Xenon Vs. Cobalt
Cobalt is a transition metal with electrons in the 3d orbital. Its configuration is more complex than xenon’s. Cobalt’s partially filled d orbitals allow it to form various oxidation states. Xenon’s full outer shell limits its oxidation states.
These differences explain why cobalt participates in many reactions and xenon mostly stays inert. Cobalt’s electron arrangement supports magnetism and conductivity, unlike xenon.

Credit: www.youtube.com
Frequently Asked Questions
How To Write The Electron Configuration Of Xenon?
Write xenon's electron configuration by filling orbitals in order: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶. This represents all 54 electrons in xenon.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element Zinc (Zn) with atomic number 30.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell can be either 2 8 8 or 2 8 18, depending on the element's period. Early elements fill shells as 2 8 8, while heavier elements may fill the third shell with 18 electrons.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 belongs to the element Cobalt (Co), atomic number 27.
Conclusion
Understanding the electron configuration of xenon helps explain its chemical behavior. Xenon has a full outer shell, making it stable and less reactive. This noble gas plays a unique role in many scientific fields. Remember, its electron arrangement follows a clear pattern.
Learning this pattern can make chemistry easier and more interesting. Keep exploring other elements to see how electron configurations vary. Each element tells a different story through its electrons. Chemistry becomes simpler when you grasp these basic ideas.



No comments