Electron Configuration of Roentgenium
Are you curious about one of the rarest and most mysterious elements on the periodic table? Roentgenium, with its superheavy atomic number, challenges our understanding of chemistry and physics alike.
Understanding its electron configuration isn’t just a matter of memorizing numbers—it reveals how this elusive element behaves and fits into the bigger picture of the periodic table. You’ll discover the secrets behind Roentgenium’s electron arrangement, why it’s so complex, and what makes it stand out from other elements.
Stick with me, and by the end, you’ll have a clear grasp of this fascinating topic that often confuses even seasoned chemists. Ready to dive in?
Basic Properties
Roentgenium is a rare and very heavy element. Its basic properties tell us about its identity and place in chemistry. Understanding these properties helps us learn more about its behavior and possible uses.
Atomic Number And Symbol
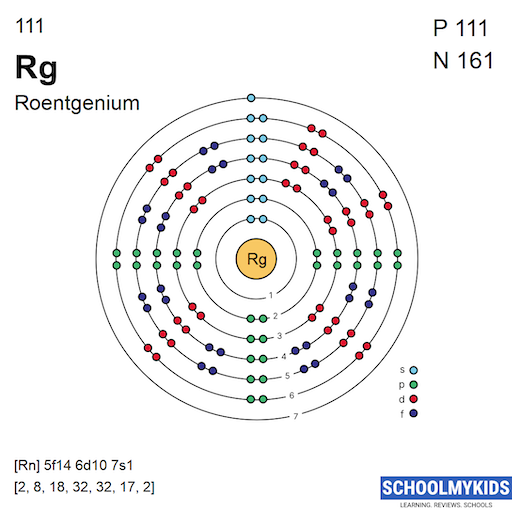
Roentgenium has the atomic number 111. Its chemical symbol is Rg. The atomic number shows the number of protons in its nucleus. This number defines the element and its position in the periodic table.
Position In Periodic Table
Roentgenium is located in group 11 of the periodic table. This group also contains copper, silver, and gold. It belongs to period 7, which includes superheavy elements. Its place suggests some similar chemical traits to these metals.
Physical And Chemical Characteristics
Roentgenium is a synthetic element, made in labs. It is highly unstable and decays quickly. Its physical properties are mostly unknown due to this instability. Scientists predict it is a dense metal with a shiny surface. Chemically, it may behave like other group 11 metals but with unique differences caused by relativistic effects on its electrons.
Credit: www.schoolmykids.com
Electron Arrangement
Roentgenium is a superheavy element with unique electron arrangements. Understanding its electron configuration helps explain its chemical behavior and place in the periodic table. This section breaks down how electrons fill Roentgenium’s orbitals and the effects that influence this pattern.
Electron arrangement in Roentgenium is complex due to its high atomic number. The electrons fill various shells and subshells following specific rules and sequences. Relativistic effects also impact the orbitals, making this element an interesting case for study.
Ground State Configuration
The ground state configuration shows the lowest energy arrangement of electrons in Roentgenium. It has 111 electrons, filling orbitals up to the 7p subshell. The expected configuration is [Rn] 5f¹⁴ 6d¹⁰ 7s² 7p¹. This means it follows the noble gas radon core and fills the f, d, s, and p orbitals in sequence.
Orbital Filling Sequence
Electrons fill orbitals based on energy levels from low to high. For Roentgenium, filling starts with the 1s orbital and moves outward. The d and f orbitals fill between the s and p orbitals at higher energy levels. The sequence follows the Aufbau principle with some variations due to relativistic effects.
Relativistic Effects On Orbitals
Roentgenium’s heavy nucleus causes electrons to move very fast, near the speed of light. This changes their mass and energy, altering orbital sizes and shapes. The 7s and 7p orbitals contract and stabilize, while 6d orbitals expand. These effects shift electron energies and impact chemical properties.
Valence Electrons
Valence electrons are the outermost electrons of an atom. They play a key role in chemical reactions and bonding. Understanding the valence electrons of roentgenium helps predict its behavior in compounds. Despite roentgenium's high atomic number, its valence electrons follow certain patterns found in transition metals. These electrons determine how roentgenium interacts with other elements.
Valence Shell Composition
Roentgenium has an atomic number of 111. Its valence shell includes electrons in the 6d and 7s orbitals. The expected valence electron configuration is 6d97s2. This places roentgenium in group 11, alongside copper, silver, and gold. The 6d orbital electrons influence its chemical properties. The 7s electrons are more loosely held and often involved in bonding.
Impact On Chemical Behavior
The valence electrons give roentgenium properties similar to gold and silver. It may show a +1 or +3 oxidation state in compounds. The electrons in the 6d orbital cause subtle differences from lighter group 11 elements. Roentgenium's chemical behavior is expected to be less reactive than copper. Its valence electrons also affect its bonding style and stability. Understanding these electrons helps scientists predict how roentgenium will react.
Comparison With Neighbors
Roentgenium’s electron configuration shows interesting patterns compared to its neighbors. Examining these patterns helps us understand its chemical behavior. The neighbors of Roentgenium, especially Gold and Platinum, share some electronic traits. These similarities arise from their position in the periodic table. Yet, Roentgenium also displays unique features due to its superheavy status. These differences affect its stability and properties. Let’s explore these aspects in detail.
Similarities With Gold And Platinum
Roentgenium, Gold, and Platinum belong to the same group in the periodic table. Their electron configurations end in d-block orbitals. This gives them similar chemical characteristics. All three have filled d-subshells and partially filled s-subshells in their outer layers. These traits influence their metallic nature and bonding styles. They often exhibit similar oxidation states. These common features make Roentgenium’s chemistry somewhat predictable based on Gold and Platinum’s behavior.
Differences Due To Superheavy Status
Roentgenium is a superheavy element with a very high atomic number. This causes strong relativistic effects on its electrons. These effects alter the energy levels of orbitals, changing the electron configuration slightly. Its outer electrons are held more tightly than those in Gold or Platinum. Roentgenium’s atoms are also less stable and decay quickly. These factors lead to differences in chemical reactivity and bonding. The superheavy nature creates unique challenges in studying Roentgenium’s properties directly.
Experimental Challenges
Studying the electron configuration of roentgenium presents many experimental challenges. This element is synthetic and extremely unstable. Scientists face hurdles in creating and analyzing it. These difficulties limit direct knowledge of its electronic structure.
Synthesis Difficulties
Roentgenium does not exist naturally. Researchers produce it by colliding lighter atoms in particle accelerators. The process requires exact conditions and high energy. The chance of successful synthesis is very low. Few atoms are made at a time. This limits the amount available for study.
Short Half-life Implications
Roentgenium atoms decay within seconds or less. Their short half-life restricts observation time. Measuring properties before decay is tough. Rapid decay also complicates experiments on electron behavior. Scientists must work quickly and with advanced equipment.
Limitations In Direct Observation
Directly observing roentgenium’s electrons is nearly impossible. Its fleeting existence prevents detailed measurements. Researchers rely on theoretical models and indirect methods. These include computer simulations and comparisons with similar elements. Such approaches provide clues but lack full certainty.
Theoretical Predictions
Theoretical predictions play a vital role in understanding the electron configuration of Roentgenium. This element is highly unstable and rare, making direct observation almost impossible. Scientists rely on models and calculations to estimate its electron arrangement. These predictions help reveal how Roentgenium might behave chemically and physically.
Two main approaches guide these predictions. Quantum mechanical models provide the fundamental framework. Computational chemistry offers detailed simulations. Both methods complement each other in studying this superheavy element.
Quantum Mechanical Models
Quantum mechanical models use mathematical equations to describe electron behavior. They consider the effects of relativity due to Roentgenium’s high atomic number. These models predict that electrons fill orbitals in a pattern similar to lighter elements, but with slight shifts.
The predicted configuration suggests that Roentgenium’s outer electrons occupy the 7s and 6d orbitals. This arrangement affects its chemical properties and bonding abilities. Quantum models also anticipate subtle changes in energy levels caused by strong nuclear forces.
Computational Chemistry Insights
Computational chemistry uses advanced software to simulate Roentgenium’s electron structure. These simulations use quantum mechanics and relativity combined. They generate data on orbital energies, shapes, and electron interactions.
Results show that Roentgenium’s electrons experience strong relativistic effects. These effects alter the expected order of orbital filling. Computational studies predict that the element behaves like other group 11 metals but with unique features. This insight guides experiments and theoretical research on superheavy elements.
Applications And Research
Roentgenium is a superheavy element with a complex electron configuration. Its unique structure attracts scientists worldwide. Understanding this element helps reveal fundamental atomic behaviors. Research focuses on its properties, stability, and potential uses. Due to its rarity and instability, practical applications remain limited. Scientists continue exploring its features through experiments and theoretical models.
Current Scientific Studies
Researchers study Roentgenium to understand heavy element chemistry. They analyze its electron configuration and nuclear properties. Experiments use particle accelerators to create and observe Roentgenium atoms. These studies test theories about electron shells in superheavy elements. Scientists also investigate how Roentgenium interacts with other atoms. Current work aims to map its place in the periodic table. Such research expands knowledge about atomic structure and forces.
Potential Future Uses
Roentgenium’s potential applications remain mostly theoretical. Its radioactivity could lead to new insights in nuclear physics. Some hope it may help develop advanced materials or catalysts. The element’s unique electron setup might influence future technologies. However, creating enough Roentgenium for practical use is a challenge. Continued research may unlock new scientific tools or methods. Its role in science can grow as more is learned.

Credit: www.youtube.com

Credit: www.youtube.com
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element Zinc (Zn). It shows filled 3d and 4s orbitals.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration is usually 2, 8, 18 for the third shell. The third shell can hold up to 18 electrons, not just 8.
Is Element 119 Theoretically Possible?
Element 119 is theoretically possible but remains unconfirmed. Scientists aim to create it via nuclear fusion in particle accelerators. Its extreme instability and short half-life make detection difficult. If synthesized, element 119 would start the eighth period of the periodic table.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn). It is a transition metal with atomic number 25.
Conclusion
Roentgenium’s electron configuration reveals its unique atomic structure. It follows patterns seen in heavy transition metals. Scientists predict its electrons fill orbitals in complex ways. The element’s position in the periodic table guides this arrangement. Understanding its configuration helps in studying superheavy elements.
Though unstable, Roentgenium provides insight into atomic behavior at high atomic numbers. Research continues to clarify its properties and electron distribution. This knowledge supports advancements in chemistry and physics fields. Exploring Roentgenium deepens our grasp of the periodic table’s limits.



No comments