Electron Configuration of Barium
If you want to truly understand barium and its role in chemistry, knowing its electron configuration is key. This simple arrangement of electrons shapes how barium behaves, reacts, and interacts with other elements.
Whether you're a student tackling chemistry homework, a science enthusiast curious about the periodic table, or someone preparing for exams, grasping the electron configuration of barium will give you a clear edge. Keep reading, and you’ll discover an easy-to-follow breakdown that makes this important concept straightforward and memorable.
Your chemistry knowledge is about to get a powerful boost!
Atomic Structure Of Barium
Barium has 56 electrons arranged to fill energy levels in a specific order. Its electron configuration ends with 6s², showing two electrons in the outermost shell. This setup helps explain barium’s chemical behavior and placement in the periodic table.
Barium is a chemical element with the symbol Ba and atomic number 56. It belongs to the alkaline earth metals group in the periodic table. The atomic structure of barium defines its chemical behavior and physical properties.
Understanding the atomic structure starts with the arrangement of electrons in various energy levels or shells. These electrons orbit the nucleus, which contains protons and neutrons. The way electrons fill these shells follows specific rules based on energy and stability.
Electron Shells And Energy Levels
Barium has 56 electrons distributed in different shells around the nucleus. The shells are labeled as K, L, M, N, O, and so on, starting from the closest to the nucleus. Each shell holds a specific number of electrons, filling from inner to outer shells.
In barium, the first five shells (K to O) are fully or partially filled. The outermost shell contains electrons that participate in chemical reactions. These electrons are called valence electrons.
Subshells And Orbital Filling
Electron shells contain subshells named s, p, d, and f. Each subshell has orbitals that hold electrons in pairs. Electrons fill these orbitals following the Aufbau principle, which means filling lower energy levels first.
For barium, the electron configuration shows a complete filling of inner subshells and two electrons in the outer 6s subshell. This configuration contributes to barium's reactivity and bonding characteristics.
Nuclear Composition And Stability
The nucleus of barium contains 56 protons, giving it a positive charge equal to the atomic number. Neutrons add mass and contribute to nuclear stability. The most common isotope of barium has 81 neutrons.
The balance between protons and neutrons ensures that the nucleus remains stable under normal conditions. This stability affects the element's physical traits and its presence in nature.

Credit: www.youtube.com
Electron Shell Arrangement
Barium’s electron configuration shows how its electrons fill energy levels. It ends with 6s² after the noble gas xenon core. This arrangement helps explain barium’s chemical properties and behavior in reactions.
Barium is a chemical element with atomic number 56. This number tells us it has 56 electrons. These electrons are arranged in shells around the nucleus.
Electron shells are layers that hold electrons at different energy levels. Each shell can hold a certain number of electrons. The arrangement of these electrons affects the element's chemical behavior.
Distribution Of Electrons In Barium Shells
Barium's electrons fill shells in order from the closest to the farthest from the nucleus. The first shell holds 2 electrons. The second shell can hold up to 8 electrons. The third shell also holds 18 electrons. The fourth shell holds 18 electrons as well. The fifth shell holds 8 electrons. The sixth shell holds 2 electrons for barium.
Energy Levels And Stability
Electrons fill lower energy levels before higher ones. This makes the atom more stable. Barium’s outermost shell has 2 electrons in the sixth shell. These electrons play a key role in chemical reactions and bonding.
Electron Configuration Notation For Barium
The electron configuration shows the exact arrangement of electrons. For barium, it is written as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s². This notation helps understand how electrons occupy shells and subshells.
Orbital Filling Sequence
The orbital filling sequence explains how electrons occupy the orbitals in an atom. Electrons fill orbitals in a specific order to achieve the lowest energy state. This order follows the Aufbau principle, which guides the arrangement of electrons from lower to higher energy orbitals.
Understanding this sequence helps in writing the electron configuration of barium accurately. Barium has 56 electrons, and these electrons fill orbitals step by step according to their energy levels. The process involves filling s, p, d, and f orbitals in the correct order.
What Is Orbital Filling Sequence?
Orbital filling sequence shows the order electrons fill atomic orbitals. Electrons first fill the lowest energy orbitals before moving to higher ones. This order is not always straightforward but follows specific rules based on energy levels.
Order Of Subshells In Barium
Barium’s electrons fill orbitals in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s. The 6s orbital fills last for barium’s 56 electrons. This sequence matches the increasing energy of the orbitals.
Applying The Aufbau Principle
The Aufbau principle states electrons fill the lowest energy orbitals first. For barium, the 1s orbital fills before 2s, and 4s fills before 3d. This ensures the atom’s stability by lowering its energy.
Electron Configuration Of Barium
Barium’s full electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s². The two electrons in the 6s orbital give barium its chemical properties as an alkaline earth metal.
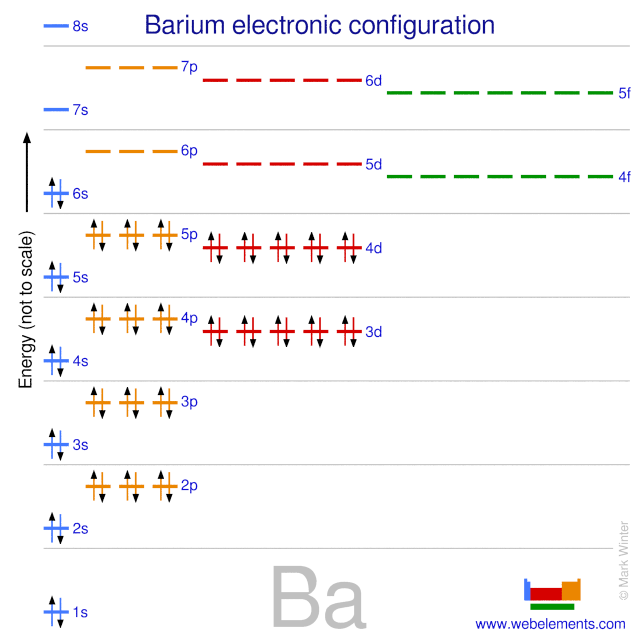
Credit: winter.group.shef.ac.uk
Full Electron Configuration
Barium’s full electron configuration shows how its 56 electrons are arranged in shells and subshells. It ends with 6s² after filling the xenon core. This layout explains barium’s chemical behavior and position in the periodic table.
Full Electron Configuration Of Barium
The full electron configuration shows how electrons fill the atomic orbitals of barium. It reveals the order of energy levels occupied by electrons. This configuration helps explain barium’s chemical behavior and position in the periodic table.
Barium has an atomic number of 56, meaning it has 56 electrons. These electrons fill orbitals starting from the lowest energy level to higher ones. The configuration follows the Aufbau principle, Pauli exclusion principle, and Hund’s rule.
Step-by-step Electron Filling
First, the 1s orbital fills with 2 electrons. Next, the 2s and 2p orbitals fill with 8 electrons total. Then, the 3s, 3p, and 4s orbitals fill, adding 18 electrons.
After that, the 3d and 4p orbitals fill with 18 more electrons. The 5s and 4d orbitals fill next, adding 10 electrons. Finally, the 5p and 6s orbitals fill, completing barium's electron shell.
Detailed Configuration Notation
The full electron configuration for barium is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s².
This notation lists each subshell and the number of electrons it contains. It shows the precise distribution of electrons across orbitals.
Importance Of Full Configuration
Knowing the full electron configuration helps predict barium’s reactivity. The two electrons in the 6s subshell are the outermost and most reactive. This explains barium’s tendency to form +2 ions.
It also clarifies barium’s placement in group 2 of the periodic table, the alkaline earth metals. The full configuration provides a complete view of its atomic structure.
Noble Gas Core Notation
The noble gas core notation simplifies the electron configuration of elements. It uses the symbol of the nearest noble gas before the element to represent inner electrons. This shorthand method makes reading and writing electron configurations easier. For barium, this notation highlights its outer electrons clearly.
What Is Noble Gas Core Notation?
Noble gas core notation replaces inner electron shells with a noble gas symbol. This method shows only the electrons beyond the noble gas core. It reduces long electron configurations into a compact form. The noble gas chosen is the one that comes before the element in the periodic table.
Noble Gas Core Notation For Barium
Barium has the atomic number 56, meaning it has 56 electrons. The closest noble gas before barium is xenon (Xe), with 54 electrons. Using noble gas core notation, barium’s electron configuration is written as [Xe] 6s2. This shows xenon's full shells plus two electrons in the 6s orbital.
Benefits Of Using Noble Gas Core Notation
This notation saves time and space when writing configurations. It helps focus on the valence electrons, which are important for chemical reactions. It also makes learning electron configuration easier for students. The notation highlights the element’s unique outer electrons quickly.

Credit: wayground.com
Valence Electrons Role
Valence electrons are the outermost electrons of an atom. They decide how an atom reacts with others. For barium, these electrons are key to its chemical behavior. They determine how barium bonds and forms compounds. Understanding valence electrons helps explain barium’s place in the periodic table and its uses.
What Are Valence Electrons In Barium?
Barium has two valence electrons. These electrons are found in the 6s orbital. The full electron configuration ends with 6s2. These two electrons are outside the inner shells and are free to interact.
How Valence Electrons Affect Barium’s Properties
Valence electrons control barium’s reactivity. Barium easily loses its two 6s electrons. This loss forms a +2 charged ion. The ion then bonds with other elements. This makes barium very reactive, especially with nonmetals.
Valence Electrons And Barium’s Role In Compounds
Barium’s two valence electrons allow it to form stable compounds. It often forms ionic bonds by giving away these electrons. Common compounds include barium sulfate and barium chloride. These compounds have many industrial and medical uses.
Electron Configuration Of Ba2+ Ion
The electron configuration of the Ba2+ ion reveals important details about its chemical behavior and stability. Barium, a group 2 element, loses electrons to form this ion. Understanding which electrons are lost helps explain its reactivity and bonding.
When barium forms a Ba2+ ion, it loses two electrons from its outer shell. These electrons come from the 6s orbital, the highest energy level occupied in the neutral atom. Removing these electrons changes the electron arrangement significantly.
Electron Configuration Of Neutral Barium
Neutral barium has an atomic number of 56. Its electron configuration is [Xe] 6s2. This means it has the same electron configuration as xenon, plus two electrons in the 6s orbital. The 6s electrons are the most easily removed.
Electron Loss in Ba2+ IonIn the Ba2+ ion, the two 6s electrons are lost. This loss makes the ion more stable. The ion now has the electron configuration of xenon, a noble gas. This configuration is very stable due to a full outer shell.
Final Electron Configuration of Ba2+After losing two electrons, Ba2+ has the configuration [Xe]. It contains 54 electrons, matching the noble gas xenon. This stable electron setup explains Ba2+'s common presence in compounds and reactions.
Comparison With Other Alkaline Earth Metals
Comparing the electron configuration of barium with other alkaline earth metals reveals clear trends. These elements belong to Group 2 of the periodic table. They all have two electrons in their outermost s-orbital. This similarity defines many of their chemical properties.
Barium stands out because it is heavier and has more electrons. Its electron configuration extends into the sixth energy level. Lighter alkaline earth metals, like magnesium and calcium, have electrons filling lower energy levels. This difference affects their reactivity and atomic size.
Electron Configuration Trends Across The Group
Alkaline earth metals share a general pattern: ending with ns² electrons. For example, beryllium ends with 2s², magnesium with 3s², and barium with 6s². As you move down the group, the principal quantum number increases. This means electrons occupy higher energy shells in heavier elements.
Impact On Chemical Behavior
Barium’s outer electrons are farther from the nucleus than those in lighter metals. This makes barium more reactive in some reactions. The larger atomic size also means barium forms compounds differently. These differences influence how barium interacts with other elements and ions.
Comparison Of Electron Shells And Subshells
While all alkaline earth metals have two electrons in the s-orbital, the inner shells differ. Barium’s inner shells include filled d and f subshells. These additional electrons provide shielding. Shielding reduces the pull of the nucleus on outer electrons, affecting ionization energy.
Applications Influenced By Electron Configuration
The electron configuration of barium plays a crucial role in its applications across various fields. Its unique arrangement of electrons influences how it reacts and interacts with other elements. This arrangement affects barium's physical and chemical properties.
Understanding these properties helps explain why barium is useful in specific industries. The electron configuration determines barium’s ability to form compounds and its behavior in different environments. These factors guide its practical uses.
Medical Imaging and RadiologyBarium’s electron configuration allows it to absorb X-rays effectively. This makes barium sulfate a common contrast agent in medical imaging. It helps highlight parts of the digestive system in X-ray scans. The stable electron arrangement ensures safety during these procedures.
Manufacturing of AlloysBarium’s electrons contribute to its role in strengthening metals. It is added to alloys to improve hardness and durability. The electron configuration affects how barium atoms bond with other metals. This enhances the mechanical properties of the final product.
Electronics and Vacuum TubesBarium’s electron configuration allows it to emit electrons easily. This quality makes it useful in vacuum tubes and cathode ray tubes. The element helps control electrical currents in these devices. Its electron structure supports efficient electron flow.
Glass and Ceramics IndustryBarium’s chemical behavior, shaped by its electron configuration, improves glass quality. It increases glass density and refractive index. This leads to clearer, stronger glass products. Barium compounds also add color and durability to ceramics.
Common Misconceptions
Many people misunderstand the electron configuration of barium. This confusion leads to wrong assumptions about its chemical behavior. Knowing the correct configuration helps in learning more about barium’s properties and reactions.
This section clears up common mistakes and explains the true electron arrangement of barium.
Misconception About The Noble Gas Core
Some think barium’s electron configuration starts from scratch. Actually, it builds on the noble gas xenon’s configuration. This shorthand uses [Xe] to save space and show core electrons clearly.
Incorrect Filling Of 5d Orbitals
People often add electrons to 5d orbitals for barium. Barium’s 5d orbitals stay empty in its ground state. The last electrons fill the 6s orbital, not 5d.
Confusing Barium With Its Ions
Another mistake is mixing up barium’s neutral atom with its ions. The Ba²⁺ ion loses two 6s electrons, changing its configuration. This difference is key in chemical reactions.
Assuming Electron Configuration Changes In Compounds
Many believe barium’s electron configuration changes inside compounds. The electron count stays the same; only electron sharing or transfer happens. The core configuration remains consistent.
Frequently Asked Questions
How Do You Write The Electron Configuration For Barium?
Write barium's electron configuration as [Xe] 6s². It shows barium has the same core as xenon plus two 6s electrons. This notation simplifies its 56-electron arrangement. Use the periodic table to confirm this configuration accurately.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element Zinc (Zn) with atomic number 30.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 belongs to cobalt (Co), atomic number 27.
What Element Is 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 represents the element Chromium (Cr) with atomic number 24.
Conclusion
Understanding the electron configuration of barium helps explain its chemical behavior. Barium has 56 electrons arranged as [Xe] 6s². This setup shows its place in the periodic table as an alkaline earth metal. Knowing this pattern aids in predicting how barium reacts with other elements.
It also supports learning about similar elements nearby. Keep these basics in mind for future chemistry studies. Electron configuration is a simple but powerful tool to understand elements better.



No comments