Electron Configuration of Samarium
When you dive into the world of chemistry, understanding how electrons arrange themselves in an atom is key. If you’ve ever wondered about the electron configuration of samarium, you’re in the right place.
Knowing samarium’s electron setup isn’t just a detail—it unlocks insights into its chemical behavior and how it fits into the bigger picture of the periodic table. You’ll discover exactly how samarium’s electrons are distributed, what makes this element unique, and why this knowledge matters for both students and science enthusiasts like you.
Ready to clear up the mystery behind samarium’s electron arrangement? Let’s get started!
Samarium Basics
Samarium is a rare earth metal found in the lanthanide series. It has the atomic number 62. This element plays a role in magnets, lasers, and nuclear reactors. Its unique properties come from its electron arrangement.
Understanding samarium's electron configuration helps explain its chemical behavior. The arrangement of electrons impacts how it bonds and interacts with other elements.
What Is Samarium?
Samarium is a silvery-white metal. It is soft and has a high melting point. This element is not very common but is important for technology. Scientists extract it from minerals like monazite and bastnäsite.
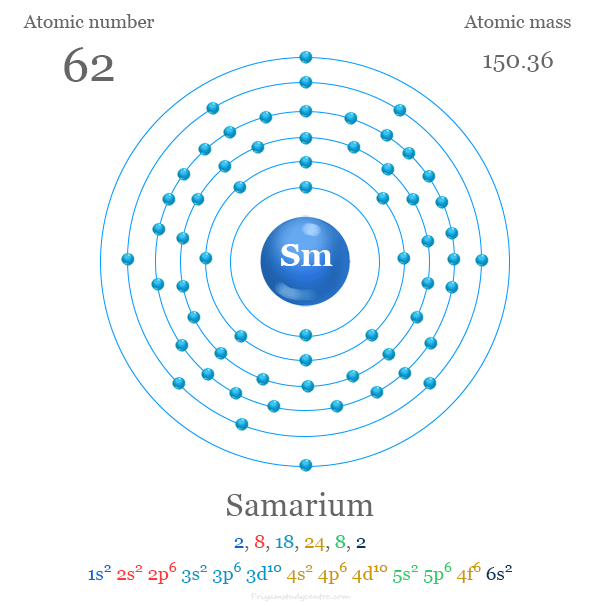
The atomic number of samarium is 62. Its chemical symbol is Sm. This number shows the total protons in its nucleus. It also equals the number of electrons in a neutral atom.
Position In The Periodic Table
Samarium is part of the lanthanide group. It sits in period 6 of the periodic table. This group includes elements with similar properties. Samarium shares traits like magnetism and reactivity with them.
Credit: www.priyamstudycentre.com
Atomic Number And Symbol
Samarium’s atomic number is 62, and its symbol is Sm. Its electron configuration shows how electrons fill energy levels in the atom. Understanding this helps explain Samarium’s chemical behavior.
Atomic Number
The atomic number of samarium is 62. This number shows how many protons are in its nucleus. It also tells the total electrons in a neutral atom. Atomic number helps place samarium in the periodic table. It belongs to the lanthanide series, which includes elements with atomic numbers 57 to 71.
Chemical Symbol
Samarium's chemical symbol is Sm. Symbols are short forms used for elements. They make writing and reading easier in chemistry. The symbol Sm comes from the element's name. This symbol is recognized worldwide and used in scientific formulas and equations.
Electron Shell Structure
Samarium’s electron configuration shows how its electrons fill shells and subshells. It has electrons in the 4f, 6s, and 5d orbitals. This arrangement affects its chemical behavior and place in the periodic table.
Understanding The Electron Shell Structure Of Samarium
The electron shell structure shows how electrons arrange around the nucleus. It explains the energy levels or shells where electrons reside. Each shell holds a limited number of electrons. The arrangement affects the chemical properties of the element.
Samarium is a lanthanide with atomic number 62. It has many electrons filling several shells. Its complex structure includes inner f orbitals. These orbitals influence its unique chemical behavior.
Energy Levels And Electron Distribution
Samarium’s electrons fill shells in order of increasing energy. The first shell holds 2 electrons. The second and third shells hold 8 electrons each. After these, electrons fill the 4th and 5th shells with more electrons.
The 4th shell contains 18 electrons, and the 5th shell holds 8. The 6th shell begins filling with 2 electrons before reaching the 4f sublevel. The 4f sublevel holds 6 electrons in samarium, defining its position in the lanthanide series.
Role Of F-orbitals In Samarium’s Electron Shell
F-orbitals are unique and complex in shape. They start filling at the 4th energy level in samarium. These orbitals hold up to 14 electrons but samarium fills only 6. The 4f electrons are important for magnetic and optical properties.
This partial filling makes samarium useful in specialized technology. It also affects the element’s reactivity. The f-orbitals contribute to samarium’s role in strong magnets and lasers.
Summary Of Samarium’s Electron Shell Structure
Samarium’s shell structure shows a clear pattern of filling. Electrons fill lower energy levels first. Then, electrons occupy the 4f sublevel, which is key to its chemistry. The total number of electrons is 62, matching its atomic number.
This shell structure helps explain samarium’s physical and chemical traits. It reveals why samarium behaves differently from other elements. Understanding this structure is essential for studying lanthanides.

Credit: pilgaardelements.com
Full Electron Configuration
Samarium’s full electron configuration shows how its 62 electrons fill atomic orbitals. It follows the pattern: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f⁶ 5d⁰. This arrangement explains its chemical behavior and properties.
Understanding Full Electron Configuration
The full electron configuration shows how electrons fill atomic orbitals. It lists each orbital and the number of electrons inside. This pattern follows specific rules based on energy levels.
Samarium is an element with atomic number 62. Its electrons fill orbitals in a unique way. The full configuration helps explain its chemical behavior and properties.
Electron Configuration Of Samarium
Samarium’s full electron configuration is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 4f⁶ 5d⁰ 6s²
This shows all electrons arranged from the lowest to highest energy orbitals. The 4f orbitals contain six electrons, which is important for samarium's magnetic and chemical properties.
Significance Of The 4f Subshell
The 4f subshell fills after the 6s orbital in samarium. It holds six electrons here. These electrons influence the element’s magnetic strength and color.
Electrons in the 4f subshell are less shielded. This makes samarium's atoms interact in special ways. It also affects how samarium bonds with other elements.
Placement In The Periodic Table
Samarium is part of the lanthanide series. This group has electrons filling the 4f orbitals. The full electron configuration confirms samarium’s position among these rare earth metals.
Its configuration explains why samarium shows similar properties to other lanthanides. The 4f electron count is a key factor in these similarities.
Valence Electrons In Samarium
Valence electrons are the outermost electrons in an atom. They play a key role in chemical reactions and bonding. Understanding valence electrons in samarium helps explain its unique properties. Samarium is a rare earth element with the symbol Sm and atomic number 62.
Its electron configuration reveals how its valence electrons are arranged. This arrangement affects samarium’s behavior in compounds. The valence electrons mainly occupy the 4f, 5d, and 6s orbitals. These electrons determine how samarium interacts with other elements.
Electron Shells Involved In Valence
Samarium has electrons in the 4f, 5d, and 6s shells. The 4f shell holds electrons that are partly shielded from the nucleus. The 5d and 6s shells are more exposed and active in bonding. These shells together form samarium’s valence electron cloud.
Number Of Valence Electrons
Samarium typically has three valence electrons. Two electrons are in the 6s orbital. One electron is in the 4f orbital, sometimes the 5d orbital also participates. This count can change slightly in different chemical environments.
Role Of 4f Electrons
The 4f electrons are less reactive but crucial for magnetic properties. They influence samarium’s ability to form complex compounds. These electrons are buried deeper but still affect chemical bonding indirectly.
Chemical Behavior Linked To Valence Electrons
Samarium’s valence electrons allow it to form bonds with various elements. It can lose or share these electrons to create stable compounds. This ability makes samarium valuable in materials science and electronics.
Role Of 4f Orbitals
Samarium’s electron configuration fills its 4f orbitals with electrons, impacting its chemical behavior. These 4f orbitals hold electrons that shape its magnetic and optical properties. Understanding this helps explain samarium’s place among lanthanides.
Understanding The 4f Orbitals In Samarium
The 4f orbitals play a key role in samarium's electron configuration. These orbitals are part of the lanthanide series and are filled after the 6s orbitals. In samarium, electrons start to occupy the 4f orbitals, which are deeper inside the atom compared to outer orbitals.
These orbitals have a unique shape and energy level. They affect samarium's chemical and physical properties. The 4f electrons are less shielded by outer electrons, which influences how samarium interacts with other elements.
Electron Filling In The 4f Subshell
Samarium has 6 electrons in its 4f subshell. This filling follows the Aufbau principle, where electrons fill orbitals from lower to higher energy. The 4f orbitals fill after the 6s orbital but before the 5d orbital. This order creates a specific pattern in samarium’s electron arrangement.
These electrons contribute to samarium’s magnetic and optical properties. Their arrangement also affects the atom’s size and reactivity. The 4f electrons are tightly held and do not easily participate in bonding.
Impact Of 4f Orbitals On Samarium's Chemistry
The 4f orbitals influence samarium's chemical behavior. Because these orbitals are shielded by outer electrons, samarium shows unique bonding characteristics. The electrons in 4f orbitals do not easily leave the atom, making samarium less reactive than some other metals.
This shielding effect leads to specific magnetic properties. Samarium can form compounds with distinct colors and shapes due to its 4f electrons. These orbitals also help samarium exhibit strong magnetic moments, useful in many applications.
Comparison With Neighboring Elements
Comparing the electron configuration of samarium with its neighbors helps us understand its unique properties. Samarium sits in the lanthanide series, surrounded by elements with similar but distinct arrangements of electrons. These differences affect their chemical behavior and physical traits.
Examining neighboring elements such as promethium and europium reveals subtle shifts in electron filling. These changes influence magnetic properties, reactivity, and atomic size. The gradual filling of the 4f orbitals plays a key role in this pattern.
Electron Configuration Of Promethium (pm)
Promethium follows samarium in the periodic table. Its electron configuration is [Xe] 4f5 6s2. This means promethium has one fewer electron in the 4f orbital than samarium. This difference leads to slightly different chemical properties. Promethium is radioactive, unlike samarium, which is stable.
Electron Configuration Of Europium (eu)
Europium comes after samarium and has the configuration [Xe] 4f7 6s2. This half-filled 4f orbital provides europium with extra stability. Samarium’s 4f6 configuration is close but less stable. Europium often exhibits a +2 oxidation state, while samarium mostly shows +3.
Differences In Chemical Behavior
These small changes in electron configuration affect how each element reacts. Samarium tends to form compounds with three positive charges. Europium can form both +2 and +3 compounds. Promethium’s radioactivity limits its chemical use. Samarium has more practical applications due to its stability.
Common Configuration Exceptions
Electron configurations often follow predictable patterns based on atomic numbers. Yet, some elements show exceptions due to electron interactions and energy stability. Samarium is one such element with unique behaviors in its electron arrangement.
These common configuration exceptions affect how samarium's electrons fill its orbitals. Understanding these exceptions helps explain its chemical and physical properties.
Why Samarium Shows Configuration Exceptions
Samarium belongs to the lanthanide series. Lanthanides have electrons filling 4f orbitals. These orbitals have close energy levels, causing unusual electron arrangements. Electrons sometimes jump to lower energy states for stability.
Typical Vs. Actual Electron Configuration Of Samarium
The expected configuration fills 4f orbitals sequentially. For samarium, it would be [Xe] 4f6 6s2. But samarium’s actual configuration slightly differs to increase stability. It shows subtle shifts in electron placement within 4f and 5d orbitals.
Impact Of Configuration Exceptions On Samarium’s Properties
These electron shifts influence magnetic and spectral properties. Samarium’s chemical reactivity also changes due to these exceptions. This explains its unique behavior compared to other lanthanides.
Electron Configuration And Chemical Properties
Samarium is a rare earth element with unique chemical traits. Its electron setup helps explain these traits clearly.
Studying samarium’s electrons shows why it behaves in certain ways. This knowledge is key for using it in technology and science.
Basic Electron Configuration Of Samarium
Samarium has the atomic number 62. This means it has 62 electrons. These electrons fill energy levels in a specific order.
The electron configuration for samarium is: [Xe] 4f⁶ 6s². This shows it has six electrons in the 4f orbital and two in the 6s orbital. The 4f electrons are important for its chemical behavior.
Effect Of Electron Configuration On Chemical Properties
Electrons in the 4f orbital are less reactive but influence magnetic and optical properties. Samarium often forms +3 oxidation states by losing electrons from the 6s and 4f orbitals.
This electron loss leads to compounds with unique magnetic features. These compounds are useful in permanent magnets and electronic devices.
Electron Configuration And Reactivity
Samarium’s electron configuration makes it moderately reactive. It reacts slowly with water and oxygen at room temperature. Heating increases its reactivity.
Its ability to form stable compounds depends on the 4f electrons. These electrons shield the nucleus, affecting how samarium bonds with other elements.

Credit: www.shutterstock.com
Applications Of Samarium's Electron Setup
Samarium’s unique electron configuration shapes its special properties. This element has electrons arranged in a way that influences magnetism and chemical behavior. The electron setup helps samarium perform well in many fields.
Its partially filled 4f orbitals give samarium strong magnetic and optical traits. These traits lead to important uses in technology and industry. Understanding samarium’s electron setup shows why it fits certain applications.
Permanent Magnets And Electronics
Samarium’s electron arrangement creates strong magnetic moments. This feature makes samarium ideal for permanent magnets. These magnets are smaller but very powerful. They appear in headphones, microphones, and small motors. Electronics benefit from samarium magnets due to their stability and strength.
Catalysts In Chemical Reactions
Samarium’s electron configuration allows it to act as a catalyst. It helps speed up reactions without being consumed. This is useful in organic chemistry for producing medicines and fine chemicals. The unique electron setup helps samarium interact with other elements easily.
Optical Materials And Lasers
Samarium ions absorb and emit light in special ways. The electron arrangement supports these optical transitions. This property makes samarium useful in laser technology. Samarium-doped crystals produce light for medical and scientific lasers. The electron setup enables precise control of light energy.
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 represents the element Zinc (Zn), atomic number 30.
Which Element Is This 1s22s22p63s23p64s23d104p5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5 belongs to the element Bromine (Br).
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ is Ruthenium (Ru). It has atomic number 44 and belongs to the transition metals group.
Which Element Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 is Chromium (Cr), atomic number 24.
Conclusion
Samarium’s electron configuration reveals its place in the lanthanide series. It helps explain the element’s chemical behavior and properties. Knowing its electron arrangement aids understanding of atomic structure. This knowledge supports learning about rare earth elements. Samarium’s electrons fill the 4f orbitals uniquely.
Such details matter in chemistry and material science studies. Understanding these basics makes complex concepts easier to grasp. Keep exploring electron configurations to deepen your chemistry skills.



No comments