Electron Configuration of Oganesson
Have you ever wondered how electrons arrange themselves around the heaviest known element, Oganesson? Understanding the electron configuration of Oganesson is not just a matter of curiosity—it unlocks insights into its chemical behavior and its place on the periodic table.
But here’s the catch: Oganesson’s electron configuration is anything but straightforward. Because it is a superheavy, synthetic element, its electrons behave in unusual ways that challenge traditional rules. If you want to grasp what makes Oganesson unique and why its electron arrangement fascinates scientists, keep reading.
This article will guide you through the basics and the complexities, making it easy for you to understand this mysterious element’s electronic blueprint.
Basic Properties Of Oganesson
Oganesson is one of the most fascinating elements in the periodic table. It has unique properties due to its position and atomic structure. Understanding its basic properties helps reveal its nature and behavior. This section covers its place in the periodic table, atomic details, and special characteristics.
Position In Periodic Table
Oganesson belongs to group 18, the noble gases. It is located at the bottom of this group. This position suggests it shares some traits with other noble gases. However, its properties might differ due to its high atomic number. It marks the end of the 7th period on the table.
Atomic Number And Mass
Oganesson has the atomic number 118. This means it has 118 protons in its nucleus. Its atomic mass is approximately 294 atomic mass units. These values make it the heaviest element currently known. The large number of protons impacts its electron arrangement.
Synthetic And Radioactive Nature
Oganesson does not occur naturally. Scientists create it in laboratories through nuclear reactions. It is highly radioactive, breaking down quickly into lighter elements. Its short half-life limits the study of its properties. Safety measures are crucial when working with this element.

Credit: www.shutterstock.com
Electron Shells And Orbitals
The electron shells and orbitals of oganesson reveal how its electrons arrange around the nucleus. This arrangement affects the element’s chemical behavior. Oganesson, with atomic number 118, has a complex structure due to its many electrons. Understanding its shells and orbitals helps us grasp its unique properties.
Electron Shell Capacity
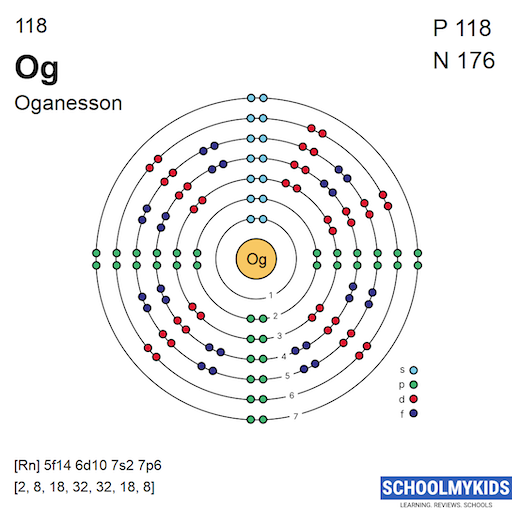
Electron shells hold electrons in fixed energy levels around the nucleus. Each shell can hold a limited number of electrons. The first shell holds up to 2 electrons. The second and third shells can each hold up to 8 electrons. The fourth and fifth shells can hold up to 18 electrons. The sixth shell can carry 32 electrons, and so can the seventh shell, where oganesson’s outer electrons are located.
Oganesson’s 118 electrons fill these shells following their capacity limits. The outermost shell is the seventh, which contains the valence electrons responsible for chemical reactions.
Orbital Types And Energy Levels
Orbitals are regions where electrons are likely to be found. They come in different shapes: s, p, d, and f. The s orbitals are spherical and hold up to 2 electrons. The p orbitals are dumbbell-shaped and hold up to 6 electrons. The d orbitals hold up to 10 electrons, while the f orbitals hold up to 14 electrons.
Electrons fill orbitals based on energy levels, starting with the lowest. In heavy elements like oganesson, relativistic effects can change these energy levels slightly. This makes the electron behavior more complex than in lighter elements.
Filling Order In Heavy Elements
The filling order of orbitals follows the Aufbau principle, which means electrons fill the lowest energy orbitals first. For oganesson, the filling goes beyond the known shells of lighter elements. Electrons occupy the 7s, 5f, 6d, and 7p orbitals.
Oganesson’s electron configuration ends in the 7p orbital. This completion of the 7p shell marks it as a noble gas. Still, strong relativistic effects cause differences in how electrons fill orbitals here compared to lighter noble gases.
Oganesson Electron Configuration
Oganesson is the heaviest element in the periodic table. It has the atomic number 118. Scientists study its electron configuration to understand its chemical behavior. Oganesson is a noble gas but may act differently from others in its group. Its electron arrangement is unique due to its high atomic number and special effects.
Unabbreviated Configuration
The full electron configuration of oganesson lists all occupied orbitals. It starts from 1s and goes up to 7p. The configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 4f¹⁴ 5s² 5p⁶ 5d¹⁰ 5f¹⁴ 6s² 6p⁶ 6d¹⁰ 7s² 7p⁶. This completes 118 electrons around the nucleus.
Noble Gas Core And Valence Electrons
Oganesson’s core matches the noble gas radon (Rn). Radon has 86 electrons filling inner shells. The valence electrons are in the 7s and 7p orbitals. Specifically, oganesson has 7s² and 7p⁶ valence electrons. These outer electrons define its chemical properties.
Relativistic Effects On Configuration
Relativistic effects change electron behavior in heavy elements. Oganesson’s electrons move very fast, near light speed. This alters their mass and energy levels. These effects cause the 7p orbitals to split and shift energy. Such changes may make oganesson more reactive than lighter noble gases.
Credit: www.schoolmykids.com
Relativistic Influence On Electrons
Oganesson is the heaviest noble gas, with unique electron behavior. Its electrons move at speeds close to light. This causes relativistic effects that change their properties. These effects alter how electrons arrange and interact around the nucleus. Understanding these changes is key to studying Oganesson's electron configuration.
Spin-orbit Coupling
Spin-orbit coupling is stronger in Oganesson than lighter elements. It happens because electrons move very fast near the nucleus. The electron’s spin interacts with its orbit, splitting energy levels. This splitting affects the atom's electron configuration and stability. It leads to unusual arrangements of electrons in outer shells.
Electron Density Distribution
Relativistic effects cause electron clouds to spread differently in Oganesson. Electrons are pulled closer to the nucleus but also pushed outward in some areas. This changes the shape and size of electron density around the atom. Such changes affect how Oganesson interacts with other atoms and molecules.
Impact On Chemical Behavior
The relativistic changes alter Oganesson’s chemical properties. Unlike other noble gases, it may show unexpected reactivity. Its electron configuration suggests it could form compounds more easily. Scientists believe these effects make Oganesson less inert than lighter noble gases.
Comparisons With Other Noble Gases
Oganesson, the heaviest noble gas, stands out in the periodic table. Comparing it with other noble gases reveals interesting trends. These differences arise from its unique position and electron configuration. Understanding these comparisons helps clarify Oganesson’s chemical and physical behavior.
Trends Down The Group
Noble gases show clear trends moving down the group. Atomic size increases with each element. Oganesson has the largest atomic radius among them. Its outer electrons are farther from the nucleus. This causes weaker attraction between nucleus and electrons. As a result, Oganesson’s properties differ from lighter noble gases.
Reactivity Differences
Most noble gases are very unreactive. Their full outer shells make them stable. Oganesson breaks this pattern slightly. Theoretical studies predict it to be more reactive. Relativistic effects influence its electron behavior. This effect increases reactivity compared to xenon or radon. Thus, Oganesson may form compounds more easily.
Electron Configuration Variations
Oganesson’s electron configuration is unique among noble gases. It fills the 7p orbital, unlike lighter elements. Strong relativistic effects cause unexpected shifts. These shifts alter orbital energies and shapes. This leads to subtle changes in its chemical properties. The configuration contrasts with the simple ns2 np6 pattern seen in others.
Challenges In Configuration Determination
Determining the electron configuration of oganesson poses unique challenges. This element has a very high atomic number, leading to strong relativistic effects. These effects alter electron behavior, making predictions difficult. Experimental data is scarce due to oganesson's short half-life and rarity. Scientists rely heavily on theoretical models and computational methods. Each approach offers different insights but also introduces uncertainties. Understanding these challenges helps explain why oganesson's electron configuration remains debated.
Theoretical Vs Experimental Data
Experimental data on oganesson is limited. Its extreme radioactivity prevents direct measurement of electron arrangements. Theoretical calculations fill this gap, using quantum mechanics principles. These models predict electron shells and subshells. Some predictions match known trends in noble gases. Others differ due to relativistic effects unique to superheavy elements. The lack of experimental confirmation leaves room for multiple interpretations.
Different Ground State Models
Scientists propose several ground state electron configurations for oganesson. Some models extend the expected noble gas pattern. Others suggest new electron filling orders. These differences arise from how relativistic effects are treated. Changes in orbital energies affect which electrons fill first. The models try to balance these effects with known chemical properties. This leads to competing views on oganesson's true ground state.
Computational Chemistry Approaches
Advanced computational methods help explore oganesson’s electron structure. Techniques like Dirac-Fock and density functional theory are common. They simulate relativistic interactions and electron correlations. Results vary depending on computational parameters and assumptions. These studies provide valuable predictions but not definitive answers. Computational chemistry remains essential for studying elements beyond experimental reach.
Chemical Implications
The chemical implications of oganesson's electron configuration reveal unusual traits. Oganesson, element 118, belongs to the noble gases group. Its electron setup differs from lighter noble gases due to relativistic effects. These effects influence the behavior of its electrons, changing chemical properties. Scientists predict interesting outcomes for its reactivity, possible compounds, and stability.
Studying oganesson helps understand superheavy elements and their place in the periodic table. The chemical implications guide researchers in exploring its potential uses and limits.
Predicted Reactivity
Oganesson may show higher reactivity than other noble gases. Its outer electrons are less tightly held. This is due to relativistic changes that weaken electron binding. Such changes could allow oganesson to form bonds more easily. This contrasts with lighter noble gases, which rarely react. Oganesson's reactivity might resemble some metals more than noble gases.
Possible Compounds
The formation of oganesson compounds remains mostly theoretical. Scientists suggest oganesson could bond with halogens or alkali metals. These compounds might be stable under extreme conditions. Predicted molecules include oganesson fluorides and oxides. Their properties would differ from known noble gas compounds. Research continues to identify the exact nature of these potential compounds.
Stability And Radioactivity Effects
Oganesson is highly unstable and radioactive. Its half-life lasts only milliseconds to seconds. This short lifespan limits chemical studies and compound creation. Radioactive decay affects electron arrangement and chemical behavior. The instability adds complexity to understanding its chemistry. Despite this, insights into oganesson help expand knowledge of superheavy elements.

Credit: www.youtube.com
Frequently Asked Questions
What Element Has An Electron Configuration Of 1s 2 2s 2 2p 6 3s 2 3p 4?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴ belongs to sulfur (S). Sulfur has 16 electrons and is a nonmetal.
Why Are 2,8,8,18,18,32 Called Magic Numbers?
2, 8, 8, 18, 18, 32 are called magic numbers because they represent fully filled electron shells. Fully filled shells give atoms extra stability. These numbers correspond to the maximum electrons each shell can hold. This concept helps explain atomic structure and chemical behavior.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ is Ruthenium (Ru).
What Is The 2 8 8 18 18 Rule?
The 2 8 8 18 18 rule describes the maximum electron capacity of the first five atomic shells. It helps predict electron arrangement in atoms.
Conclusion
Oganesson’s electron configuration is unique and complex. It reflects its place as a superheavy noble gas. Relativistic effects influence its electrons strongly. This makes Oganesson different from lighter noble gases. Scientists still study its properties to learn more. Understanding its electron setup helps explain its chemistry.
The element remains fascinating despite limited experimental data. Its configuration shows how atoms behave under extreme conditions. Oganesson challenges our knowledge of the periodic table. Exploring it deepens insight into atomic structure and behavior.



No comments