Electron Configuration of Mercury
Have you ever wondered what makes mercury so unique among the elements? The secret lies deep within its electron configuration.
Understanding how mercury's electrons are arranged can reveal why it behaves the way it does—its shiny appearance, its liquid state at room temperature, and its chemical reactions. If you want to uncover the mysteries behind this fascinating metal and see how its electron setup influences its properties, keep reading.
This simple guide will break it down for you, step by step, making complex science easy and interesting. Your curiosity is about to be rewarded!
Basic Electron Configuration
Mercury’s electron configuration shows how its 80 electrons fill energy levels. The pattern follows the order of orbitals around the nucleus. This arrangement explains many of mercury’s chemical properties.
Understanding Electron Configuration
Electron configuration shows how electrons are arranged in an atom. It helps explain chemical properties and behavior. Each electron occupies a specific energy level and orbital.
Mercury's electron configuration is unique because it is a heavy metal. It has many electrons filling different shells and subshells.
Mercury’s Basic Electron Configuration
Mercury has 80 electrons. These electrons fill the shells starting from the lowest energy level.
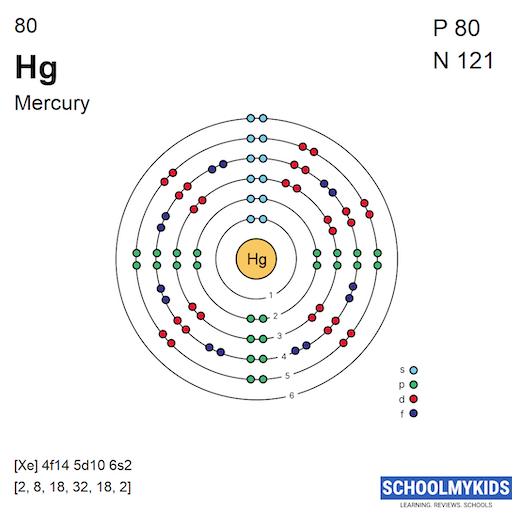
The basic electron configuration of mercury is: [Xe] 4f14 5d10 6s2. This shows the distribution of electrons in orbitals.
Electron Shells And Subshells In Mercury
Mercury’s electrons fill shells in a specific order. The inner shells fill first, followed by outer shells.
The 4f and 5d subshells are fully filled in mercury. This gives mercury its stable electron structure.
The outermost electrons are in the 6s subshell. These electrons influence mercury’s chemical reactions.
Mercury's Position In The Periodic Table
Mercury is a unique element in the periodic table. It is a heavy metal with interesting properties. Its place in the table tells us about its behavior and characteristics.
Mercury is found in the transition metals section. It belongs to Group 12 and Period 6. This position shows it shares traits with zinc and cadmium.
Group And Period
Mercury is in Group 12. This group includes zinc and cadmium. These metals have similar electron arrangements.
Mercury is in Period 6. Elements in this period have electrons filling the 6th energy level. This affects their size and reactivity.
Block Classification
Mercury is part of the d-block elements. The d-block contains transition metals. These metals often have multiple oxidation states.
Being in the d-block means mercury has electrons in d orbitals. This influences its chemical behavior and bonding.
Relation To Neighboring Elements
Mercury lies between gold and thallium. Gold shares some physical traits with mercury. Thallium belongs to a different group but is close in the table.
Its neighbors help explain mercury’s unique liquid state at room temperature. This is rare among metals.
Filling Order Of Orbitals
The filling order of orbitals explains how electrons fill the energy levels in an atom. Electrons fill orbitals starting from the lowest energy to the highest. This order follows specific rules to keep the atom stable.
For mercury, understanding this order helps us see how its electrons arrange in different shells and subshells. The way electrons fill these orbitals affects mercury's chemical behavior and properties.
Principles Guiding Orbital Filling
Electrons fill orbitals based on three main principles. The first is the Aufbau principle, which states electrons occupy the lowest energy orbitals first. Next is the Pauli exclusion principle, meaning each orbital holds two electrons with opposite spins. Lastly, Hund's rule says electrons fill orbitals singly before pairing up.
Order Of Orbital Filling For Mercury
Mercury has 80 electrons. These electrons fill orbitals in this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, and 6p. Each orbital fills completely before electrons move to the next higher energy level.
Unique Filling In D And F Orbitals
Mercury’s electrons fill the 5d orbital after the 6s orbital fills. This is because 5d has slightly higher energy but fills after 6s. The 4f orbitals fill even later, influencing mercury’s complex electron structure.
Detailed Orbital Breakdown
The electron configuration of mercury reveals how its electrons fill different orbitals. This arrangement explains many of mercury’s chemical properties. Each orbital holds a specific number of electrons in a defined order.
Understanding mercury’s orbitals helps us see how its atoms behave and interact with others. The breakdown starts from the lowest energy level to the highest.
S-orbitals In Mercury
Mercury’s s-orbitals hold electrons closest to the nucleus. The 1s, 2s, 3s, and 4s orbitals fill first, each holding two electrons. The outermost s-orbital, 6s, contains two electrons that influence mercury’s reactivity.
P-orbitals In Mercury
p-Orbitals hold up to six electrons each. Mercury’s 2p, 3p, 4p, and 5p orbitals are fully occupied. These orbitals contribute to the shape and energy of mercury atoms.
D-orbitals In Mercury
Mercury has filled d-orbitals in its 3d, 4d, and 5d shells. Each d-orbital holds up to ten electrons. These electrons affect mercury’s color and bonding behavior.
F-orbitals In Mercury
Mercury’s electron structure does not include f-orbitals. Elements heavier than mercury often fill these orbitals. Mercury’s electron count stops before the f-orbitals begin.
Comparison With Other Transition Metals
Mercury's electron configuration is unique among transition metals. It has a full d-subshell, unlike many others with partially filled d-orbitals. This affects its chemical properties and stability compared to other elements in the same group.
Electron Configuration Of Mercury
Mercury has a unique electron setup among transition metals. Its configuration ends with 5d10 6s2. This full d-subshell gives mercury special properties.
Electron Configuration Of Other Transition Metals
Most transition metals have partially filled d-orbitals. For example, iron ends with 3d6 4s2. These incomplete d-orbitals cause their typical metallic behavior.
Unlike mercury, many transition metals easily lose d electrons. This loss helps them form various oxidation states.
Impact On Chemical Properties
Mercury’s full d-subshell means it is less reactive than others. It often shows only +1 and +2 oxidation states.
Other transition metals show multiple oxidation states. This variety makes them useful in different reactions and compounds.
Physical Differences Due To Electron Configuration
Mercury is a liquid at room temperature. This unusual trait links to its electron structure and weak metallic bonding.
Most transition metals are solid and have stronger bonds. Their partially filled d-orbitals strengthen metallic connections.
Relativistic Effects On Mercury
Mercury is a unique element with special atomic properties. These properties come from the effects of relativity on its electrons. Relativistic effects happen because electrons near mercury’s nucleus move very fast. This speed changes how the electrons behave and how mercury’s atoms form.
These effects impact mercury’s electron configuration. They make mercury different from other elements in its group. Understanding these changes helps explain mercury’s unusual chemical and physical traits.
Relativistic Contraction of s-OrbitalsElectrons in mercury’s 6s orbital move close to light speed. This causes a contraction of the 6s orbital. The orbital shrinks and holds electrons tighter. This contraction lowers the energy of the 6s electrons. It makes these electrons less likely to participate in chemical reactions.
Expansion of d and f OrbitalsWhile s orbitals contract, d and f orbitals expand slightly. This expansion changes how electrons fill these orbitals. It also affects the shape and size of mercury’s electron cloud. These changes influence mercury’s bonding ability and reactivity.
Impact on Mercury’s Chemical PropertiesRelativistic effects cause mercury to be less reactive. The tightly held 6s electrons resist bonding with other atoms. This makes mercury a liquid metal at room temperature. It also explains mercury’s low melting point compared to its neighbors.
Electron Configuration And Chemical Properties
Electron configuration plays a key role in defining mercury’s chemical nature. It explains how electrons arrange themselves around mercury atoms. This arrangement affects how mercury reacts with other elements. Understanding this helps us grasp mercury’s unique properties.
Mercury has a full set of electrons in its inner shells. Its outer electrons are arranged in a way that makes it less reactive than many metals. This influences mercury’s behavior in chemical reactions and its physical state at room temperature.
Electron Configuration Of Mercury
Mercury has 80 electrons. These electrons fill different energy levels and sublevels. The full configuration is [Xe] 4f14 5d10 6s2. This means mercury’s 6s electrons are the outermost. The 5d and 4f electrons form a stable inner core.
This stable arrangement makes mercury less likely to lose or gain electrons. It is why mercury is liquid at room temperature and does not easily form compounds.
Chemical Properties Linked To Electron Configuration
Mercury’s electron setup leads to low reactivity. It does not react quickly with air or water. Mercury forms compounds mainly with halogens and sulfur. These reactions involve the outer 6s electrons.
The filled 5d and 4f orbitals provide extra stability. Mercury’s chemistry is different from other metals in the same group. Its electron structure limits the number of oxidation states it can have.
Impact On Mercury’s Oxidation States
Mercury mostly shows +1 and +2 oxidation states. The +2 state is more common. The electron configuration explains why higher oxidation states are rare. Removing electrons beyond the 6s shell needs more energy.
This limits mercury’s ability to form complex ions. It also affects its bonding with other atoms. The stable electron arrangement keeps mercury less reactive overall.

Credit: www.youtube.com
Mercury's Oxidation States
Mercury is a unique metal with interesting oxidation states. These states describe how many electrons mercury loses or gains when it forms compounds. Understanding these states helps explain mercury’s chemical behavior and uses.
Mercury mainly shows two common oxidation states. Each state has different properties and forms various compounds. These compounds can be found in nature and used in industry.
Oxidation State +1
Mercury can lose one electron, forming the +1 oxidation state. This state is rare and less stable. It usually appears in compounds where two mercury atoms bond together. These compounds are called mercurous compounds.
Mercurous chloride (Hg2Cl2) is a common example. It has mercury atoms connected by a bond sharing one electron each. This state is important in some chemical reactions but less common than +2.
Oxidation State +2
The +2 oxidation state is the most common for mercury. Mercury loses two electrons in this state. It forms many stable compounds, called mercuric compounds.
Mercuric oxide (HgO) and mercuric chloride (HgCl2) are examples. These compounds are more reactive and used in various applications. The +2 state explains much of mercury’s chemistry.
Less Common Oxidation States
Mercury rarely shows other oxidation states. Some complex compounds may have mercury in unusual states. These are not common and usually unstable.
Scientists study these rare states to understand mercury’s full chemical nature. They help in advanced research but have limited practical use.
Role In Mercury's Toxicity
Mercury’s electron configuration affects how it binds with other elements, leading to its toxic effects. Its unique arrangement makes it react strongly with proteins in the body. This reaction disrupts normal cell functions and causes harm.
Electron Configuration And Chemical Behavior
Mercury’s electron configuration influences its chemical reactions. Its outer electrons are arranged as 5d10 6s2. These electrons are tightly held, making mercury less reactive than other metals. This stability affects how mercury interacts with living organisms and the environment.
Formation Of Toxic Compounds
Mercury forms toxic compounds by bonding with sulfur and carbon. These bonds arise because of its electron arrangement. Compounds like methylmercury are highly toxic. They easily enter the food chain and accumulate in fish and humans.
Interaction With Biological Molecules
Mercury binds strongly to proteins and enzymes in the body. This binding disrupts normal cell functions. The electron configuration allows mercury to attach to sulfur atoms in proteins. This attachment causes damage to the nervous system and organs.

Credit: www.chegg.com
Applications Influenced By Electron Configuration
Electron configuration plays a key role in determining how an element behaves. Mercury's electron arrangement affects its properties and uses. This section explores how mercury’s electron configuration influences its applications in different fields.
Mercury In Thermometers And Electrical Devices
Mercury has a unique electron setup that makes it liquid at room temperature. This property allows mercury to expand and contract with temperature changes. It conducts electricity well because of its electron flow. These traits make mercury useful in thermometers and electrical switches.
Use In Amalgams For Dental Work
Mercury’s electrons enable it to mix easily with metals like silver. This creates amalgams, which dentists use for fillings. The electron structure helps mercury bond with other metals tightly. This bonding provides durability and strength to dental materials.
Role In Scientific Instruments
Mercury’s electron configuration gives it high density and reflectivity. These features make mercury valuable in scientific tools. Devices like barometers and vacuum pumps rely on mercury’s stable electron behavior. This ensures accuracy and longevity in measurements.
Credit: www.schoolmykids.com
Frequently Asked Questions
What Is The Electron Configuration Of Mercury?
Mercury's electron configuration is [Xe] 4f14 5d10 6s2. It shows a filled d-subshell and s-subshell. This explains mercury's chemical properties and stability.
Why Is Mercury's Electron Configuration Important?
Understanding mercury's electron configuration helps explain its reactivity and bonding behavior. It also aids in predicting its placement in the periodic table.
How Does Mercury's Electron Configuration Affect Its Properties?
The filled 5d10 and 6s2 orbitals make mercury dense and less reactive. This unique configuration results in mercury being liquid at room temperature.
What Is The Significance Of The 5d10 Subshell In Mercury?
The 5d10 subshell is fully filled in mercury, contributing to its chemical inertness and metallic characteristics. It stabilizes the atom's structure.
Conclusion
Mercury’s electron configuration reveals its unique chemical traits. It holds electrons in filled d and s orbitals. This setup affects how mercury bonds with other elements. Understanding this helps explain mercury’s behavior in nature and industry. Simple knowledge, big impact.
Keep exploring elements to see how electron patterns shape our world.



No comments