Electron Configuration of Tennessine
Have you ever wondered how the electrons arrange themselves in one of the rarest elements on the periodic table? Understanding the electron configuration of Tennessine can unlock fascinating insights into its chemical behavior and place in the periodic table.
Whether you’re a student tackling chemistry homework or simply curious about this mysterious, superheavy element, knowing how its electrons are organized will deepen your grasp of atomic structure. You’ll discover exactly how Tennessine’s electrons are distributed and why this matters for its properties and potential uses.
Keep reading to unravel the secrets hidden in Tennessine’s electron configuration and boost your understanding of the elements around you.

Credit: www.youtube.com
Basic Properties Of Tennessine
Tennessine is a superheavy element with unique properties. It belongs to the halogen group but shows unusual behavior. Understanding its basic properties helps us grasp its place in chemistry.
Scientists study tennessine mainly through experiments. Its short half-life makes detailed research difficult. Still, its atomic structure reveals important information.
Atomic Number And Symbol
Tennessine has the atomic number 117. Its chemical symbol is Ts. This number shows it has 117 protons in its nucleus. The atomic number helps identify and classify the element.
Position In The Periodic Table
Tennessine is in group 17, the halogen group. It sits in period 7, the seventh row. This position links tennessine to other halogens like iodine and astatine. It lies in the p-block, where outer electrons fill p-orbitals.
Physical And Chemical Characteristics
Tennessine is a radioactive element with a very short half-life. It is expected to be a solid under normal conditions. Chemically, it may behave like other halogens but with some differences. It likely forms compounds by gaining one electron. Its heavy atomic mass affects its reactivity and bonding.
Electron Shell Structure
The electron shell structure shows how electrons arrange around an atom's nucleus. This structure helps explain an element's chemical behavior. Tennessine is a rare and heavy element with unique electron distribution. Its shell structure follows patterns found in the periodic table. Understanding this helps scientists predict how Tennessine reacts with other elements.
Total Number Of Electrons
Tennessine has 117 electrons. This matches its atomic number. Each electron occupies a specific energy level or shell. The total count defines Tennessine's chemical properties. Electrons fill shells in order from lower to higher energy. This filling follows the Pauli exclusion principle and Hund's rule. The large number of electrons makes Tennessine complex to study.
Distribution Across Energy Levels
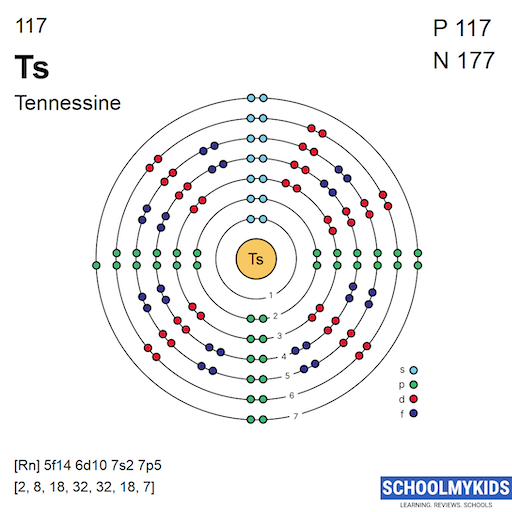
Electrons in Tennessine spread across seven main energy levels. The inner shells fill first with lower energy electrons. Outer shells contain electrons with higher energy. The distribution follows this pattern: 2, 8, 18, 32, 32, 18, and 7 electrons in each shell. The last shell holds valence electrons, which influence bonding. Tennessine’s outer shell electrons give it properties similar to halogens.
Long-form Electron Configuration
The long-form electron configuration shows how electrons fill all orbitals in an atom. It lists every subshell occupied by electrons from the lowest to the highest energy levels. This detailed notation helps understand the distribution of electrons in complex elements like Tennessine.
Tennessine, with atomic number 117, has electrons arranged in many shells and subshells. The long-form configuration reveals the exact order in which these electrons fill the orbitals. It follows the principles of quantum mechanics and the Pauli exclusion rule. This helps scientists predict chemical behavior and bonding patterns of Tennessine.
Filling Order Of Orbitals
Electrons fill orbitals starting from the lowest energy level first. The order follows the Aufbau principle. For Tennessine, electrons fill orbitals in this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, and finally 7s and 5f orbitals. This order respects the increasing energy of each subshell.
In Tennessine, the 7p orbital is partially filled last, making it part of the halogen group. The filling stops after the 7p subshell gains five electrons. This pattern shows similarities with other halogens but includes effects from relativistic electrons due to its high atomic number.
Complete Electron Configuration Notation
The complete electron configuration of Tennessine is written by listing each subshell and its electron count. It starts from the innermost shell and moves outward. The notation is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p⁶ 7s² 5f¹⁴ 6d¹⁰ 7p⁵
This full notation shows Tennessine’s electrons filling up to the 7p subshell. Each superscript indicates the number of electrons in that subshell. This format helps chemists and students visualize the element’s electron structure clearly.
Abbreviated Electron Configuration
The abbreviated electron configuration helps show how electrons are arranged in an atom. It uses a noble gas symbol to shorten the full electron configuration. This method makes it easier to understand and write the electron setup of elements, especially those with many electrons like tennessine. Tennessine is a heavy element with many electrons, so its abbreviated electron configuration is very useful.
Using Noble Gas Core
The noble gas core is a shortcut in electron configuration. It starts with the nearest noble gas before the element. For tennessine, the noble gas core is radon, which has 86 electrons. This core replaces the inner electrons that do not change. It simplifies the configuration by focusing only on the electrons added after radon.
Simplified Notation For Tennessine
Tennessine has an atomic number of 117. Its abbreviated electron configuration begins with [Rn], the symbol for radon. After radon, the electrons fill the 5f, 6d, 7s, and 7p orbitals. The full notation is [Rn] 5f14 6d10 7s2 7p5. This shows the outermost electrons and their positions clearly. Using this short form saves space and makes it easy to read.
Comparison With Other Halogens
Tennessine belongs to the halogen group in the periodic table. It shares many traits with other halogens like fluorine, chlorine, bromine, and iodine. Comparing its electron configuration helps us see patterns in this group. This comparison also highlights unique features caused by its higher atomic number. Understanding these similarities and differences reveals Tennessine’s place among halogens.
Similarities In Electron Configuration
Like all halogens, Tennessine has seven electrons in its outermost shell. This is a key trait that defines halogens. These seven valence electrons make Tennessine highly reactive. It tends to gain one electron to complete its outer shell. The electron configuration ends with a p^5 sublevel for all halogens, including Tennessine. This shared pattern explains why halogens have similar chemical properties.
Differences Due To Atomic Number
Tennessine’s atomic number is much higher than other halogens. It has 117 protons, compared to 9 in fluorine or 53 in iodine. This large atomic number adds many inner electrons. These electrons fill higher energy levels and sublevels, like 7p and 6d orbitals. The inner electrons cause stronger electron shielding effects. This changes how Tennessine’s outer electrons behave. Its electron cloud is larger and more complex than lighter halogens.
Relativistic effects also influence Tennessine’s electron configuration. These effects occur at high atomic numbers. They cause electrons to move faster and gain more mass. This changes orbital shapes and energy levels. Such changes do not happen in lighter halogens. These differences affect Tennessine’s chemical and physical properties.
Relativistic Effects On Electron Configuration
The electron configuration of tennessine, element 117, shows unique traits. These traits arise from relativistic effects. Relativistic effects occur due to electrons moving near the speed of light. This changes how electrons behave around the nucleus. It impacts the energy and shape of atomic orbitals. Understanding these effects helps explain tennessine’s special properties.
Influence On Orbital Energies
Relativistic effects cause orbitals to shift in energy. Inner electrons move very fast, increasing their mass slightly. This makes s and p orbitals contract and lower in energy. Meanwhile, d and f orbitals expand and rise in energy. For tennessine, this changes the order of orbital filling. The 7p orbitals split into two groups with different energies. This splitting affects how electrons fill these orbitals.
Impact On Chemical Behavior
The changes in orbital energies affect tennessine’s chemistry. Its outer electrons are held more tightly. This lowers the element's reactivity compared to lighter halogens. Tennessine may show unusual oxidation states due to these effects. It can form bonds differently than expected. These behaviors make tennessine an interesting subject for chemistry research.
Electron Configuration And Reactivity
Tennessine is a superheavy element with unique electron configuration and reactivity. Its electron arrangement shapes how it interacts chemically. Understanding this helps predict its behavior among halogens.
The electron configuration influences Tennessine’s chemical properties and reactivity. This section explores its expected chemical traits and possible oxidation states.
Expected Chemical Properties
Tennessine’s electron configuration ends in the 7p orbitals. It resembles other halogens like iodine and astatine. This suggests it may form similar compounds. Its large atomic size may reduce reactivity compared to lighter halogens. It could show some metallic character due to relativistic effects. The element might be less reactive and more unstable.
Potential Oxidation States
Tennessine likely shows oxidation states of -1, +1, +3, and +5. The -1 state matches typical halogen behavior. Positive states arise from its ability to lose p electrons. Higher oxidation states may appear due to relativistic stabilization of orbitals. These states help form compounds with metals and nonmetals. Such variety indicates complex chemistry despite its rarity.

Credit: www.shutterstock.com
Methods To Determine Electron Configuration
Determining the electron configuration of tennessine requires specialized methods. Tennessine is a superheavy element, making direct observation difficult. Scientists rely on both experimental and theoretical approaches. These methods help predict how electrons arrange around its nucleus. Understanding these methods offers insight into the element's chemical behavior and properties.
Experimental Techniques
Direct experimental methods for tennessine are limited. The element exists only briefly and in tiny amounts. Scientists use advanced instruments like spectroscopy to study decay products. They analyze emitted radiation and energy levels. This data gives clues about electron arrangements. Particle accelerators create tennessine atoms for these tests. Experiments often focus on chemical reactions with known elements. These reactions hint at the electron structure indirectly.
Theoretical Calculations
Theoretical models play a major role in electron configuration. Quantum mechanics helps predict electron positions and energies. Scientists use computer simulations to solve complex equations. These calculations consider relativistic effects due to tennessine’s high atomic number. Different models compare predicted results with known elements. Theoretical results fill gaps left by experimental limits. They provide probable electron configurations and guide future research.
Credit: www.schoolmykids.com
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 represents the element calcium (Ca) with atomic number 20.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The element with the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ is Ruthenium (Ru). It has atomic number 44 and belongs to the transition metals group.
Which Element Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 belongs to Chromium (Cr), atomic number 24.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration can be either 2 8 8 or 2 8 18, depending on the element. Early shells hold 2 and 8 electrons. The third shell can hold 8 or expand to 18 electrons due to available d-orbitals.
Conclusion
Tennessine’s electron configuration reveals its place in the periodic table. It follows the pattern of heavy halogens but shows unique properties. Understanding its electrons helps predict chemical behavior and reactivity. This knowledge aids scientists in exploring superheavy elements. Studying Tennessine expands our grasp of atomic structure and element classification.
The topic remains exciting for future research and discovery.



No comments