Electron Configuration of Francium
Are you curious about what makes francium such a unique element? Understanding the electron configuration of francium can unlock the secrets behind its chemical behavior and place in the periodic table.
By diving into how its electrons are arranged, you’ll gain clear insights that make this rare element less mysterious and more fascinating. Keep reading to discover exactly how francium’s electron setup shapes its properties—and why this knowledge could change the way you see the elements around you.
Credit: commons.wikimedia.org
Francium Basics
Francium is a rare and highly radioactive metal. It belongs to the alkali metals group. This element is the heaviest in its group and very unstable.
Scientists have studied francium only in small amounts. It is hard to find because it breaks down quickly. Its properties are similar to other alkali metals but with unique differences.
What Is Francium?
Francium is element number 87 on the periodic table. It has the symbol "Fr". This element was discovered in 1939. It is named after France.
Francium is very rare in nature. Only tiny amounts exist naturally. It forms when uranium or thorium decays.
Physical Properties Of Francium
Francium is a soft metal. It looks silvery but darkens quickly in air. It melts at about 27 degrees Celsius. This low melting point makes it one of the easiest metals to melt.
Because it is radioactive, francium has a short lifespan. It decays into other elements fast. This limits its use in experiments.
Where Is Francium Found?
Francium is found in trace amounts in uranium ores. It forms through radioactive decay processes. The amount is extremely small and hard to collect.
Most francium used in labs is made artificially. Scientists create it in particle accelerators. This helps study its properties safely.

Credit: chemistnotes.com
Atomic Structure Of Francium
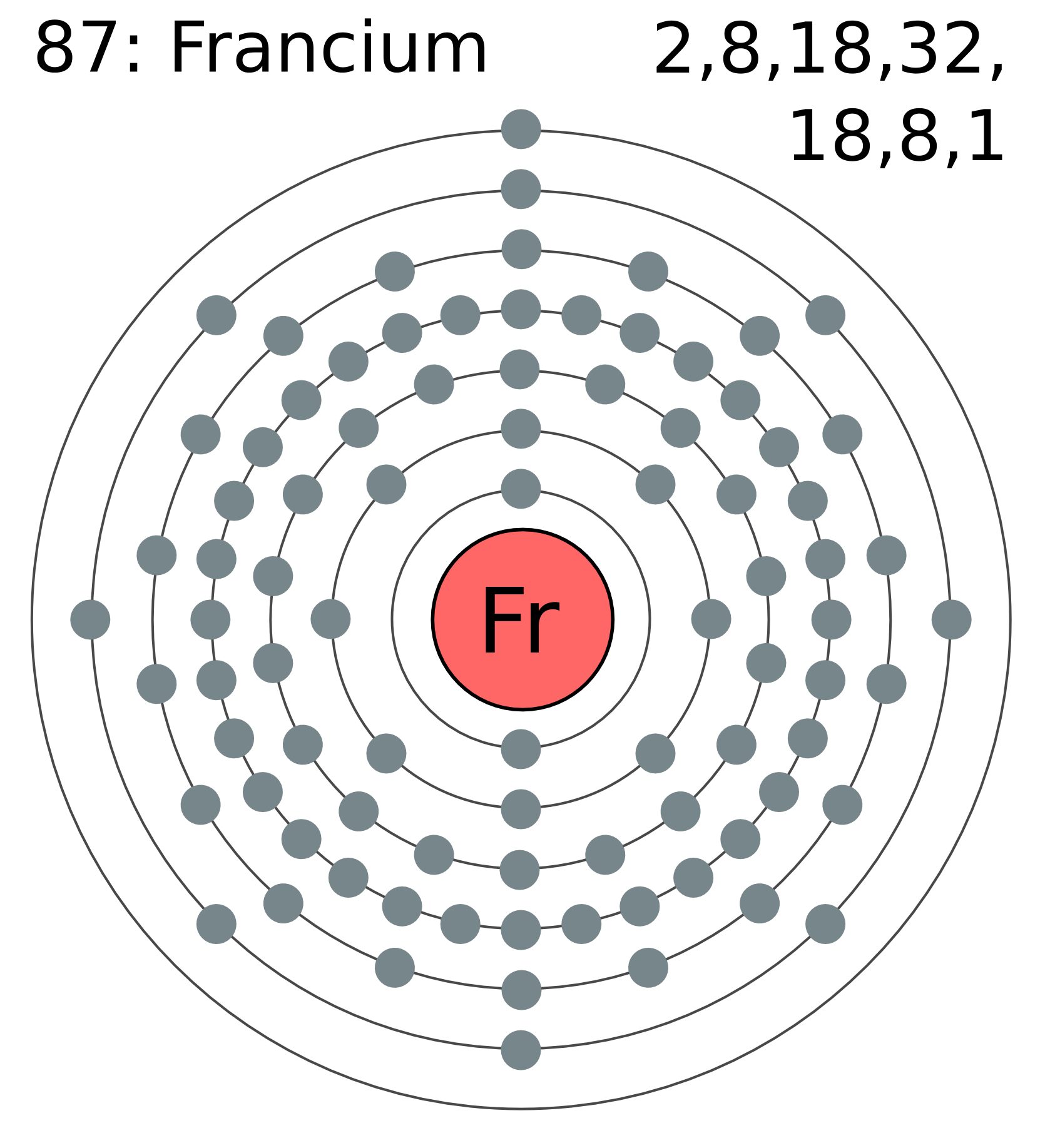
Francium has a simple electron setup with 87 electrons arranged in shells. Its electron configuration ends in 7s1, showing one electron in the outer shell. This setup helps explain francium’s chemical behavior.
Electron Shells Of Francium
Francium has a complex atomic structure with many electron shells. It contains 87 electrons arranged in different shells. These shells are labeled from the innermost to the outermost as K, L, M, N, O, P, and Q. Each shell holds a specific number of electrons. The outermost shell of francium contains just one electron.
Subshell Arrangement In Francium
Inside each shell, electrons fill smaller groups called subshells. These are named s, p, d, and f. Francium’s electrons fill these subshells in a specific order based on energy levels. The last electron enters the 7s subshell, making francium highly reactive. This single electron in the outer shell defines many of francium’s properties.
Significance Of Electron Configuration
The electron configuration explains francium’s position in the periodic table. It is in group 1, the alkali metals. Its single outer electron makes it very unstable and reactive. This electron easily leaves to form positive ions. Understanding this helps predict francium’s chemical behavior and reactions.
Electron Shells And Subshells
Understanding electron shells and subshells is key to grasping francium's electron configuration. Electrons orbit the nucleus in layers called shells. Each shell holds a specific number of electrons.
Within these shells are smaller groups called subshells. Subshells come in types: s, p, d, and f. Each type has a different shape and capacity. These shapes affect how electrons fill the atom.
Electron Shells Of Francium
Francium has seven electron shells. Each shell holds more electrons as you move outward. The first shell holds 2 electrons, while outer shells hold many more. The seventh shell contains the outermost electron of francium.
Subshell Structure In Francium
Francium's subshells fill in a specific order. It starts with the 1s subshell and moves up to 7s. The 7s subshell has one electron. This electron defines francium's chemical properties.
Importance Of Subshells In Electron Configuration
Subshells determine how electrons arrange in an atom. Each subshell fills based on energy levels. Lower energy subshells fill before higher ones. This pattern explains francium’s unique electron setup.
Francium Electron Configuration
Francium has a simple electron setup with 87 electrons filling its shells. Its electron configuration ends with 7s¹, showing it is in the seventh period. This setup helps explain francium’s chemical behavior.
Francium is a rare and highly radioactive element. Its electron configuration shows how electrons are arranged in its atoms. Understanding this helps explain its chemical behavior and place in the periodic table.
Francium has an atomic number of 87. This means it has 87 electrons orbiting its nucleus. These electrons fill energy levels and sublevels in a specific order. The arrangement follows the rules of quantum mechanics.
How Francium's Electrons Are Arranged
The electron configuration of francium starts with the lower energy levels. These levels fill up completely before electrons occupy higher levels. The pattern follows the sequence of s, p, d, and f orbitals.
Its configuration ends with a single electron in the 7s orbital. This single electron is key to francium’s reactivity. The full configuration is written as [Rn] 7s¹, where [Rn] represents the electron setup of radon.
Why Electron Configuration Matters For Francium
The single electron in the 7s orbital makes francium very reactive. It behaves like other alkali metals, which also have one electron in their outermost shell. This lone electron is easily lost during chemical reactions.
This explains francium’s strong tendency to form positive ions. The electron configuration helps predict its chemical properties and reactions with other elements. It also shows francium’s place in Group 1 of the periodic table.
Role Of Valence Electrons
Valence electrons in francium determine its chemical properties and reactivity. Its electron configuration shows one valence electron in the 7s orbital. This single electron makes francium highly reactive and unstable.
Understanding Valence Electrons In Francium
Valence electrons are the outermost electrons in an atom. They control how an element reacts with others. For francium, these electrons are crucial in defining its chemical behavior.
Francium has one valence electron in its outer shell. This single electron makes francium very reactive. It easily loses this electron to form positive ions.
How Valence Electrons Affect Francium’s Reactivity
The valence electron in francium is loosely held. This causes francium to react quickly with other substances. It often forms compounds by giving away its valence electron.
This high reactivity is typical of alkali metals. Francium’s valence electron plays a key role in its strong chemical activity.
Valence Electrons And Francium’s Position In The Periodic Table
Francium is in Group 1 of the periodic table. Elements in this group have one valence electron. This shared trait explains similarities in their chemical properties.
The single valence electron determines francium’s placement and behavior. It is why francium behaves like other alkali metals.
Francium In The Periodic Table
Francium is one of the rarest elements on Earth. It belongs to the group called alkali metals. These metals are very reactive and found in the first column of the periodic table.
Francium is placed in period 7, which means it has seven energy levels. It sits below cesium and above no other element in its group. This position helps us understand its properties and behavior.
Position And Group
Francium is in group 1, the alkali metals. Elements in this group have similar chemical features. They usually have one electron in their outer shell. This electron makes them highly reactive.
Period And Energy Levels
Francium is in period 7, the last row of the table. This period has seven electron shells. Francium’s electrons fill these shells from the inside out. The outermost shell contains one electron.
Electron Configuration Basics
The electron configuration shows how electrons arrange around the nucleus. For francium, it helps explain its high reactivity. It also shows why francium behaves like other alkali metals.
Comparison With Other Alkali Metals
Francium shares traits with lithium, sodium, potassium, rubidium, and cesium. All have one electron in their outer shell. Francium’s larger size affects its chemical reactions. This makes it more reactive than cesium.
Comparison With Alkali Metals
Francium’s electron configuration is similar to other alkali metals, with one electron in the outermost shell. This single outer electron defines its chemical behavior. Compared to lighter alkali metals, francium’s electrons occupy higher energy levels.
Electron Configuration Of Francium
Francium is the heaviest alkali metal in the periodic table. Its electron configuration shows how electrons are arranged in its atoms. This arrangement affects its chemical properties and reactions. Francium has a unique electron setup compared to other alkali metals.
Similarities With Other Alkali Metals
All alkali metals have one electron in their outermost shell. This single electron makes them very reactive. Francium, like lithium, sodium, and potassium, has this lone outer electron. It defines their position in Group 1 of the periodic table.
Francium’s electron configuration ends with 7s¹. This matches the pattern of alkali metals ending with ns¹. This similarity explains why francium behaves like its lighter cousins.
Differences Due To Higher Energy Levels
Francium’s electrons fill higher energy levels than other alkali metals. It has electrons in the 7th shell, while others have fewer shells filled. This affects its size, reactivity, and stability. Francium atoms are larger and less stable than lighter alkali metals.
The electron cloud in francium is more spread out. This causes weaker attraction between the outer electron and the nucleus. As a result, francium loses its outer electron more easily than others.
Impact On Chemical Behavior
The outer electron’s position influences francium’s chemical reactions. Francium reacts quickly with water and air. Its reactivity is higher than cesium, the next alkali metal above it. The electron configuration makes francium very unstable and short-lived.
Despite these differences, francium still forms similar compounds. It forms ionic bonds by losing its outer electron. This behavior is common to all alkali metals.

Credit: www.pointlomahigh.com
Relativistic Effects On Electrons
Electrons in heavy elements like francium behave differently. Their speed near the nucleus is very high. This causes changes in how electrons arrange around the atom. These changes are called relativistic effects.
Relativistic effects alter the energy and shape of electron orbitals. They affect the chemical properties of francium too. Understanding these effects helps explain francium’s unique behavior.
Relativistic Speed Of Electrons
Electrons close to the francium nucleus move almost at light speed. This high speed increases their mass slightly. The increased mass changes the way electrons orbit the nucleus. It causes inner electrons to pull closer to the nucleus.
Impact On Orbital Energies
Relativistic effects lower the energy of s orbitals in francium. This makes s electrons more tightly bound. Other orbitals, like p and d, are less affected. The shift in orbital energies changes francium’s chemical reactions.
Contraction And Expansion Of Orbitals
S orbitals contract due to relativistic effects. This contraction pulls electrons closer to the nucleus. Meanwhile, outer orbitals expand slightly. This expansion happens because of less attraction to the nucleus. These changes affect how francium bonds with other elements.
Chemical Properties Linked To Configuration
The chemical behavior of francium is deeply connected to its electron configuration. Francium has one electron in its outermost shell. This single electron shapes how francium reacts with other elements. Its position in the periodic table also plays a key role.
Francium belongs to the alkali metals group. Elements in this group share similar chemical properties. The electron configuration of francium causes it to be highly reactive. This reactivity impacts how francium forms compounds and bonds.
Reactivity Due To Single Valence Electron
Francium has one electron in the 7s orbital. This electron is easy to lose during reactions. Losing this electron forms a positive ion, Francium⁺. This ion forms strong bonds with non-metals. Such reactivity makes francium a very active metal.
Formation Of Ionic Compounds
Francium tends to form ionic bonds by donating its valence electron. It reacts mostly with halogens and oxygen. The electron transfer creates stable ionic compounds. These compounds have francium ions and negatively charged ions. This behavior is typical for alkali metals.
Low Ionization Energy And Its Effects
The ionization energy of francium is very low. This means it takes little energy to remove the outer electron. Low ionization energy increases francium’s chemical activity. It reacts quickly with water and air. This property limits francium’s natural occurrence and stability.
Applications And Research On Francium
Francium is a rare and highly unstable element. It exists only in trace amounts in nature. Its short half-life limits many practical uses. Despite this, scientists are very interested in francium for research. The element helps us learn about atomic structure and nuclear physics.
Researchers study francium to better understand electron behavior. Its electron configuration is unique among alkali metals. This makes francium valuable in scientific experiments. Research focuses on its atomic and nuclear properties.
Scientific Studies On Francium's Electron Configuration
Francium's electron arrangement helps scientists test atomic models. Its position in the periodic table predicts certain behaviors. Experiments confirm or challenge these predictions. This improves knowledge about heavy atoms and their electrons.
Francium In Nuclear Physics Research
Francium's unstable nature makes it important for nuclear studies. Researchers use it to study radioactive decay processes. It provides insights into nuclear reactions and forces. This research aids the understanding of atomic nuclei.
Potential Uses In Atomic Clocks
Scientists explore francium for use in atomic clocks. Its unique properties could improve clock accuracy. However, francium's short life limits current practical use. Experiments continue to explore its potential in precision timekeeping.
Frequently Asked Questions
What Is The Electron Configuration Of Francium?
Francium's electron configuration is [Rn] 7s¹. It has 87 electrons filling shells up to the 7th energy level. This configuration places Francium in the alkali metals group, showcasing its single valence electron in the 7s orbital.
Why Is Francium's Electron Configuration Important?
Francium's electron configuration explains its chemical properties and reactivity. Its single electron in the outer shell makes it highly reactive and similar to other alkali metals. Understanding this helps predict its behavior in chemical reactions and bonding.
How Does Francium's Electron Configuration Compare To Cesium?
Francium’s configuration [Rn] 7s¹ is similar to Cesium’s [Xe] 6s¹. Both have one valence electron in the s orbital. Francium is heavier and more unstable, but their configurations explain their similar chemical characteristics as alkali metals.
Can Francium's Electron Configuration Change In Compounds?
Francium typically loses its single 7s electron in compounds. This forms a +1 ion, Francium ion (Fr⁺). The electron configuration then resembles the stable radon core, [Rn], making it chemically reactive and eager to bond.
Conclusion
Francium’s electron configuration shows its place in the periodic table. It has a simple but important pattern. Understanding this helps explain its chemical behavior. Francium is rare and unstable, so it is less studied. Still, knowing its electrons helps scientists predict reactions.
This knowledge supports learning about other elements too. Electron patterns tell a story about each element’s nature. Keep exploring to see how elements connect and change.



No comments