Electron Configuration of Iodine
Are you curious about what makes iodine tick at the atomic level? Understanding the electron configuration of iodine is key to unlocking how this element behaves in chemical reactions and why it holds such a unique place in the periodic table.
By exploring how iodine’s electrons are arranged, you’ll gain insights that can help you predict its properties, reactivity, and role in everything from medicine to industry. Stick with me, and you’ll discover a simple, clear breakdown of iodine’s electron setup—no complicated jargon, just the facts you need to grasp this essential concept fully.
Ready to dive in? Let’s get started!

Credit: www.shutterstock.com
Basic Properties Of Iodine
Iodine is a chemical element with unique basic properties. It is a non-metal and belongs to the halogen group on the periodic table. Its atomic number is 53, meaning it has 53 protons and electrons in a neutral state.
This element appears as a shiny, dark gray solid at room temperature. Iodine easily sublimates, turning directly from solid to gas with a violet color. It is less reactive than other halogens like chlorine or fluorine.
Physical State And Appearance
Iodine exists as a solid under normal conditions. It has a metallic luster and is brittle. When heated, iodine changes into a violet gas without melting first.
Chemical Reactivity
Iodine reacts moderately with other elements. It forms compounds mainly by gaining one electron to become iodide ions. Its reactivity is lower compared to fluorine and chlorine.
Electron Configuration
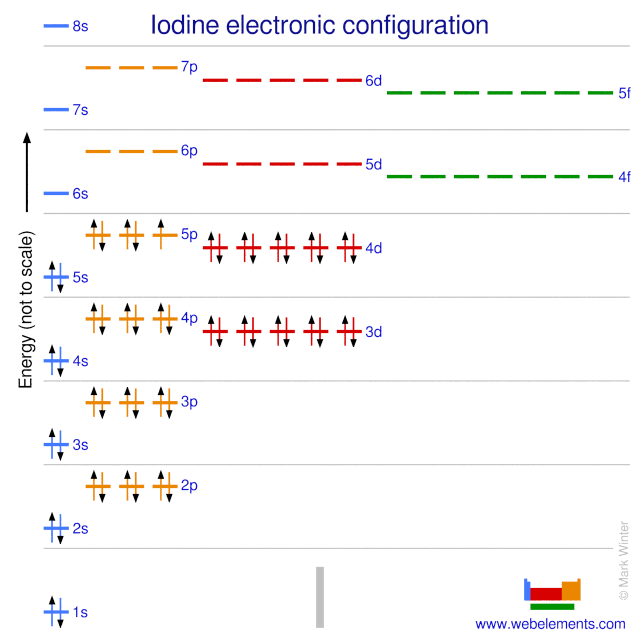
The electron configuration of iodine is key to its behavior. It is written as [Kr] 4d10 5s2 5p5. This means iodine has seven electrons in its outer shell, ready to form bonds.
Occurrence And Uses
Iodine naturally occurs in small amounts in seawater and minerals. It is essential in human nutrition, especially for thyroid health. Iodine is used in medicine, disinfectants, and photography.
Atomic Structure And Electrons
The atomic structure of iodine defines its chemical behavior. Atoms consist of a nucleus surrounded by electrons. These electrons occupy specific energy levels or shells. Their arrangement follows strict rules that shape iodine's properties.
Electrons fill shells based on energy levels, starting from the lowest. The distribution of electrons affects how iodine reacts with other elements. Understanding this distribution helps explain iodine's role in chemistry and biology.
Electron Shells In Iodine
Iodine has 53 electrons arranged in shells around its nucleus. These shells hold electrons in increasing order of energy. The first shell holds 2 electrons. The second shell can hold up to 8 electrons. The third shell holds 18 electrons, and so on.
For iodine, the electrons fill five shells. The outermost shell contains 7 electrons. This outer shell is also called the valence shell. Valence electrons determine iodine's chemical bonding.
Subshells And Their Role
Inside each shell are subshells labeled s, p, d, and f. These subshells hold specific numbers of electrons. For iodine, the electron filling follows the order of these subshells.
The s subshell holds 2 electrons, p holds up to 6, d holds up to 10, and f holds up to 14. Iodine’s electrons fill these subshells in a pattern that matches its atomic number.
Electron Configuration Notation
Electron configuration shows how electrons are distributed among shells and subshells. For iodine, it is written as 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁵.
This notation reveals that iodine has seven electrons in its outermost shell. These electrons are in the 5s and 5p subshells. This arrangement explains iodine’s ability to gain one electron and form iodide ions.
Electron Shells And Their Capacities
Electron shells are layers around an atom's nucleus. Each shell holds a set number of electrons. These shells determine how atoms behave and bond with others. Understanding their capacities helps explain iodine’s unique properties.
Electrons fill shells starting from the closest to the nucleus. Each shell can hold a maximum number of electrons based on a simple formula. This formula guides the arrangement of electrons in iodine.
The capacity of each shell follows the rule 2n², where n is the shell number. For example, the first shell (n=1) can hold up to 2 electrons. The second shell (n=2) holds up to 8 electrons. The third shell (n=3) can carry up to 18 electrons.
Higher shells hold even more electrons. The fourth shell (n=4) can contain up to 32 electrons. Iodine’s electrons fill these shells in order to create its stable electron configuration.
How Electron Shells Fill Up
Electrons fill shells from the lowest energy level to the highest. This pattern is called the Aufbau principle. It means iodine’s electrons first occupy shells closest to the nucleus.
After filling the first three shells, iodine’s electrons enter the fourth and fifth shells. This filling order explains iodine’s chemical behavior and its place on the periodic table.
Significance Of Shell Capacities For Iodine
Iodine has 53 electrons. These electrons distribute across five shells. The first shell holds 2 electrons, the second 8, the third 18, the fourth 18, and the fifth 7 electrons.
The outermost shell has 7 electrons, making iodine reactive. This arrangement influences how iodine bonds and interacts with other elements.
Credit: winter.group.shef.ac.uk
Aufbau Principle And Iodine
The Aufbau Principle guides how electrons fill atomic orbitals. It states electrons occupy the lowest energy orbitals first. This principle helps us understand iodine’s electron arrangement.
Iodine has 53 electrons. These electrons fill orbitals in a specific order. The Aufbau Principle shows the step-by-step filling of these orbitals.
What Is The Aufbau Principle?
The Aufbau Principle means "building up" in German. It explains electron order in atoms. Electrons fill orbitals from low to high energy. This order affects chemical behavior and bonding.
Applying The Aufbau Principle To Iodine
Start filling with the 1s orbital. Then, fill 2s, 2p, 3s, and so on. Iodine’s electrons fill orbitals up to 5p. The 5p orbital holds the outermost electrons.
This filling order follows strict rules. Each orbital has a maximum electron capacity. The 1s orbital holds 2 electrons, 2p holds 6, and 5p holds 6 as well.
Electron Configuration Of Iodine Using Aufbau
The full electron configuration of iodine is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁵
This shows all electrons from the lowest to highest energy orbitals. The 5p⁵ means iodine has five electrons in its outermost p orbital.
Iodine's Electron Configuration Notation
Iodine's electron configuration notation shows how its electrons arrange in shells and orbitals. This notation helps us understand iodine’s chemical behavior and bonding.
Iodine has 53 electrons. These electrons fill orbitals in a specific order. The notation uses numbers and letters to represent energy levels and orbital types.
Basic Structure Of Iodine’s Electron Configuration
The electron configuration starts from the lowest energy level. Electrons fill the 1s orbital first, then move to higher orbitals. The order follows the Aufbau principle.
For iodine, this filling goes through many orbitals until reaching the 5p orbital. This orbital holds the outermost electrons.
Electron Configuration Notation For Iodine
The full electron configuration for iodine is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁵.
This notation breaks down the 53 electrons into their respective orbitals and energy levels clearly.
Understanding The Symbols In Notation
Numbers like 1, 2, 3, 4, and 5 show the energy levels or shells. Letters s, p, and d represent orbital types. Superscripts indicate how many electrons occupy each orbital.
For example, 5p⁵ means five electrons fill the p orbital in the fifth shell.
Valence Electrons In Iodine
Iodine is a chemical element with unique electron arrangements. Its valence electrons play a key role in chemical bonding and reactivity. Understanding these valence electrons helps explain iodine’s properties and behavior in compounds.
Valence electrons are the outermost electrons of an atom. They determine how atoms interact with each other. For iodine, these electrons are found in specific atomic orbitals that influence its chemical nature.
Number Of Valence Electrons In Iodine
Iodine has seven valence electrons. These electrons reside in the fifth energy level. This means iodine can form up to one single bond or gain one electron to complete its octet.
Electron Configuration Of Iodine’s Valence Shell
The valence shell of iodine contains electrons in the 5s and 5p orbitals. Specifically, the configuration is 5s² 5p⁵. This shows five electrons in the p orbital and two in the s orbital.
Role Of Valence Electrons In Chemical Reactions
Valence electrons in iodine participate in reactions by sharing or gaining electrons. This allows iodine to form compounds such as iodides. The seven valence electrons make iodine highly reactive with metals and nonmetals alike.
Iodine In The Periodic Table
Iodine’s electron configuration shows how its 53 electrons fill energy levels around the nucleus. Electrons arrange in shells following specific rules. This arrangement helps explain iodine’s chemical behavior and position in the periodic table.
Iodine is a fascinating element found in the halogen group. It belongs to group 17 and period 5 of the periodic table. This position influences its properties and behavior.
The atomic number of iodine is 53. This means it has 53 protons and, in a neutral state, 53 electrons. The arrangement of these electrons defines iodine’s electron configuration.
Position And Group Characteristics
Iodine shares its group with fluorine, chlorine, bromine, and astatine. These elements have seven electrons in their outer shell. This makes iodine highly reactive, especially with metals.
Its position in period 5 means iodine’s electrons fill up to the fifth energy level. This affects its size and chemical activity compared to lighter halogens.
Electron Configuration Of Iodine
The electron configuration of iodine follows a specific order. Electrons fill the shells and subshells according to energy levels. For iodine, the configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁵.
This arrangement shows iodine has five electrons in its outermost p subshell. These electrons are responsible for iodine’s chemical bonding and reactions.
Significance Of Electron Configuration
Understanding iodine’s electron configuration helps explain its reactivity and compounds it forms. The five outer electrons make iodine a strong oxidizer. It tends to gain one electron to complete its octet.
This property is essential in many applications, including medicine and industry. The electron structure also determines iodine’s color and physical state at room temperature.
Iodide Ion Electron Configuration
The iodide ion forms when iodine gains an extra electron. This extra electron changes its electron configuration. Understanding the iodide ion electron configuration helps explain its chemical behavior and properties.
Iodide ion carries a negative charge (I⁻). This means it has one more electron than a neutral iodine atom. The extra electron fills the outermost shell, making the ion more stable.
Electron Configuration Of Neutral Iodine Atom
Neutral iodine has 53 electrons. Its electron configuration is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁵
This shows electrons fill shells in a set order. The 5p shell holds five electrons in iodine.
How The Iodide Ion Gains An Electron
Iodine gains one electron to become iodide (I⁻). The added electron fills the 5p orbital. So, the 5p shell now contains six electrons.
This extra electron completes the outer shell. It follows the octet rule, making the ion stable.
Electron Configuration Of The Iodide Ion (i⁻)
The electron configuration of I⁻ is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶
This configuration shows a full 5p orbital, unlike neutral iodine. The ion has 54 electrons, one more than iodine.
Importance Of Iodide Ion Configuration
The filled 5p shell makes iodide chemically stable. It behaves differently from neutral iodine in reactions. It often acts as a reducing agent or forms salts with metals.
The electron configuration explains why iodide is common in compounds like sodium iodide (NaI).
Comparing Iodine With Other Halogens
Iodine belongs to the halogen group in the periodic table. Halogens include fluorine, chlorine, bromine, iodine, and astatine. They share similar chemical properties and electron configurations. Understanding iodine’s electron configuration compared to other halogens helps explain its unique traits.
All halogens have seven electrons in their outer shell. This similarity leads to comparable reactivity and bonding behavior. Differences in electron shells affect their physical properties and atomic size.
Electron Configuration Patterns Among Halogens
Halogens follow a clear pattern in electron filling. Fluorine’s configuration ends in 2p5, chlorine’s in 3p5, bromine’s in 4p5, and iodine’s in 5p5. Each element adds a new electron shell as you move down the group. This increase causes iodine’s electrons to be farther from the nucleus.
Atomic Size And Electron Shells
Iodine has more electron shells than fluorine and chlorine. This makes iodine larger in size. The extra shells reduce the attraction between the nucleus and outer electrons. Atomic size impacts iodine’s reactivity and physical state at room temperature.
Reactivity Differences Explained By Electron Configuration
All halogens seek one electron to complete their outer shell. Fluorine is the most reactive due to its small size and strong nuclear pull. Iodine is less reactive because its outer electrons are shielded by inner shells. This shielding lowers its ability to attract electrons.
Physical States And Electron Configuration
Electron configuration influences halogens’ physical states. Fluorine and chlorine are gases at room temperature. Bromine is a liquid, and iodine is a solid. The increase in electron shells and atomic mass causes these changes as you move down the group.
Applications Of Electron Configuration In Chemistry
Electron configuration is a key concept in chemistry that reveals how electrons are arranged in an atom. Understanding these arrangements helps explain many chemical properties and behaviors of elements like iodine. This knowledge guides scientists and students in predicting reactions, bonding, and physical characteristics.
Electron configuration also supports the study of atomic structure and periodic trends. It provides a simple way to visualize how atoms interact with each other. For iodine, its electron configuration shows the distribution of electrons across shells and subshells. This layout influences iodine’s reactivity and its role in chemical compounds.
Electron Configuration And Chemical Bonding
The electron configuration of iodine helps explain its bonding patterns. Iodine has seven electrons in its outer shell. These electrons participate in forming bonds with other atoms. The configuration shows which electrons are available for bonding. It predicts iodine’s tendency to form single covalent bonds. This is common in many iodine-containing molecules.
Predicting Reactivity Using Electron Configuration
Electron configuration reveals iodine’s reactivity with other elements. Iodine’s seven valence electrons make it reactive but less so than halogens like fluorine. The arrangement shows why iodine tends to gain one electron to complete its octet. This helps in understanding iodine’s behavior in reactions, especially in forming iodide ions (I⁻).
Role In Periodic Table Trends
Electron configurations explain iodine’s position in the periodic table. Iodine belongs to group 17, the halogens. Its configuration ends in 5p⁵, indicating five electrons in the p subshell of the fifth shell. This pattern matches other halogens, defining similar chemical traits. The configuration helps predict changes in atomic size, ionization energy, and electronegativity across the group.
Use In Spectroscopy And Quantum Chemistry
Scientists use iodine’s electron configuration to analyze its spectra. The arrangement of electrons affects how iodine absorbs and emits light. This data helps identify elements in samples and study atomic energy levels. Quantum chemistry relies on electron configurations to model iodine’s electron cloud and predict properties at the atomic level.

Credit: www.youtube.com
Frequently Asked Questions
How To Write Electron Configuration For Iodine?
Write iodine's electron configuration by filling orbitals in order: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁵. This represents 53 electrons, matching iodine's atomic number.
What Element Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 corresponds to the element calcium. It has 20 electrons and atomic number 20.
What Is The 2 8 8 18 18 Rule?
The 2 8 8 18 18 rule describes the maximum electrons in an atom’s first five shells. It follows the sequence: 2, 8, 8, 18, and 18 electrons per shell. This rule helps predict electron distribution in elements.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 2?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p² corresponds to Germanium (Ge). It has atomic number 32.
Conclusion
Understanding iodine’s electron configuration helps explain its chemical behavior. Electrons fill orbitals in a specific order. Iodine has 53 electrons arranged to follow this order. This arrangement influences iodine’s reactivity and bonding. Knowing the configuration aids in learning about halogens and periodic trends.
Simple and clear, this knowledge builds a strong chemistry foundation. Keep exploring elements to see how electron configurations shape their properties.



No comments