Electron Configuration of Tellurium
Are you curious about what makes tellurium unique in the world of chemistry? Understanding the electron configuration of tellurium can unlock insights into its chemical behavior, reactivity, and place in the periodic table.
You’ll discover how the electrons are arranged around tellurium’s nucleus and why this arrangement matters for everything from bonding to material properties. Whether you’re a student, a chemistry enthusiast, or just someone eager to learn more about this fascinating element, this clear and simple guide will help you grasp the essentials quickly.
Keep reading to uncover the secrets hidden in tellurium’s electron configuration and how it influences its role in science and industry.
Basic Properties Of Tellurium
Tellurium is a unique element with interesting physical and chemical properties. It sits in the periodic table between metals and nonmetals, giving it traits of both categories. This makes tellurium important in various scientific and industrial fields.
Its atomic structure influences how it reacts and bonds with other elements. Understanding tellurium's basic properties helps explain its behavior in different environments and applications.
Atomic Number And Symbol
Tellurium has the atomic number 52. Its chemical symbol is Te. This number shows it has 52 protons and, in a neutral atom, 52 electrons.
Physical Appearance
Tellurium is a brittle, silvery-white solid. It has a metallic luster but is not a true metal. It breaks easily and forms crystals with a unique shape.
Atomic Mass
The atomic mass of tellurium is about 127.6 atomic mass units. This value reflects the average weight of its naturally occurring isotopes.
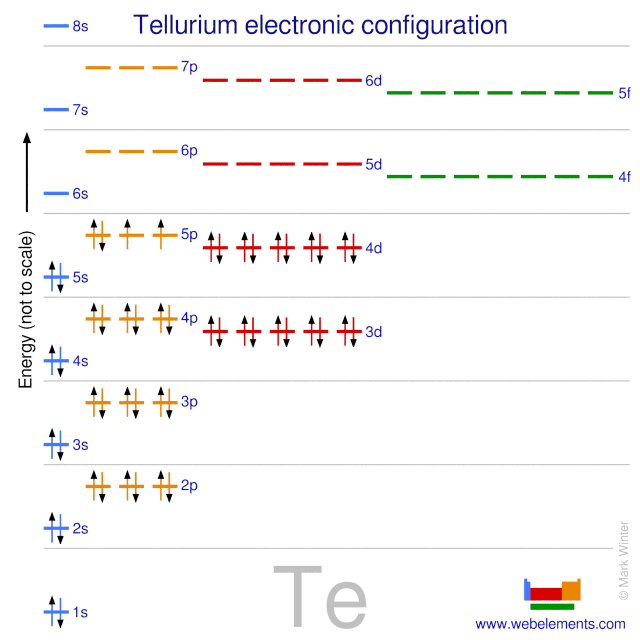
Electron Configuration
Tellurium’s electron configuration is key to its properties. Its full electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁴. This shows how electrons fill its atomic orbitals.
State At Room Temperature
Tellurium is solid at room temperature. It melts at 449.5 degrees Celsius and boils at 988 degrees Celsius.
Electronegativity And Reactivity
Tellurium has moderate electronegativity, about 2.1 on the Pauling scale. It reacts slowly with oxygen and other elements, forming compounds like tellurides.
Credit: winter.group.shef.ac.uk
Atomic Structure And Number
Understanding the atomic structure and number of tellurium is key to grasping its electron configuration. The atomic number defines the number of protons in the nucleus. It also tells the number of electrons in a neutral atom. These electrons arrange themselves in shells around the nucleus.
Tellurium has unique properties because of its atomic structure. Its electrons fill specific energy levels or orbitals. This arrangement impacts how tellurium behaves chemically and physically.
Atomic Number Of Tellurium
Tellurium has an atomic number of 52. This means it contains 52 protons in its nucleus. In a neutral atom, it also has 52 electrons orbiting the nucleus. The atomic number places tellurium in group 16 of the periodic table.
Electron Shells And Orbitals
Electrons in tellurium fill several shells and subshells. The first shell holds 2 electrons, the second 8, and the third 18. The outermost shell contains 6 electrons. These electrons occupy s, p, and d orbitals.
Effect On Chemical Properties
The electron arrangement affects tellurium's chemical behavior. Its 6 valence electrons make it reactive with metals. Tellurium often forms compounds by gaining or sharing electrons. This structure also influences its conductivity and bonding patterns.
Full Electron Configuration
Tellurium has 52 electrons arranged in energy levels around its nucleus. Its full electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁴. This setup explains its chemical behavior and placement in the periodic table.
The full electron configuration of Tellurium shows how its electrons fill each atomic orbital. It details the exact distribution of all electrons in the atom. This helps us understand its chemical behavior and physical properties.
Tellurium has an atomic number of 52, meaning it has 52 electrons in a neutral state. These electrons occupy orbitals in a specific order based on energy levels.
Electron Distribution In Energy Levels
The electrons start filling the lowest energy orbitals first. For Tellurium, the 1s orbital fills first, followed by 2s and 2p orbitals. This pattern continues through higher energy levels.
The configuration includes s, p, and d orbitals. Each orbital type holds a specific number of electrons. This arrangement affects Tellurium’s reactivity and bonding.
Complete Electron Configuration Of Tellurium
The full electron configuration for Tellurium is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁴
This shows all the electrons from the first to the fifth energy level. The superscripts indicate the number of electrons in each subshell.
Importance Of Full Electron Configuration
Knowing the full electron configuration helps predict Tellurium's chemical properties. It reveals the number of valence electrons, which participate in bonding.
This information is crucial for students and scientists studying element behavior. It also aids in understanding periodic trends and element classification.
Credit: www.chegg.com
Abbreviated Electron Configuration
The abbreviated electron configuration simplifies the full configuration of an element. It uses the nearest noble gas to shorten the notation. This method saves space and makes the configuration easier to read. It also highlights the valence electrons that participate in chemical reactions.
For tellurium, the abbreviated electron configuration begins with the symbol of the noble gas krypton. Krypton represents the core electrons that fill inner shells. Then, the configuration continues with the electrons beyond krypton. This shows the outer electrons important for bonding and properties.
Noble Gas Core For Tellurium
Krypton is the noble gas before tellurium in the periodic table. Its electron configuration is [Kr]. This symbol replaces all the electrons in tellurium up to krypton. It includes 36 electrons in the first four shells. Using [Kr] simplifies the configuration and focuses on the outer electrons.
Electrons Beyond Krypton
After [Kr], tellurium has additional electrons in the 4d and 5p orbitals. The configuration continues as 4d10 5s2 5p4. These electrons control tellurium’s chemical behavior. The 5p4 means tellurium has four electrons in the 5p subshell. These are the valence electrons that interact with other atoms.
Abbreviated Configuration Of Tellurium
The full abbreviated electron configuration of tellurium is [Kr] 4d10 5s2 5p4. This notation is concise and clear. It shows the filled d subshell and the valence s and p electrons. The simplicity helps students and chemists quickly understand tellurium’s electron structure.
Electron Shells And Subshells
Understanding the electron shells and subshells of tellurium is key to grasping its chemical behavior. Electrons in an atom arrange themselves in layers called shells. Each shell contains smaller groups known as subshells. These shells and subshells follow specific energy levels.
Tellurium's electron configuration reveals how its electrons fill these shells and subshells. This arrangement affects its properties and how it bonds with other elements. Let’s explore these shells and subshells in detail.
Electron Shells Of Tellurium
Tellurium has 52 electrons distributed across several shells. These shells are numbered 1 to 5, starting from the closest to the nucleus. The first shell holds 2 electrons. The second shell holds 8 electrons. The third shell contains 18 electrons. The fourth shell has 18 electrons. The fifth shell holds the remaining 6 electrons. These layers create the atom’s structure.
Subshells Within Each Shell
Each shell divides into subshells named s, p, d, and f. Subshells differ by shape and energy. The s subshell holds up to 2 electrons. The p subshell holds up to 6 electrons. The d subshell holds up to 10 electrons. The f subshell holds up to 14 electrons. Tellurium's electrons fill these subshells in order of increasing energy.
Filling Order Of Tellurium’s Subshells
The electrons fill subshells starting with the lowest energy level. For tellurium, the order begins with 1s, then 2s and 2p, followed by 3s, 3p, and 4s. Next come 3d, 4p, 5s, and 4d subshells. Finally, electrons fill the 5p subshell. This sequence determines tellurium’s full electron configuration and influences its chemical traits.
Valence Electrons In Tellurium
Valence electrons play a key role in defining the chemical behavior of tellurium. These electrons are found in the outermost shell of the atom. They determine how tellurium bonds with other elements and its reactivity.
Tellurium has six valence electrons. These electrons are located in the 5th energy level. Understanding these electrons helps explain tellurium's properties and its place in the periodic table.
Electron Shells And Valence Electrons
Tellurium's electrons are arranged in shells around its nucleus. The inner shells hold electrons tightly. The outer shell contains the valence electrons. For tellurium, the outer shell is the 5th shell with six electrons.
How Valence Electrons Affect Tellurium's Reactivity
Tellurium tends to gain or share two electrons to fill its outer shell. This makes it reactive with metals and nonmetals. Its six valence electrons explain why it often forms -2 oxidation states.
Valence Electrons And Chemical Bonding
The six valence electrons allow tellurium to form covalent bonds. It can also participate in metallic bonding in certain compounds. The number of valence electrons influences the type and strength of bonds formed.
Tellurium’s Position In The Periodic Table
Tellurium is a unique element with an interesting spot in the periodic table. It belongs to the group of elements called metalloids. Metalloids share properties of both metals and non-metals. This makes tellurium important in many scientific and industrial fields.
Its position affects its chemical behavior and the way its electrons are arranged. Understanding where tellurium sits on the table helps explain its electron configuration and reactivity.
Group And Period
Tellurium is located in group 16 of the periodic table. This group is also called the chalcogens. Elements here often form compounds with oxygen. Tellurium is in period 5. This means it has five electron shells around its nucleus.
Block And Classification
Tellurium belongs to the p-block of elements. The p-block contains elements with electrons filling p orbitals. It is classified as a metalloid because its properties are between metals and non-metals. This unique nature influences its electron arrangement.
Atomic Number And Symbol
Tellurium’s atomic number is 52. This means it has 52 protons in its nucleus and 52 electrons around it. The chemical symbol for tellurium is Te. This symbol is used worldwide to represent the element in scientific writing.
Comparison With Neighboring Elements
Comparing the electron configuration of tellurium with its neighboring elements reveals interesting patterns. These patterns explain differences in chemical behavior and physical properties. The elements around tellurium share some electron arrangement traits but also show clear differences.
Tellurium has the atomic number 52. It lies between antimony (Sb, atomic number 51) and iodine (I, atomic number 53). Examining their electron configurations helps understand their similarities and differences.
Electron Configuration Of Tellurium
Tellurium's full electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁴. It ends with 5p⁴, indicating four electrons in the outermost p orbital.
This arrangement gives tellurium its unique chemical characteristics, especially in forming compounds.
Comparison With Antimony (sb)
Antimony has atomic number 51. Its electron configuration ends with 5p³. This means it has one less electron in the 5p orbital than tellurium.
Because of this, antimony acts as a metalloid with different bonding behavior. Tellurium is more likely to gain or share electrons due to its higher p-electron count.
Comparison With Iodine (i)
Iodine has atomic number 53. Its electron configuration ends with 5p⁵, which is one more electron than tellurium.
This gives iodine a stronger tendency to gain an electron and form negative ions. Tellurium, with 5p⁴, shows more metallic properties than iodine.
Trends In The Periodic Table
The elements in this part of the table gradually fill the 5p orbital. This filling changes their properties from metallic to non-metallic.
Tellurium stands in the middle, showing a mix of metal and non-metal traits. Its electron configuration plays a key role in this behavior.
Chemical Reactivity And Electron Configuration
The chemical reactivity of tellurium is closely linked to its electron configuration. This configuration explains how tellurium atoms interact with other elements. Understanding this helps predict tellurium’s behavior in chemical reactions.
Tellurium has electrons arranged in specific energy levels and sublevels. These arrangements determine how easily tellurium gains, loses, or shares electrons. Reactivity depends on these electron movements.
Chemical Properties Influenced By Electron Arrangement
Tellurium’s outer electrons are in the 5p sublevel. These valence electrons mainly control its chemical properties. Tellurium tends to gain or share two electrons to complete its outer shell. This leads to common oxidation states of -2 and +4.
Because of its electron configuration, tellurium acts like a metalloid. It can form both ionic and covalent bonds. This dual nature affects how it reacts with metals and nonmetals.
Electron Configuration And Reactivity Trends
Tellurium’s electron configuration is [Kr] 4d10 5s2 5p4. The four electrons in the 5p orbitals are key to its reactivity. These electrons seek to pair up, making tellurium moderately reactive.
Compared to other group 16 elements, tellurium is less reactive than sulfur but more than polonium. Its reactivity also varies with physical state and environment.
Impact On Compound Formation
Tellurium forms a variety of compounds due to its electron configuration. It reacts with oxygen to form tellurium dioxide (TeO2). In metal alloys, it improves hardness and corrosion resistance.
Its ability to share or gain electrons allows tellurium to participate in diverse chemical reactions. This versatility is a direct result of its electron structure.

Credit: www.youtube.com
Applications Influenced By Electron Structure
The electron configuration of tellurium plays a key role in its applications. The way electrons are arranged affects tellurium’s chemical and physical properties. These properties determine how tellurium interacts in different fields. Understanding this helps explain why tellurium is valuable in various industries.
Tellurium's unique electron structure influences its behavior in electronics, materials science, and chemistry. Each application relies on the element's ability to gain, lose, or share electrons. This makes tellurium useful in many technological and industrial processes.
Electrical Conductivity In Semiconductors
Tellurium’s electron arrangement allows it to act as a semiconductor. It can conduct electricity under certain conditions. This property makes it important in electronic devices. Tellurium is used in alloys and compounds that improve semiconductor performance. Its electron configuration helps control electrical flow efficiently.
Role In Thermoelectric Materials
The electron structure of tellurium supports its use in thermoelectric materials. These materials convert heat into electricity. Tellurium's electron arrangement helps balance electrical conductivity with low thermal conductivity. This balance is essential for good thermoelectric efficiency. Devices using tellurium can power small gadgets or recover waste heat.
Chemical Reactivity And Catalyst Function
Tellurium’s outer electrons influence its chemical reactivity. It can form compounds with different elements easily. This makes tellurium useful as a catalyst in some chemical reactions. Its electron configuration allows it to interact selectively with other substances. Catalysts containing tellurium improve reaction rates and product yields.
Applications In Optical Devices
Tellurium’s electron structure affects its optical properties. It can absorb and emit light in specific ways. This feature is useful in optical storage and sensors. Tellurium compounds help create materials with unique light responses. These materials find use in lasers and other photonic devices.
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 corresponds to calcium (Ca), atomic number 20.
What Element Has The Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d10?
The element with the electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d10 is Silver (Ag).
Which Atom Only Symbol Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d2 Blank 1?
The atom with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d2 5p1 is scandium (Sc).
What Is 1s2 2s2 2p6 3s2 3p6 4s2 3d8?
The notation 1s2 2s2 2p6 3s2 3p6 4s2 3d8 represents the electron configuration of a nickel (Ni) atom. It shows the distribution of electrons in atomic orbitals.
Conclusion
Tellurium’s electron configuration reveals its chemical behavior clearly. It has 52 electrons arranged in specific orbitals. This arrangement helps explain its place in the periodic table. Knowing the configuration aids understanding of its bonding and properties. Simple patterns emerge when studying tellurium’s electrons.
This knowledge supports learning about other elements too. Understanding electron configurations makes chemistry easier and more fun. Keep exploring to deepen your grasp of atomic structures.



No comments