Electron Configuration of Darmstadtium
Have you ever wondered how the electrons arrange themselves in one of the heaviest elements known to science? Darmstadtium, with its elusive nature and complex structure, holds secrets that can unlock a deeper understanding of chemistry’s frontier.
By exploring the electron configuration of darmstadtium, you’ll uncover how its electrons fill energy levels, why this matters for its properties, and what makes it so unique compared to lighter elements. This guide will make the complex clear, helping you grasp the fundamentals and nuances of darmstadtium’s electron setup—so keep reading and empower your knowledge of this fascinating superheavy element.
Credit: www.schoolmykids.com
Basic Properties
Darmstadtium is a synthetic element with unique properties. Understanding its basic properties helps grasp its place in chemistry. These properties include its atomic number, symbol, and position in the periodic table. Each reveals essential details about this rare element.
Atomic Number And Symbol
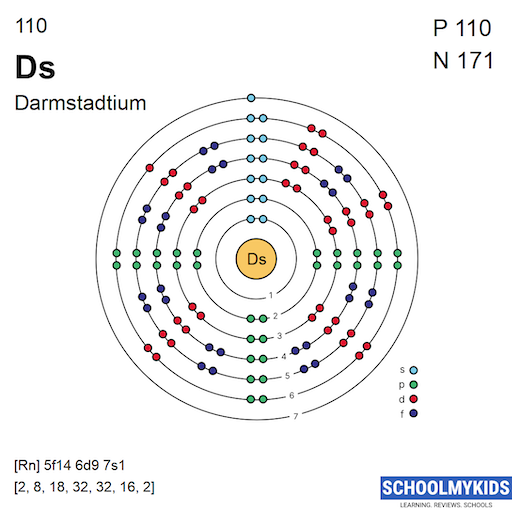
Darmstadtium has the atomic number 110. This means it has 110 protons in its nucleus. Its chemical symbol is Ds. The symbol Ds comes from the element's name. Atomic number defines its identity in the periodic table.
Position In Periodic Table
Darmstadtium is located in group 10 of the periodic table. It belongs to the d-block elements. It is part of the transition metals category. Darmstadtium is in period 7, the seventh row. This position reflects its electron configuration and properties.

Credit: winter.group.shef.ac.uk
Electron Shell Structure
The electron shell structure of Darmstadtium defines how its electrons arrange in various energy levels around the nucleus. This arrangement affects the element's chemical and physical properties. Understanding this structure helps explain Darmstadtium's place in the periodic table and its behavior in reactions.
Darmstadtium is a superheavy element with complex electron arrangements. Its electron shells follow patterns seen in other heavy transition metals but with unique relativistic effects due to its high atomic number.
Principal Energy Levels
Darmstadtium has electrons filling principal energy levels from 1 to 7. These levels are labeled with numbers 1 through 7, representing shells closest to farthest from the nucleus. Electrons fill the lower energy levels first before moving to higher ones.
The seventh energy level is the outermost shell for Darmstadtium. It contains the electrons that mostly influence its chemical properties. The sixth and seventh levels hold the electrons in the d and s orbitals, respectively. These levels are key to understanding Darmstadtium’s electron configuration.
Subshell Filling Order
Electrons fill subshells in a specific order based on energy levels. For Darmstadtium, the filling follows the pattern of d and s orbitals in the sixth and seventh shells. The 6d subshell fills before the 7s subshell completes.
The typical electron configuration ends with 6d and 7s electrons. This order follows the Aufbau principle but is affected by relativistic effects. These effects cause shifts in orbital energies, slightly changing electron filling order in superheavy elements like Darmstadtium.
Ground State Configuration
The ground state configuration of darmstadtium reveals how its electrons are arranged in the lowest energy state. Darmstadtium is a superheavy element with the atomic number 110. Scientists cannot observe its electron configuration directly. Instead, they rely on theoretical models and predictions.
Understanding the ground state helps explain the chemical behavior of darmstadtium. It also shows how it fits into the periodic table. Due to its high atomic number, relativistic effects influence its electron arrangement.
Predicted Electron Arrangement
The predicted ground state electron configuration of darmstadtium is [Rn] 5f14 6d8 7s2. This means it follows radon’s electron core, then fills 5f, 6d, and 7s orbitals. The 6d orbital holds eight electrons, and the 7s orbital holds two. Some sources suggest a slightly different arrangement, like 6d9 7s1, but this is less common.
Relativistic effects cause shifts in orbital energy levels. These shifts make the 6d orbital more stable and favor the 6d8 7s2 arrangement. This arrangement matches trends seen in other group 10 elements like platinum.
Comparison Of Models
Two main models predict darmstadtium’s electron configuration: the non-relativistic and the relativistic model. The non-relativistic model ignores high-speed electron effects. It tends to predict the configuration as 6d9 7s1. This model is simpler but less accurate for heavy elements.
The relativistic model includes corrections for electrons moving near light speed. It predicts 6d8 7s2 as the ground state. This model is more reliable for superheavy elements like darmstadtium. It explains the orbital contractions and energy shifts observed in calculations.
Overall, the relativistic model aligns better with chemical trends. It helps scientists understand darmstadtium’s behavior in theory. This knowledge aids research into superheavy element chemistry.

Credit: chemistry.stackexchange.com
Relativistic Effects
Darmstadtium is a superheavy element with complex electron behavior. Relativistic effects influence its electron configuration strongly. These effects occur because electrons move at speeds close to light speed near the nucleus. This changes how orbitals behave and how electrons fill them.
Orbital Contraction
Relativistic speeds cause orbitals, especially s-orbitals, to shrink or contract. This contraction happens because electrons gain more mass at high speeds. The shrinking pulls electrons closer to the nucleus. As a result, s-electrons become more tightly bound than predicted by simple models.
This contraction affects Darmstadtium’s 7s orbital the most. It lowers the size and energy of this orbital. The contracted s-orbital influences chemical properties and stability of the atom.
Energy Level Shifts
Relativistic effects also shift the energy levels of orbitals. In Darmstadtium, d and f orbitals change energy differently than s and p orbitals. The 6d orbitals rise in energy relative to the 7s orbital. This shift alters electron filling order.
The energy shifts cause electrons to fill orbitals in a way that differs from lighter elements. This explains the unusual electron configuration of Darmstadtium. Understanding these shifts helps predict its chemical behavior more accurately.
Electron Configuration Notations
Understanding the electron configuration notations of darmstadtium helps clarify its atomic structure. These notations describe how electrons fill the atom’s orbitals. Each notation style offers a unique way to represent electron arrangements clearly and concisely. Exploring these notations makes studying this superheavy element easier.
Standard Notation
The standard notation lists all electron shells and subshells filled in order. For darmstadtium, it shows electrons in each orbital explicitly. This notation uses numbers and letters, such as 1s, 2p, 3d, to denote orbitals. It helps visualize the exact electron count in each orbital. Though long, it provides full details about electron distribution.
Noble Gas Shorthand
The noble gas shorthand simplifies electron configuration. It uses the symbol of the nearest noble gas before darmstadtium to represent core electrons. For example, [Rn] stands for radon’s electron configuration. Then, only the electrons after radon are listed. This method shortens the notation and highlights outer electrons. It saves space and improves readability.
Quantum Number Representation
Quantum number representation describes electrons by their quantum states. It uses four numbers: principal, angular, magnetic, and spin quantum numbers. These numbers detail an electron’s energy level and position. For darmstadtium, this shows how electrons behave in complex orbitals. This notation is useful for advanced study and quantum chemistry. It explains electron properties beyond simple counting.
Comparison With Neighboring Elements
Comparing the electron configuration of darmstadtium with its neighboring elements reveals intriguing patterns. These patterns reflect its place in the periodic table and its chemical behavior. Examining similarities and differences helps to understand darmstadtium’s unique properties and its relation to nearby elements.
Similarities With Platinum Group
Darmstadtium shares many traits with platinum group metals. These elements have filled or nearly filled d-orbitals. Like platinum and iridium, darmstadtium’s electron configuration includes a d-block arrangement. This similarity suggests comparable chemical properties. They all exhibit strong metallic bonding and high density. Their electron structures support similar catalytic activities. These traits make darmstadtium part of this exclusive group despite its radioactivity.
Differences In Electron Arrangement
Darmstadtium’s electron arrangement shows subtle differences from its neighbors. It has a higher atomic number, affecting electron distribution. Relativistic effects cause shifts in its orbital energies. These shifts alter the filling order of orbitals slightly. For example, the 6d and 7s orbitals interact differently than in lighter elements. This causes variations in chemical reactivity and bonding. Such differences highlight the impact of atomic size and nuclear charge on electron structure.
Applications And Implications
Darmstadtium is a superheavy element with unique electron configuration. Understanding its applications and implications helps reveal its chemical behavior. This knowledge guides researchers in predicting how it might interact with other elements. The electron arrangement influences its stability and reactivity, which are key for scientific studies.
Due to its position in the periodic table, darmstadtium shows interesting properties linked to its electron structure. These properties might impact future research in chemistry and physics. Studying darmstadtium also challenges scientists to improve experimental methods for superheavy elements.
Impact On Chemical Properties
The electron configuration of darmstadtium shapes its chemical properties. Its electrons fill the 6d and 7s orbitals, affecting bonding and reactivity. This layout suggests darmstadtium behaves like other group 10 elements but with differences. Relativistic effects cause shifts in orbital energies, altering expected properties.
These effects may lead to lower reactivity than lighter homologs. Predictions indicate it could form unusual compounds or oxidation states. Understanding this helps chemists anticipate how darmstadtium might act in experiments. Its chemical nature remains mostly theoretical due to limited sample availability.
Challenges In Experimental Confirmation
Confirming darmstadtium’s electron configuration experimentally is difficult. It has a very short half-life and is produced in tiny amounts. These facts limit the ability to study its chemical traits directly. Advanced instruments and techniques are needed to detect its electron arrangement.
High radiation and rapid decay make experiments complex and expensive. Researchers rely on indirect methods and theoretical models for insights. These challenges slow progress but push innovation in experimental chemistry. Each step forward improves knowledge of superheavy elements like darmstadtium.
Frequently Asked Questions
What Is The Electron Configuration Of Darmstadtium?
Darmstadtium’s electron configuration is [Rn] 5f14 6d8 7s2. Relativistic effects influence its orbital structure significantly.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 4?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴ corresponds to the element sulfur.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn).
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell can be either 2 8 8 or 2 8 18 depending on the element and its energy level filling.
Conclusion
Darmstadtium’s electron configuration shows its place in the periodic table. It follows patterns seen in other heavy elements. Relativistic effects affect its electron behavior significantly. Scientists use this to predict chemical properties. Understanding its configuration helps in studying superheavy elements.
Although rare, Darmstadtium’s structure teaches us about atomic theory. Each discovery adds to knowledge of chemistry’s complexity. Exploring these elements opens doors to new science.



No comments