Electron Configuration of Holmium
Have you ever wondered what makes holmium unique among the elements? Understanding its electron configuration is key to unlocking its chemical behavior and role in advanced technologies.
You’ll discover exactly how holmium’s electrons are arranged and why that matters to scientists and engineers alike. By the end, you’ll have a clear grasp of this fascinating rare earth metal’s atomic structure, giving you a fresh perspective on the periodic table and the elements that shape our world.
Keep reading to uncover the secrets hidden in holmium’s electron configuration—and see how this knowledge can expand your understanding of chemistry.
Holmium Basics
Holmium’s electron configuration is [Xe] 4f¹¹ 6s², showing its place in the lanthanide series. This setup affects its magnetic and chemical behavior. Understanding these electrons helps explain holmium’s unique properties.
Holmium is a rare earth element with the symbol Ho and atomic number 67. It belongs to the lanthanide series in the periodic table. Holmium is known for its unique magnetic and spectral properties. It appears as a silvery-white metal that is soft and malleable.
This element is not abundant in nature but can be found in minerals like monazite and gadolinite. Holmium has important uses in scientific research and industrial applications. Understanding its electron configuration helps explain its chemical behavior and reactivity.
Atomic Structure Of Holmium
Holmium has 67 electrons arranged in specific energy levels around its nucleus. These electrons occupy orbitals in a way that follows the Aufbau principle. The distribution of electrons affects how holmium interacts with other elements.
Electron Configuration Explained
The electron configuration of holmium is [Xe] 4f11 6s2. This means it has eleven electrons in the 4f orbital and two in the 6s orbital. The [Xe] represents the electron configuration of xenon, a noble gas preceding holmium. This shorthand helps simplify complex electron arrangements.
Chemical Properties Linked To Configuration
Holmium’s electron configuration leads to a stable +3 oxidation state. It easily loses its 6s and some 4f electrons during reactions. This behavior makes holmium useful in magnets and lasers. The partially filled 4f shell gives holmium unique magnetic properties.
Atomic Structure
The atomic structure of holmium defines its unique chemical and physical traits. This rare earth element belongs to the lanthanide series. Its atoms contain a specific arrangement of electrons that influences its reactivity and magnetic properties.
Understanding holmium's atomic structure starts with its electron configuration. This arrangement reveals how electrons fill the energy levels and orbitals around the nucleus. Each electron shell and subshell holds a fixed number of electrons, shaping the element’s behavior.
Electron Shells And Subshells
Holmium atoms have 67 electrons distributed among several shells. These shells are divided into subshells: s, p, d, and f. The f subshell plays a key role in holmium's properties. It contains electrons that are less shielded and more reactive.
4f Orbital Occupation
Eleven electrons occupy the 4f orbital in holmium. This partially filled 4f subshell creates strong magnetic moments. The arrangement in this orbital distinguishes holmium from other lanthanides. It also affects its spectral and magnetic behavior.
Outer Electron Configuration
Holmium’s outermost electrons are found in the 6s orbital. Two electrons occupy this orbital, contributing to its chemical bonding. Together with the 4f electrons, these outer electrons define holmium’s typical +3 oxidation state.
Electron Shells
Holmium’s electron configuration reveals its electrons arranged in shells around the nucleus. It has electrons filling the 4f and 6s orbitals, showing its place in the lanthanide series. Understanding this helps explain holmium’s chemical behavior and properties.
Understanding Electron Shells In Holmium
Electron shells are layers around an atom's nucleus. Each shell holds electrons with specific energy levels. Holmium has several electron shells filled in a unique order.
The arrangement of electrons in these shells defines many properties of holmium. This pattern is called the electron configuration. It helps explain holmium’s behavior in chemical reactions and physical states.
Electron Distribution In Holmium
Holmium has 67 electrons distributed in shells. These electrons fill shells starting from the closest to the nucleus. The shells are labeled as K, L, M, N, O, P, and Q in order.
For holmium, the electron shells fill up to the 6th level. The outermost electrons are in the 6th shell, which affects how holmium interacts with other elements.
Role Of 4f And 6s Electrons
The 4f and 6s subshells are very important in holmium’s electron configuration. The 4f subshell contains 11 electrons. These electrons give holmium its unique magnetic and chemical properties.
The 6s subshell has 2 electrons. These electrons are in the outer shell and play a key role in bonding and reactivity of holmium atoms.
4f Orbital Role
Holmium’s electron configuration includes electrons filling the 4f orbital, specifically 4f¹¹. This orbital plays a key role in its magnetic and chemical properties. Electrons in the 4f orbital influence how holmium interacts with other elements.
Understanding The 4f Orbital In Holmium
The 4f orbital plays a crucial role in defining holmium's electron configuration. It holds electrons that are deeply embedded within the atom. These electrons influence holmium's magnetic and chemical properties.
Unlike outer orbitals, the 4f orbital is shielded by outer electrons. This shielding affects how holmium interacts with other elements. The 4f electrons are less involved in bonding but impact the atom’s behavior significantly.
Electron Distribution In The 4f Orbital
Holmium has 11 electrons in its 4f orbital. This is written as 4f11. These electrons fill the complex shape of the 4f orbital. The arrangement leads to unique magnetic properties of holmium.
The 4f electrons are not easily removed or shared. This makes holmium stable in its common +3 oxidation state. The electron distribution in the 4f orbital defines many physical characteristics.
Impact Of The 4f Orbital On Holmium's Properties
The 4f orbital electrons cause strong magnetic behavior. Holmium exhibits one of the highest magnetic moments among elements. This trait is valuable in magnets and magnetic refrigeration.
These electrons also affect holmium’s optical properties. The 4f electrons absorb and emit light in specific ways. This explains holmium's use in lasers and specialized glass.
Valence Electrons
Valence electrons are the outermost electrons of an atom. They play a key role in chemical reactions and bonding. Understanding the valence electrons of holmium helps explain its behavior in compounds and materials.
Holmium is a lanthanide element with a complex electron arrangement. Its valence electrons are found mainly in the 6s and 4f orbitals. These electrons influence holmium’s magnetic and chemical properties.
Electron Shells Involved In Holmium's Valence
Holmium has electrons in the 4f, 5d, and 6s shells. The 4f electrons are partially filled and contribute to unique magnetic characteristics. The 6s electrons are the most loosely held and often participate in bonding.
Number Of Valence Electrons In Holmium
Holmium has three valence electrons. Two electrons occupy the 6s orbital, and one electron is in the 4f orbital. These electrons are important for forming chemical bonds and determining reactivity.
Role Of 4f Electrons In Holmium's Chemistry
The 4f electrons in holmium are shielded by outer electrons. They do not easily participate in bonding but affect magnetic properties. These electrons also influence the element’s color and spectral features.

Credit: pilgaardelements.com
Electron Configuration Notation
Electron configuration notation is a way to show how electrons are arranged in an atom. It helps us understand the structure and behavior of elements. Each electron occupies a specific energy level and sublevel around the nucleus. This arrangement affects the chemical properties of the element.
For holmium, a rare earth metal, the electron configuration reveals its unique characteristics. The notation uses numbers, letters, and superscripts to represent energy levels, sublevels, and electron counts. This system follows rules based on quantum mechanics.
Understanding The Notation Symbols
The numbers in the notation show the energy levels or shells (1, 2, 3, etc.). Letters represent sublevels: s, p, d, and f. Each sublevel can hold a limited number of electrons. Superscripts indicate how many electrons are in each sublevel.
Holmium’s Electron Configuration
Holmium has 67 electrons in total. Its configuration is written as 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹¹. This shows electrons filling up shells and sublevels in order of increasing energy.
Importance Of Electron Configuration Notation
This notation helps predict how holmium reacts with other elements. It also explains its magnetic and spectral properties. Scientists use this system to study atomic structure and chemical bonding.
Common Misconceptions
Understanding the electron configuration of holmium can be tricky. Many learners hold common misconceptions that confuse the topic. Clearing these up helps in grasping holmium’s unique atomic structure.
Holmium’s position in the lanthanide series adds complexity. This leads to errors in reading its electron arrangement. Let’s explore some frequent misunderstandings to guide better learning.
Misinterpreting The 4f Electron Filling
Many assume the 4f orbitals fill in a simple, linear order. Holmium’s 4f electrons, however, follow specific rules. The 4f subshell fills with 11 electrons in holmium, not 10 or 12. This detail often confuses beginners.
The 4f electrons are shielded and behave differently than s or p electrons. This causes misconceptions about their placement and energy levels.
Confusing The Role Of 6s Electrons
People often think 6s electrons contribute mainly to chemical bonding. Holmium’s 6s electrons are indeed the outermost but remain largely non-bonding. They exist as a stable pair.
This stability leads to the common mistake of overestimating their reactivity. Holmium’s chemical traits depend more on 4f electrons than 6s.
Ignoring The Effect Of Electron Spin And Hund’s Rule
Hund’s rule states electrons fill degenerate orbitals singly first. Overlooking this results in incorrect configurations. Holmium’s 4f electrons occupy orbitals to maximize spin.
Ignoring spin leads to errors in counting electrons or their arrangement. Correct understanding reflects the true energy state of the atom.
Assuming Holmium’s Configuration Matches Lighter Lanthanides
Holmium has more electrons than lighter lanthanides like cerium or neodymium. Assuming their configurations are the same causes mistakes. Each lanthanide differs in its 4f electron count.
Holmium’s unique 4f11 configuration shapes its properties distinctly. Treating all lanthanides alike misses these nuances.
Comparison With Lanthanides
Holmium’s electron configuration closely matches other lanthanides, filling the 4f subshell. This pattern influences its magnetic and chemical properties. Compared to lighter lanthanides, holmium has more 4f electrons, affecting its reactivity and atomic behavior.
Electron Configuration Patterns In Lanthanides
Holmium belongs to the lanthanide series. Lanthanides share similar electron configurations. They fill the 4f orbitals progressively as atomic number rises. Holmium’s configuration is [Xe] 4f11 6s2. This means it has eleven electrons in the 4f subshell. This pattern matches the general trend of lanthanides filling 4f electrons one by one.
Each lanthanide element adds one electron to the 4f orbital. This gradual filling affects their chemical and physical properties. Holmium’s 4f11 configuration places it near the middle of this series. Its electron arrangement influences its magnetic and spectral behaviors.
Differences In Electron Shielding And Stability
Lanthanides have poor shielding by 4f electrons. Holmium’s 4f electrons shield less effectively than s or p electrons. This causes a strong attraction between the nucleus and outer electrons. The result is the lanthanide contraction, a decrease in atomic size across the series.
Holmium experiences this contraction like other lanthanides. Its electrons feel a stronger pull from the nucleus. This effect impacts its ionization energy and chemical reactivity. Compared to early lanthanides, holmium's electrons are held more tightly.
Comparison Of Chemical Behavior
Holmium shows typical lanthanide chemistry. It mainly forms +3 oxidation state ions. This state results from losing two 6s electrons and one 4f electron. Like most lanthanides, it reacts slowly with water and acids. It also forms stable complexes with organic ligands.
Its electron configuration influences its magnetic properties. Holmium has a high magnetic moment due to unpaired 4f electrons. This feature is common among lanthanides with partially filled 4f orbitals. Holmium’s behavior fits well within the lanthanide group.
Impact On Chemical Properties
Holmium’s electron configuration plays a key role in its chemical behavior. The arrangement of electrons in its atoms affects how it reacts with other elements. This impact shapes holmium’s unique chemical properties and its place in the periodic table.
The electron configuration of holmium is [Xe] 4f11 6s2. This means it has eleven electrons in the 4f orbital and two in the 6s orbital. These electrons determine how holmium bonds with other atoms and its typical oxidation state.
Stability Of The Trivalent State
Holmium usually forms ions with a +3 charge. This trivalent state is very stable because it results from losing the two 6s electrons and one 4f electron. The stability comes from the half-filled 4f orbitals, which lower the energy of the atom.
This stable state influences holmium’s common compounds and reactions. It tends to form ionic bonds with nonmetals and shows predictable chemical behavior in this form.
Magnetic Properties And Electron Spin
The eleven 4f electrons give holmium strong magnetic properties. Their unpaired spins create a large magnetic moment. This makes holmium useful in magnetic materials and devices.
The electron spin also affects how holmium interacts with light and other electromagnetic radiation. These properties depend directly on its electron configuration.
Reactivity With Other Elements
Holmium reacts mainly with oxygen and halogens. Its electron configuration allows it to lose electrons easily and form compounds like holmium oxide (Ho2O3) and halides.
Its chemical reactivity is moderate, not too high or too low. The stable 4f electrons shield the nucleus and reduce the reactivity compared to lighter lanthanides.
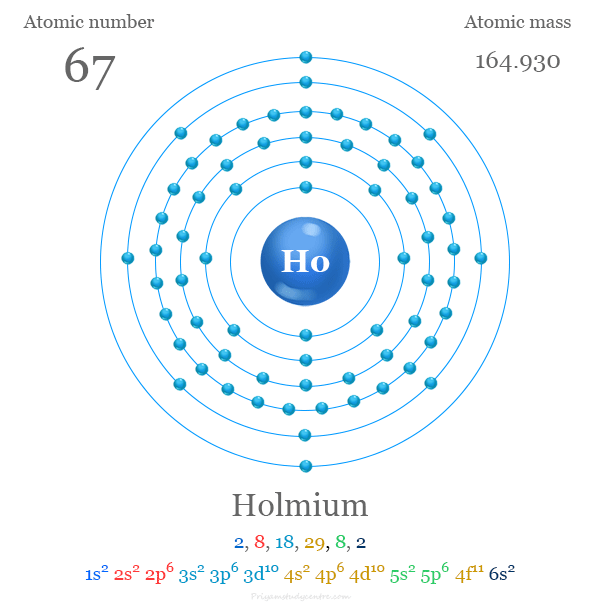
Credit: www.priyamstudycentre.com
Applications Linked To Configuration
The electron configuration of holmium plays a key role in its unique applications. This lanthanide element shows special magnetic and optical properties. These features arise from its 4f electron arrangement. Understanding this helps explain why holmium is useful in certain technologies.
Holmium’s electrons influence its magnetic strength and stability. This makes it valuable in devices requiring strong magnets. Its electron setup also affects how it interacts with light, which is important for lasers and optical instruments.
Magnetic Applications Of Holmium
Holmium’s electron configuration gives it high magnetic moments. It is one of the strongest natural magnets at low temperatures. This property is used in nuclear reactors to control neutron flux. Holmium magnets help in medical imaging machines like MRI scanners. These applications rely on its stable and strong magnetism.
Laser Technology Uses
The 4f electrons in holmium emit light at specific wavelengths. Holmium-doped lasers produce infrared light useful in surgery and dentistry. Its electron configuration allows precise light emission for these tools. These lasers can cut or remove tissue with minimal damage. This precision is vital in medical treatments.
Data Storage And Electronics
Holmium’s electron structure supports high-density data storage. It helps create materials with controlled magnetic properties. These materials improve the performance of hard drives and memory devices. The element’s stable electron arrangement ensures reliability in electronics. This makes it a candidate for future technology advancements.
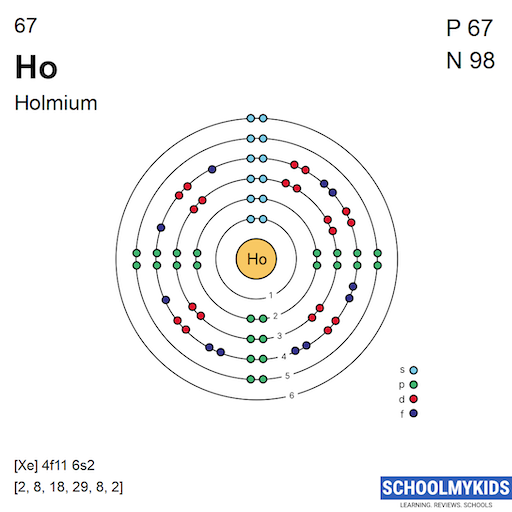
Credit: www.schoolmykids.com
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 represents the element zinc (Zn). It shows zinc’s full first four energy levels.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn).
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration can be 2, 8, 8 or 2, 8, 18 depending on the element. For lighter elements, it is 2, 8, 8, while heavier elements may have 2, 8, 18. Shell capacity varies by energy levels and subshell filling.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 5?
The electron configuration 1s² 2s² 2p⁵ belongs to fluorine. Fluorine is a highly reactive halogen with atomic number 9.
Conclusion
Understanding holmium’s electron configuration helps explain its unique behavior. It has 67 electrons arranged as [Xe] 4f¹¹ 6s². This setup influences its chemical and physical properties. Holmium belongs to the lanthanide series, known for magnetic and optical uses. Knowing its electron layout guides scientists and students alike.
The pattern shows how electrons fill orbitals in heavy elements. Simple yet important for grasping element characteristics. Keep exploring to see how electron configurations affect the periodic table. Holmium’s structure is a key piece in that puzzle.



No comments