Electron Configuration of Gold
Have you ever wondered what makes gold so unique on the atomic level? Understanding the electron configuration of gold can unlock the secrets behind its dazzling shine, incredible conductivity, and chemical behavior.
You’ll discover exactly how the electrons are arranged in a gold atom and why this arrangement matters to you—whether you’re curious about chemistry, interested in materials science, or just love learning fascinating facts about one of the most precious metals on Earth.
Keep reading, and you’ll gain clear insights that make this complex topic easy to grasp.
Atomic Structure Of Gold
Gold's electron configuration shows how its electrons are arranged in shells. This setup affects gold's chemical and physical traits. Electrons fill specific orbitals, giving gold its unique properties.
Overview Of Gold’s Atomic Structure
Gold is a chemical element with the symbol Au and atomic number 79. Its atomic structure defines its unique properties. The atom has a nucleus with protons and neutrons. Around the nucleus, electrons move in specific patterns called shells and subshells. These electrons determine how gold reacts and bonds with other elements.
Electron Shells And Energy Levels
Gold’s electrons are arranged in energy levels or shells. Each shell holds a limited number of electrons. The shells fill from the closest to the nucleus outward. Gold has electrons in five main shells. The outermost shell contains electrons that affect gold’s chemistry most.
Subshells And Orbital Configuration
Each shell divides into smaller parts called subshells: s, p, d, and f. These subshells hold electrons in orbitals. Gold’s electrons fill subshells in a specific order. The 5d and 6s subshells play a key role in gold’s behavior. The arrangement explains gold’s color and conductivity.

Credit: www.youtube.com
Basics Of Electron Configuration
Electron configuration shows how electrons are arranged around an atom's nucleus. It helps explain many chemical properties of elements. Each electron occupies a specific energy level and sublevel.
Understanding electron configuration is important to grasp the behavior of atoms in different reactions. It reveals the number of electrons in each shell and subshell.
What Is Electron Configuration?
Electron configuration is a way to describe where electrons are in an atom. Electrons fill energy levels from lowest to highest. This order follows specific rules based on quantum mechanics.
Energy Levels And Sublevels
Electrons live in energy levels, also called shells. Each shell has smaller parts called sublevels: s, p, d, and f. Each sublevel can hold a limited number of electrons.
How Electrons Fill Sublevels
Electrons fill the lowest energy sublevels first. The order is s, then p, then d, and finally f. The filling pattern affects the element’s properties and its place in the periodic table.
Electron Shells And Subshells
Electron shells and subshells describe how electrons are arranged around an atom’s nucleus. These shells form layers, each holding a limited number of electrons. Subshells divide these shells into smaller parts with specific shapes and energy levels. Understanding these arrangements helps explain gold’s chemical behavior and properties.
Gold’s electron configuration depends on filling its shells and subshells in a precise order. The way electrons fill each shell affects gold’s stability and reactivity. Exploring these shells and subshells gives insight into gold’s unique place in the periodic table.
What Are Electron Shells?
Electron shells are energy levels surrounding the nucleus. Each shell can hold a certain number of electrons. The first shell holds up to 2 electrons, the second up to 8, and the numbers increase for outer shells. Electrons fill the lowest energy shells first. This order is important for understanding gold’s electron layout.
Understanding Subshells
Subshells are parts of electron shells with different shapes and energy. They are labeled s, p, d, and f. Each subshell holds a fixed number of electrons: s holds 2, p holds 6, d holds 10, and f holds 14. Electrons fill subshells in a specific sequence, which affects how gold behaves chemically.
Electron Distribution In Gold
Gold has 79 electrons arranged in shells and subshells. Its configuration shows electrons filling up to the 5d and 6s subshells. The 5d subshell is partially filled, which influences gold’s color and conductivity. This distribution also impacts gold’s ability to form bonds.
Gold’s Position In The Periodic Table
Gold holds a special place in the periodic table. It is a transition metal with unique properties. Understanding its position helps explain its electron configuration and behavior.
Gold is found in group 11 and period 6. This location influences its chemical and physical characteristics. It shares the group with copper and silver, metals known for conductivity and malleability.
Position In Group 11
Group 11 contains elements known as coinage metals. Gold’s neighbors, copper and silver, have similar electron arrangements. These metals have a filled d-subshell and one electron in the outer s-orbital. This setup affects their conductivity and resistance to corrosion.
Placement In Period 6
Period 6 includes heavy elements with many electron shells. Gold’s electrons fill up to the 6th energy level. The presence of 5d and 6s electrons defines its chemistry. The large atomic size affects its density and melting point.
Relation To Transition Metals
Gold is a transition metal, which means it has partially filled d-orbitals. This gives gold its unique color and chemical reactivity. Transition metals often form colored compounds and have multiple oxidation states. Gold’s stable +1 and +3 states are common in compounds.
Ground State Configuration Of Gold
The ground state configuration of gold shows how its electrons arrange in the atom. This arrangement follows specific rules based on energy levels. These rules help predict gold’s chemical and physical behavior.
Understanding gold’s electron setup helps explain why it behaves uniquely in reactions. It also reveals why gold has its distinctive color and conductivity.
What Is Ground State Electron Configuration?
Ground state electron configuration means the lowest energy arrangement. Electrons fill orbitals starting with the lowest energy first. This stable state forms the basis for understanding the atom’s properties.
Gold’s Atomic Number And Electron Count
Gold has an atomic number of 79. This means it has 79 electrons in a neutral atom. These electrons fill several energy levels and sublevels around the nucleus.
Detailed Electron Configuration Of Gold
Gold’s configuration starts with filling the 1s orbital. Then electrons fill higher orbitals in order. The full configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s¹ 4f¹⁴ 5d¹⁰.
This setup shows that gold has a single electron in the 6s orbital. It also has a filled 5d sublevel, which affects its chemistry.
Why Gold’s Electron Configuration Is Unique
Gold’s electron arrangement differs from many other elements. The full 5d sublevel and one electron in 6s create special stability. This pattern results from electron interactions and energy considerations.
This unique setup gives gold its color and high resistance to corrosion.
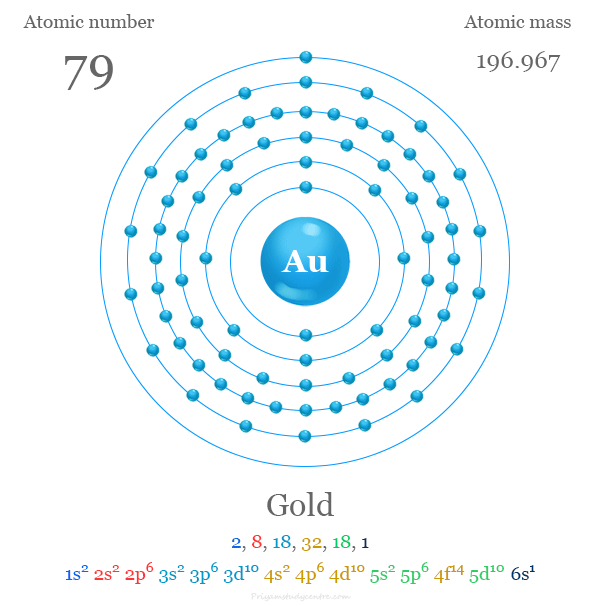
Credit: www.priyamstudycentre.com
Exceptions In Gold’s Electron Arrangement
Gold’s electron arrangement does not follow the simple rules you expect. It shows unique exceptions that make its structure special. These exceptions happen due to the way electrons fill the orbitals around the nucleus.
Understanding these exceptions helps explain gold’s properties like color and reactivity. The arrangement is not just about filling orbitals in order. Instead, it balances stability and energy, which causes some unusual patterns.
Electron Configuration Of Gold
Gold has 79 electrons to arrange around its nucleus. Normally, electrons fill the 5d and 6s orbitals in order. The expected configuration is [Xe] 4f14 5d9 6s2. But gold differs from this pattern.
Why Gold’s Electron Arrangement Is Unique
Gold’s actual electron configuration is [Xe] 4f14 5d10 6s1. One electron moves from the 6s orbital to the 5d orbital. This shift creates a full 5d shell, which is more stable.
Role Of Stability In Electron Distribution
Electrons prefer to fill orbitals to reach maximum stability. A full d-subshell is more stable than a partially filled one. So, gold’s electrons rearrange to achieve this stability. This exception is common in transition metals.
Role Of Relativistic Effects
Gold's electron configuration shows unusual behavior compared to lighter elements. This happens because of relativistic effects. These effects arise from electrons moving very fast near the gold nucleus.
Fast-moving electrons gain extra mass and energy. This changes how electrons fill the orbitals. It also shifts the energy levels of the orbitals.
What Are Relativistic Effects?
Relativistic effects occur when electrons move close to the speed of light. This happens in heavy elements like gold. The speed changes the electron's mass and shape of orbitals. These changes affect chemical properties and electron arrangement.
Impact On Gold’s 6s And 5d Orbitals
In gold, the 6s orbital shrinks and stabilizes. The 5d orbitals expand and rise in energy. This shift causes electrons to fill the 5d orbitals differently. The result is a unique electron configuration for gold.
Why Gold’s Color Is Linked To Relativistic Effects
Gold's shiny yellow color comes from relativistic effects. These effects change how gold absorbs and reflects light. Electrons absorb blue light more, making gold appear yellow. This feature is rare among metals.
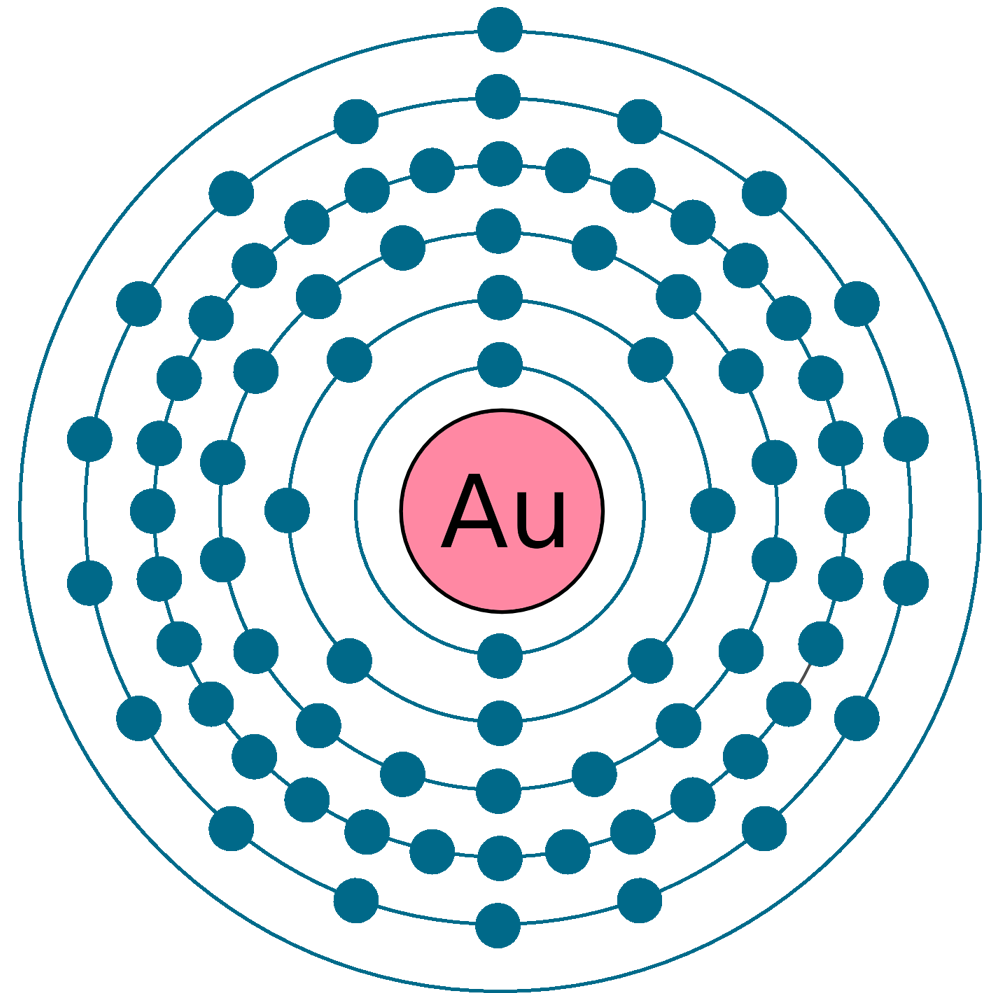
Credit: www.a-c-g.it
Impact On Gold’s Chemical Properties
Gold’s electron configuration shapes how it reacts with other elements. Its unique arrangement of electrons causes special chemical behaviors. This affects its color, stability, and ability to bond.
Understanding these effects helps explain gold’s value in science and industry. The electron structure controls gold’s softness and resistance to corrosion.
Stability And Resistance To Corrosion
Gold’s electron configuration makes it very stable. It does not easily lose or gain electrons. This stability prevents gold from rusting or reacting with air.
This resistance to corrosion keeps gold shiny and bright for a long time. It is why gold is used in jewelry and electronics.
Color And Optical Properties
Gold’s electron arrangement causes its distinct yellow color. Electrons absorb and reflect light differently than in other metals.
This unique light interaction gives gold its warm and rich appearance. Other metals like silver appear white or gray because of their electrons.
Bonding And Chemical Reactivity
Gold forms fewer chemical bonds compared to other metals. Its full d-subshell holds electrons tightly, limiting bonding sites.
This low reactivity means gold rarely forms compounds. It reacts mostly with strong acids or halogens under special conditions.
Comparison With Other Transition Metals
Gold’s electron configuration differs from many transition metals by filling the 5d and 6s orbitals uniquely. This arrangement gives gold its distinct chemical and physical properties. Comparing it helps understand its special place in the periodic table.
Electron Configuration Of Gold
Gold's electron configuration is unique among transition metals. It ends with 5d10 6s1, showing a filled d-subshell and a single s electron. This arrangement affects gold's chemical behavior and color.
Electron Configuration Of Other Transition Metals
Most transition metals have partially filled d-orbitals. For example, iron has 3d6 4s2. Copper is similar to gold but ends with 3d10 4s1. These differences influence their reactivity and properties.
Stability Differences In Electron Configurations
Gold's full d-subshell provides extra stability. This stability is less common in early transition metals. Their d-orbitals are often only partially filled. Stability affects their chemical bonds and uses.
Impact On Physical And Chemical Properties
Gold's electron setup leads to high corrosion resistance. Other metals may oxidize or rust easily. The electron configuration also affects conductivity and color. Gold's unique setup gives it a distinct yellow shine.
Applications Related To Electron Configuration
Gold's electron configuration shapes many of its unique uses. The way electrons are arranged affects its color, conductivity, and chemical behavior. These properties make gold valuable beyond jewelry.
Understanding electron configuration helps explain gold's role in technology and medicine. Each application depends on how gold's electrons interact with other elements and materials.
Electrical Conductivity And Electronics
Gold's electron arrangement allows easy flow of electric current. This makes gold an excellent conductor. It is used in connectors, switches, and circuit boards. The stable electron shell prevents corrosion, ensuring long-lasting performance.
Catalysis In Chemical Reactions
Gold can speed up chemical reactions without being consumed. Its electron configuration creates active sites on its surface. These sites help convert harmful gases and improve fuel efficiency. Scientists study gold catalysts for cleaner industrial processes.
Medical Applications And Drug Delivery
Gold nanoparticles interact well with biological systems. Their electron configuration helps them bind to cells and tissues. This property aids in targeted drug delivery and diagnostic imaging. Gold is safe and effective in many medical treatments.
Frequently Asked Questions
What Is The Electron Configuration Of Gold?
The electron configuration of gold (Au) is [Xe] 4f14 5d10 6s1. This arrangement shows filled d and f subshells with one electron in 6s.
Why Does Gold Have A Unique Electron Configuration?
Gold's electron configuration is unique due to relativistic effects. These effects alter electron energies, causing the 6s orbital to hold a single electron.
How Does Gold's Electron Configuration Affect Its Properties?
Gold's electron configuration influences its high conductivity, malleability, and resistance to corrosion. The single 6s electron plays a key role in these traits.
What Is The Significance Of 5d10 In Gold's Electron Structure?
The filled 5d10 subshell in gold contributes to its chemical stability and distinctive color. These electrons are crucial in bonding and metallic behavior.
Conclusion
Gold’s electron configuration reveals its unique chemical traits. Its arrangement shows why gold is stable and shiny. Understanding these electrons helps explain gold’s place in the periodic table. This knowledge supports learning about metals and their uses. Remembering gold’s electron setup makes chemistry clearer and more fun.
Keep exploring elements to see how electrons shape our world.



No comments