Electron Configuration of Thulium
Have you ever wondered what makes thulium unique among the elements? Understanding the electron configuration of thulium can unlock insights into its chemical behavior and practical uses.
By diving into how its electrons are arranged, you’ll gain a clearer picture of why thulium behaves the way it does in reactions and materials. Whether you’re a student, a chemistry enthusiast, or just curious, this guide will simplify the complex world of electron arrangements so you can easily grasp thulium’s atomic secrets.
Keep reading to discover the key to mastering thulium’s electron configuration and how it impacts its role in science and technology.
Basic Properties
Understanding the basic properties of thulium helps us grasp its unique electron arrangement. Thulium is a rare earth metal with distinct features. These features influence its chemical behavior and physical characteristics.
The electron configuration reveals the way thulium's electrons are organized around its nucleus. This arrangement affects how thulium interacts with other elements and compounds. The study of these properties forms the foundation for deeper knowledge about this element.
Atomic Number And Symbol
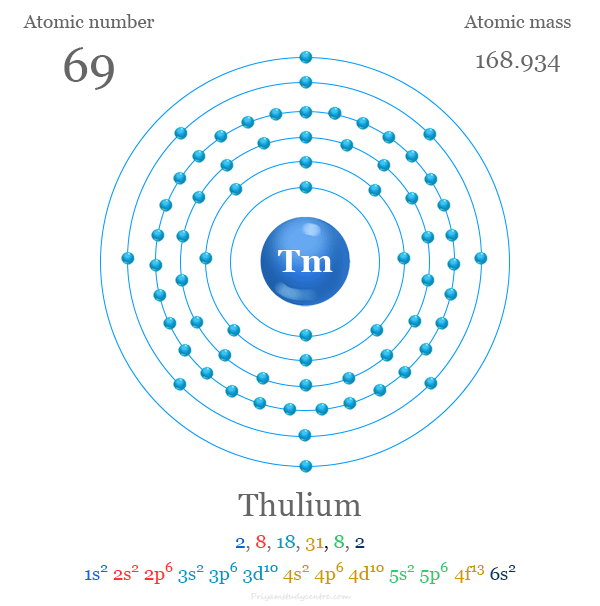
Thulium has the atomic number 69. Its chemical symbol is Tm. This number shows how many protons are in the nucleus. It also equals the total electrons in a neutral atom.
Electron Shells And Orbitals
Thulium's electrons fill several shells and orbitals. The outermost electrons play a key role in bonding. The configuration ends with the 4f orbital being partially filled.
Ground State Electron Configuration
The ground state configuration of thulium is [Xe] 4f13 6s2. This means it has a xenon core with 13 electrons in the 4f sublevel and 2 electrons in the 6s sublevel. This setup defines its chemical properties.
Thulium has three valence electrons. These electrons are in the 4f and 6s orbitals. They determine how thulium bonds with other atoms and its reactivity.
Credit: jamesmaas.com
Atomic Structure
Thulium’s electron configuration shows how its electrons fill orbitals. It follows the pattern of filling 4f, 5d, and 6s shells. This arrangement helps explain its chemical properties and place in the periodic table.
The atomic structure of thulium defines its chemical and physical traits. Thulium is a lanthanide with atomic number 69. This means it has 69 protons in its nucleus. Electrons orbit the nucleus in shells and subshells. Their arrangement influences how thulium behaves in reactions and bonding.
Understanding thulium’s atomic structure helps explain its place in the periodic table. It also clarifies its magnetic and spectral properties. The structure consists of electrons filling energy levels in a specific order. This order follows the principles of quantum mechanics.
Electron Shells And Subshells
Thulium’s electrons fill shells labeled 1 to 6 and subshells s, p, d, and f. The inner shells hold fewer electrons, while outer shells hold more. The 4f subshell, part of the f-block elements, is especially important for thulium. It holds 13 electrons, giving thulium unique characteristics.
Electron Configuration Of Thulium
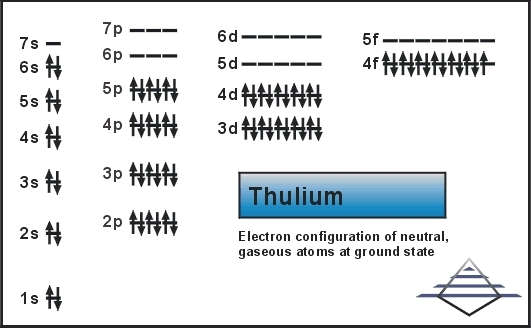
The full electron configuration of thulium is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹³ 6s². This shows how electrons fill each subshell in order. The 4f¹³ indicates thirteen electrons in the 4f subshell. The outermost electrons, 6s², affect thulium’s reactivity and bonding.
Noble Gas Notation
Using noble gas notation, thulium’s electron configuration is simplified to [Xe] 4f¹³ 6s². This means it follows xenon’s electron arrangement plus thirteen 4f electrons and two 6s electrons. This shorthand is easier to read and remember. It highlights the valence electrons that matter most for chemistry.
Electron Shells
The electron shells of an atom show how electrons are arranged around the nucleus.
Each shell holds a certain number of electrons and defines the atom's energy levels.
Understanding the electron shells of thulium helps explain its chemical behavior.
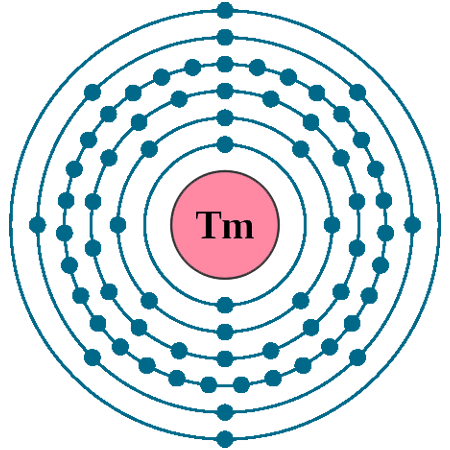
Thulium’s Electron Shell Structure
Thulium has 69 electrons arranged in several shells.
The shells are filled in order of increasing energy.
Its electron shells fill up to the 6th energy level with the outermost shell containing few electrons.
Distribution Of Electrons In Shells
Thulium's electron shell distribution is 2, 8, 18, 32, 8, 1.
The first shell has 2 electrons, the second has 8.
More electrons are packed in the middle shells before reaching the outer shell.
Significance Of Electron Shells In Thulium
The electron shell arrangement affects thulium's reactivity and bonding.
The shells also influence its magnetic and optical properties.
These shells explain why thulium behaves like other lanthanides.
Orbital Filling
Thulium’s electrons fill orbitals in a specific order following the Aufbau principle. Its electron configuration shows how electrons occupy shells and subshells step by step. This pattern explains thulium’s chemical behavior and properties clearly.
Understanding The Orbital Filling Of Thulium
Orbital filling describes how electrons fill the atomic orbitals of an element. Each orbital can hold a limited number of electrons. Electrons fill lower energy orbitals first before moving to higher ones.
For thulium, the electrons fill orbitals in a specific order. This order follows the Aufbau principle, which helps predict the electron arrangement in atoms.
The Electron Shells And Subshells In Thulium
Thulium has 69 electrons. These electrons occupy different shells and subshells. The main shells are numbered 1, 2, 3, and so on. Each shell contains one or more subshells: s, p, d, and f.
In thulium, electrons fill the 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, and 5d orbitals. The 4f subshell is very important for this element.
Filling The 4f Subshell In Thulium
The 4f subshell can hold up to 14 electrons. Thulium has 13 electrons in the 4f subshell. This makes thulium part of the lanthanide series, known for filling the 4f orbitals.
These 4f electrons influence thulium’s chemical and physical properties. They cause unique magnetic and spectral behaviors.
Orbital Filling Order For Thulium
Thulium's electrons fill orbitals in this sequence: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹³ 5d⁰ 6p⁰.
This order respects the energy levels of orbitals. Electrons fill the lowest energy orbitals first, then proceed to higher levels.
Full Electron Configuration
The full electron configuration shows the arrangement of all electrons in thulium's atoms. It details the distribution of electrons across different energy levels and orbitals. This helps us understand thulium’s chemical behavior and physical properties.
Thulium has 69 electrons. These electrons fill orbitals in a specific order based on energy levels. The configuration follows the principles of quantum mechanics and the Aufbau principle.
Ground State Electron Configuration Of Thulium
The ground state configuration of thulium is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f¹³ 5d⁰ 6s²
This shows thulium’s electrons filling the 1s orbital first, then moving outward to higher orbitals. The 4f orbital has 13 electrons, which is key to thulium’s properties.
Electron Configuration By Energy Levels
Electrons fill lower energy orbitals before higher ones. The sequence moves from 1s to 6s orbitals. The 4f orbital fills after the 6s orbital for thulium. This filling order is important for lanthanides like thulium.
Importance Of The 4f Orbital In Thulium
The 4f orbital holds 13 electrons in thulium. These electrons influence magnetic and spectral properties. The 4f electrons are shielded by outer electrons, affecting chemical reactions.
This orbital’s unique filling pattern explains thulium’s place in the lanthanide series. It also impacts how thulium interacts with other elements.
Noble Gas Shorthand
The noble gas shorthand is a simple way to write electron configurations. It uses the electron configuration of the nearest noble gas before the element. This method makes long configurations shorter and easier to read.
For thulium, the shorthand starts with the noble gas xenon (Xe). Xenon has a full set of electrons that match thulium’s inner shells. After xenon, only the electrons outside this core are shown.
Why Use Noble Gas Shorthand?
Writing full electron configurations can be long and complex. Noble gas shorthand saves space and reduces errors. It helps students and chemists quickly identify an element’s valence electrons. This method highlights the outer electrons that determine chemical behavior.
Noble Gas Shorthand For Thulium
Thulium’s atomic number is 69. The noble gas before it is xenon with atomic number 54. The shorthand starts with [Xe]. Then the electrons after xenon are added.
The configuration after xenon is: 4f13 6s2. So, thulium’s noble gas shorthand is [Xe] 4f13 6s2.
Benefits Of Using Noble Gas Shorthand For Lanthanides
Lanthanides like thulium have many electrons in inner shells. Writing full configurations can be confusing. The shorthand helps focus on the important 4f and 6s electrons. It makes comparing lanthanides easier. This saves time in chemical calculations and studies.
Valence Electrons
Valence electrons are the electrons found in the outermost shell of an atom. They play a key role in chemical reactions and bonding. Understanding valence electrons helps explain how elements interact with each other.
For thulium, these electrons determine its reactivity and placement in the periodic table. Thulium is a lanthanide, so its valence electrons involve the 4f and 6s orbitals. These electrons influence its unique chemical and physical properties.
Valence Electron Configuration Of Thulium
Thulium has an atomic number of 69. Its valence electrons mainly occupy the 4f and 6s subshells. The electron configuration for valence electrons is 4f13 6s2. This means 13 electrons fill the 4f orbital, and 2 are in the 6s orbital.
Importance Of 4f Electrons
The 4f electrons in thulium are shielded by outer electrons. They do not participate easily in bonding. Still, they affect magnetic and optical properties of thulium compounds. These electrons give thulium its distinct characteristics among lanthanides.
Role Of 6s Electrons
The 6s electrons are more exposed and can be lost or shared in reactions. They are the main valence electrons involved in chemical bonding. These electrons allow thulium to form different ions and compounds.
Inert Pair Effect
The inert pair effect describes the tendency of the outermost s electrons in heavy elements to remain non-ionized or unshared in compounds. This effect influences the chemical behavior of many elements, especially those in the lower periods of the periodic table. It makes the s electrons less available for bonding, causing variations in oxidation states.
For thulium, a lanthanide element, the inert pair effect plays a subtle but important role. Thulium's electron configuration shows a filled 6s orbital, but these electrons often do not participate fully in bonding. Understanding this helps explain thulium’s common +3 oxidation state and its chemical stability.
What Causes The Inert Pair Effect?
The inert pair effect arises from poor shielding of nuclear charge by inner d and f electrons. This causes the outer s electrons to be held more tightly by the nucleus. As a result, these s electrons resist ionization or sharing in bonds.
Inert Pair Effect In Thulium's Electron Configuration
Thulium's electron configuration ends in 4f13 6s2. The 6s electrons form the inert pair. They are less reactive due to strong attraction from the nucleus and shielding by f electrons. This explains why thulium commonly forms +3 ions by losing the 4f and one 6s electron.
Impact On Thulium's Chemical Properties
The inert pair effect limits thulium to fewer oxidation states, mainly +3. This leads to predictable chemistry and stable compounds. It also influences thulium’s role in materials and technology, where consistent valence is crucial.
Comparison With Lanthanides
The electron configuration of thulium shows unique traits among lanthanides. Comparing it with other lanthanides helps understand its chemical behavior and position in the periodic table. The lanthanides share similarities but also have distinct differences in their electron arrangements.
Thulium’s electron configuration reflects its place near the end of the lanthanide series. This affects its properties, such as magnetism and reactivity. Examining these differences clarifies how thulium fits within the group.
Electron Shell Structure Differences
Thulium has the electron configuration [Xe] 4f13 6s2. This means it has 13 electrons in the 4f orbital. Other lanthanides have varying numbers of 4f electrons, ranging from 1 to 14. The gradual filling of 4f orbitals causes slight changes in atomic size.
Compared to earlier lanthanides, thulium’s 4f orbitals are more filled. This leads to a smaller atomic radius because of stronger attraction between electrons and the nucleus. This trend is called the lanthanide contraction.
Valence Electron Similarities
Lanthanides usually have two valence electrons in the 6s orbital. Thulium follows this pattern. These valence electrons play a key role in chemical bonding. The 4f electrons are less involved in bonding but influence the element’s magnetic and spectral properties.
Because thulium’s valence electrons are similar to other lanthanides, its chemical reactions often resemble theirs. Yet, the nearly full 4f shell gives thulium some unique magnetic behaviors.
Magnetic And Spectral Properties
Thulium’s electron configuration leads to specific magnetic properties. The unpaired 4f electrons cause paramagnetism. Other lanthanides also show paramagnetism, but the strength varies with 4f electron count.
Spectral lines of thulium differ from other lanthanides due to its electron arrangement. These lines help identify thulium in mixtures and applications. The nearly complete 4f shell creates distinct energy transitions.
Credit: pilgaardelements.com
Applications And Uses
Thulium has several important uses in science and technology. Its unique electron configuration helps make these applications possible. The element's properties suit various fields well.
People use thulium in areas such as medicine, electronics, and research. Its specific traits allow for precise and useful functions. Below are key uses of thulium in modern industries.
Medical Applications Of Thulium
Thulium ions are valuable in laser technology for medicine. These lasers treat eye problems and tumors with high accuracy. Thulium-doped lasers provide controlled energy release. This makes them safer and more effective.
Thulium is also used in portable X-ray devices. These devices help doctors perform quick scans outside hospitals. The element’s electron setup helps produce clear images.
Role In Electronic Devices
Thulium is part of some special electronic components. It improves the performance of materials used in magnets and batteries. This element helps enhance energy storage and transfer.
It also plays a role in improving the brightness of certain display screens. Thulium’s electrons emit light when energized, making screens clearer and sharper.
Scientific Research Uses
Scientists study thulium for its magnetic and optical properties. Its electron configuration makes it useful for experiments in physics and chemistry. Research on thulium helps develop new materials and technologies.
The element is also used in calibration standards for instruments. This ensures accuracy in various measurements across scientific fields.
Credit: www.priyamstudycentre.com
Frequently Asked Questions
What Is The Electronic Configuration Of Thulium?
The electronic configuration of thulium (Tm) is [Xe] 4f¹³ 6s². It belongs to the lanthanide series.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element zinc (Zn), atomic number 30.
Is Electron Shell 2 8 8 Or 2 8 18?
Electron shells usually follow 2, 8, 18 for the first three levels. The second shell holds 8 electrons, and the third can hold up to 18. So, electron shells can be 2, 8, 8 or 2, 8, 18 depending on the element’s electron configuration.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn). It has atomic number 25.
Conclusion
Understanding thulium’s electron configuration helps explain its chemical behavior. Its unique arrangement affects how it bonds and reacts. This knowledge supports studies in chemistry and material science. Remember, thulium’s electrons fill specific orbitals in a set order. This pattern influences its place in the periodic table.
Knowing this makes learning about thulium easier and clearer. Keep exploring to see how electron configurations shape elements around us.



No comments