Electron Configuration of Europium
Are you curious about what makes europium unique in the periodic table? Understanding the electron configuration of europium reveals the hidden pattern of its electrons and explains its special chemical behavior.
Whether you’re a student, a chemistry enthusiast, or just someone eager to learn, getting to know europium’s electron arrangement will deepen your grasp of how this fascinating element interacts and forms compounds. You’ll discover the step-by-step breakdown of europium’s electron configuration, why it matters, and how it influences the element’s properties.
Keep reading to unlock the secrets behind europium’s electrons and see how this knowledge can enhance your understanding of chemistry.
Basic Properties Of Europium
Europium is a rare earth element with unique chemical and physical properties. It belongs to the lanthanide series on the periodic table. Understanding its basic properties helps explain its behavior in compounds and applications.
This element has a silvery-white appearance and is soft enough to be cut with a knife. Europium is known for its high reactivity with air and water. It easily oxidizes, forming a protective oxide layer.
Atomic Number And Symbol
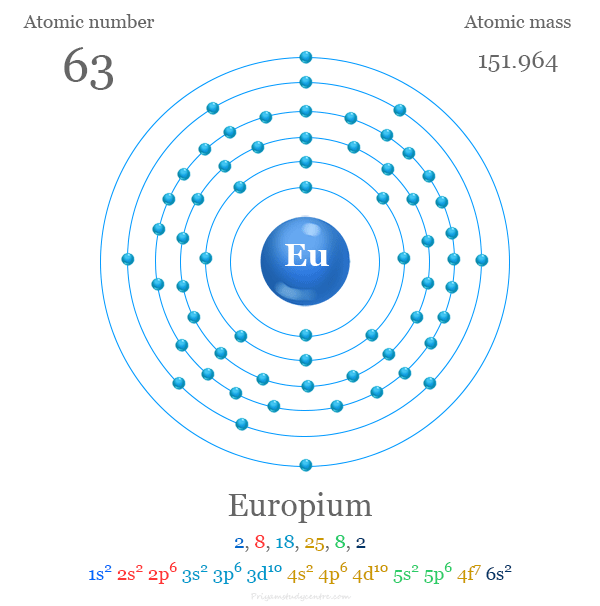
Europium has the atomic number 63. Its chemical symbol is Eu. The atomic number defines the number of protons in its nucleus.
Electron Configuration
The electron configuration of europium is [Xe] 4f7 6s2. This means it has seven electrons in the 4f orbital and two in the 6s orbital. This arrangement affects its chemical properties and magnetic behavior.
Physical Characteristics
Europium is a soft metal with a melting point around 826°C. It has a density of 5.24 grams per cubic centimeter. This metal is paramagnetic, meaning it is attracted to magnetic fields.
Chemical Reactivity
Europium easily forms compounds, mostly in the +2 and +3 oxidation states. It reacts quickly with oxygen and water. This reactivity influences how it is stored and handled.
Credit: www.priyamstudycentre.com
Atomic Structure And Number
The atomic structure of europium defines its unique chemical properties. It consists of a nucleus with protons and neutrons. Around the nucleus, electrons orbit in different energy levels or shells. Each shell holds a specific number of electrons.
Europium's atomic number tells how many protons are inside its nucleus. This number also equals the number of electrons in a neutral europium atom. Understanding this helps explain europium’s place in the periodic table and its behavior in reactions.
Atomic Number Of Europium
Europium has an atomic number of 63. This means it has 63 protons in its nucleus. The same number of electrons orbit around the nucleus. These electrons fill the shells starting from the lowest energy level.
Number Of Neutrons In Europium
Europium's most common isotope has 96 neutrons. Neutrons add to the atom's mass but have no charge. The total of protons and neutrons gives the atomic mass number. For europium, this is usually around 159.
Electron Shells And Orbitals
Electrons in europium fill shells labeled by energy levels: 1, 2, 3, and so on. Each shell contains subshells named s, p, d, and f. Europium's electrons occupy the 4f subshell, which is part of its unique chemical traits.
The filling order follows strict rules, with lower energy orbitals filling first. Europium’s electron configuration ends in the 4f subshell. This makes it one of the lanthanide series elements.
Electron Shell Capacities
Electron shell capacities explain how many electrons each shell can hold. This rule helps us understand the structure of atoms like europium.
Each electron shell corresponds to a principal energy level, labeled by n. The capacity of each shell follows a simple formula based on n.
Simplified Rule For Electron Shell Capacities
The 2n² rule states that each shell can hold up to 2 times n squared electrons. Here, n is the shell number (1, 2, 3, and so on).
For example, the first shell (n=1) holds up to 2 electrons. The second shell (n=2) holds up to 8 electrons.
Filling Order Of Electron Shells
Electrons fill shells starting from the lowest energy level. This filling order follows a specific sequence based on energy.
The shells fill as 1s, 2s, 2p, 3s, 3p, and continue higher. Europium’s electrons fill the shells up to 4f and 6s orbitals.
Key Concepts: Octet And 2n² Rules
The octet rule says atoms tend to have 8 electrons in their outer shell. It explains why atoms form bonds to complete their shell.
The 2n² rule applies to total electrons per shell, not just valence electrons. Europium’s electron shells follow this rule while filling inner and outer shells.
Orbital Filling Order
The orbital filling order explains how electrons fill the atomic orbitals in europium. Electrons occupy orbitals starting from the lowest energy level to higher ones. This order follows specific rules that shape the electron configuration.
Understanding the filling order helps reveal europium's chemical behavior. The arrangement influences its reactivity and bonding patterns. Europium, as a lanthanide, has electrons filling the 4f orbitals after the 6s and 5d orbitals.
Principle Of Aufbau In Europium
The Aufbau principle states electrons fill orbitals from low to high energy. For europium, the 6s orbital fills first with two electrons. Then electrons enter the 4f orbitals, which are higher in energy but fill before the 5d orbitals.
Pauli Exclusion Principle And Hund's Rule
The Pauli exclusion principle allows only two electrons per orbital with opposite spins. Hund's rule requires electrons to occupy empty orbitals singly before pairing. In europium, electrons spread across the seven 4f orbitals before pairing up.
Energy Levels And Subshells In Europium
Europium’s electron filling order follows energy levels 6s, then 4f, and finally 5d. The 4f subshell has seven orbitals, each holding up to two electrons. Europium has 63 electrons, with 4f orbitals partly filled, giving it unique properties.
Full Electron Configuration
Europium’s full electron configuration shows how its electrons fill energy levels. It is written as [Xe] 4f^7 6s^2, revealing seven electrons in the 4f orbital. This arrangement influences europium’s chemical behavior and placement in the periodic table.
Full Electron Configuration Of Europium
Europium is a lanthanide element with the atomic number 63. Its full electron configuration explains how electrons are arranged in its atoms. Understanding this helps in studying its chemical behavior and properties.
Electrons fill orbitals in a specific order based on energy levels. Europium’s electrons occupy shells and subshells from the lowest to higher energy states. This follows the Aufbau principle, which guides electron placement.
Electron Configuration Notation
The full electron configuration of Europium is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f⁷ 5d⁰ 6p⁶
This shows each shell and subshell filled with electrons. The numbers represent energy levels, letters denote orbital types, and superscripts show electron count.
Role Of 4f Orbital In Europium
Europium’s 4f orbital contains seven electrons. This half-filled 4f subshell provides extra stability to the atom. The 4f electrons influence europium’s magnetic and optical properties significantly.
Comparison With Neighboring Elements
Europium has more 4f electrons than samarium but fewer than gadolinium. Its electron configuration sets it apart in the lanthanide series. This affects how europium reacts and forms compounds.

Credit: www.chegg.com
Noble Gas Shorthand Configuration
The valence electrons of europium play a key role in its chemical behavior. These electrons are found in the outermost shells and determine how europium reacts with other elements. Understanding europium’s valence electrons helps explain its unique reactivity among the lanthanides.
Europium has electrons in the 4f, 5d, and 6s orbitals, but its valence electrons mainly come from the 6s and 4f orbitals. These electrons can be lost or shared during chemical reactions, affecting the element’s bonding and stability. Europium often forms compounds by losing two or three electrons, which influences its reactivity and the types of bonds it forms.
Valence Electron Configuration Of Europium
Europium’s electron configuration ends in 4f^7 6s^2. This means it has seven electrons in the 4f subshell and two in the 6s subshell. The 4f electrons are more shielded and less reactive, while the 6s electrons are more easily lost in reactions. This arrangement gives europium two common oxidation states: +2 and +3.
Impact Of Valence Electrons On Reactivity
The presence of half-filled 4f orbitals makes europium relatively stable in its +2 state. This stability is unusual among lanthanides, which typically prefer the +3 state. Europium’s ability to lose two electrons leads to unique chemical properties and reactivity. It reacts more easily with oxygen and halogens compared to other lanthanides.
Common Oxidation States And Chemical Behavior
Europium commonly forms Eu²⁺ and Eu³⁺ ions. The Eu²⁺ ion results from losing the two 6s electrons, while Eu³⁺ forms by losing two 6s and one 4f electron. The +3 state is more stable in most compounds, but the +2 state appears in reducing environments. These states affect europium’s color, magnetism, and bonding patterns.
Valence Electrons And Reactivity
Europium holds a unique place in the periodic table. It is part of the lanthanide series. This series includes elements with atomic numbers 57 through 71. Europium’s atomic number is 63, placing it among these rare earth metals.
Located in period 6, europium shares similar properties with its lanthanide neighbors. These elements are known for their f-block electron configurations. This position affects europium’s chemical behavior and electron arrangement.
Europium is found in the f-block, specifically in the lanthanides group. It lies between samarium and gadolinium. This position reflects its electron filling in the 4f orbital.
Period And Group
Europium is in period 6 of the periodic table. Its group is often labeled as part of the lanthanides, which do not have a fixed group number. The lanthanides are placed separately below the main table.
Relation To Neighboring Elements
Europium’s neighbors are samarium (atomic number 62) and gadolinium (atomic number 64). These elements share similar properties due to their close atomic numbers. Their electron configurations show gradual filling of the 4f orbitals.

Credit: www.youtube.com
Common Oxidation States
The electron configuration of europium plays a key role in its chemical behavior. Europium is a lanthanide element with unique electron arrangements. These arrangements influence how it reacts with other elements and forms compounds.
Understanding europium’s electron configuration helps explain its typical oxidation states and bonding patterns. Its valence electrons are crucial in determining its reactivity and interaction with other substances.
Electron Configuration Of Europium
Europium has the atomic number 63. Its ground-state electron configuration is [Xe] 4f7 6s2. This means europium has seven electrons in the 4f subshell and two in the 6s subshell. The filled 4f orbitals give europium special magnetic and optical properties.
Impact On Oxidation States
Europium commonly shows +2 and +3 oxidation states. The +3 state happens when it loses two 6s electrons and one 4f electron. The +2 state results from losing only the two 6s electrons. This flexibility is rare in lanthanides, making europium chemically interesting.
Chemical Reactivity And Bonding
The 4f electrons in europium are shielded by outer electrons. This shielding causes europium to be less reactive than transition metals. It forms mostly ionic bonds in compounds, especially in +3 and +2 states. Europium’s electron configuration also affects its ability to form stable complexes.
Frequently Asked Questions
Which Atom Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d10?
The atom with electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d10 is copper (Cu). It has atomic number 29.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 4?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴ belongs to sulfur. It has 16 electrons.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn).
What Is The 2 8 8 18 18 Rule?
The 2 8 8 18 18 rule describes electron distribution in atomic shells. It assigns electrons as 2 in the first, 8 in second and third, then 18 in fourth and fifth shells. This helps explain electron configuration in elements beyond argon.
Conclusion
Understanding europium’s electron configuration helps explain its chemical behavior. Its unique arrangement affects how it bonds and reacts. This knowledge is useful in fields like chemistry and material science. Europium’s position in the periodic table relates directly to its electrons.
Knowing these details makes learning about lanthanides easier. It also supports studies on rare earth elements and their uses. Exploring europium’s electrons opens doors to deeper scientific insights. Simple, clear electron patterns tell a bigger story about this element.



No comments