Electron Configuration of Dysprosium
If you’ve ever wondered how the electrons arrange themselves inside a dysprosium atom, you’re in the right place. Understanding the electron configuration of dysprosium isn’t just about memorizing numbers—it’s about unlocking the secrets behind its unique properties and behavior.
Whether you’re a student, a science enthusiast, or just curious, knowing how dysprosium’s electrons are organized will deepen your grasp of this fascinating rare-earth metal. Keep reading, and you’ll discover exactly how these tiny particles shape the element’s identity and why that matters for science and technology.
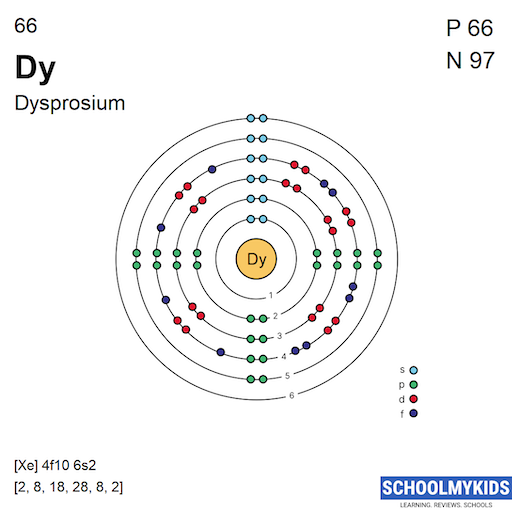
Credit: www.schoolmykids.com
Basic Properties Of Dysprosium
Dysprosium is a rare-earth metal found in the f-block of the periodic table. It has unique physical and chemical traits. These traits make it valuable in many high-tech applications.
Dysprosium’s atomic number is 66, meaning it has 66 protons and electrons in a neutral atom. It belongs to the lanthanide series, which includes elements with partially filled 4f orbitals.
Atomic Structure Of Dysprosium
The electron configuration of dysprosium is complex. Its electrons fill the 4f, 5d, and 6s orbitals. The full configuration is [Xe] 4f10 6s2. This arrangement defines its chemical behavior and bonding.
Physical Characteristics
Dysprosium is a silvery-white metal that is soft and malleable. It has a high melting point of about 1,412 °C. This makes it useful in environments with extreme heat.
Chemical Properties
Dysprosium reacts slowly with water but quickly forms oxides in air. It often shows a +3 oxidation state in compounds. Its chemistry is typical of lanthanides, showing magnetic and catalytic properties.
Atomic Number And Electron Count
Dysprosium’s atomic number is 66, which means it has 66 electrons in a neutral atom. These electrons fill energy levels following specific rules to create its unique electron configuration. This arrangement affects dysprosium’s chemical behavior and properties.
Atomic Number Of Dysprosium
Dysprosium has an atomic number of 66. This number shows how many protons are in its nucleus. The atomic number also tells the total electrons in a neutral atom. For dysprosium, that means it has 66 electrons orbiting the nucleus. This count defines the element's chemical identity. Each element in the periodic table has a unique atomic number.
Electron Count And Its Importance
The electron count affects how dysprosium reacts with other elements. Electrons move around the nucleus in layers called shells. Dysprosium’s 66 electrons fill these shells in a specific order. This order follows rules based on energy levels and stability. The number of electrons determines the element’s bonding and properties.
Electron Shells And Subshells
The electron shells and subshells explain how electrons arrange in an atom. Each shell holds a certain number of electrons. These shells surround the nucleus like layers of an onion. Inside each shell, electrons fill smaller parts called subshells.
Subshells have different shapes and energy levels. These differences affect how electrons behave and interact. Understanding shells and subshells helps reveal an element’s chemical properties.
Electron Shells Of Dysprosium
Dysprosium has 66 electrons. These electrons fill shells from the closest to the nucleus outward. The shells are numbered 1 to 6 for Dysprosium. Each shell can hold a fixed maximum number of electrons.
The first shell holds 2 electrons. The second and third shells hold 8 each. The fourth and fifth shells hold 18 each. The sixth shell holds the remaining electrons. This arrangement follows the 2n² rule.
Subshells In Dysprosium
Subshells are labeled as s, p, d, and f. Each subshell has a specific number of orbitals. Each orbital holds up to 2 electrons. Dysprosium's electrons fill these subshells in order of increasing energy.
The s subshell holds 2 electrons, p holds 6, d holds 10, and f holds 14. Dysprosium’s unique properties come from filling the 4f subshell. This subshell holds 10 electrons in Dysprosium.
How Electrons Fill Dysprosium’s Subshells
Electrons fill subshells starting from the lowest energy. The order follows the Aufbau principle. For Dysprosium, the filling ends with the 4f subshell. This subshell is partially filled, which makes Dysprosium special.
The full electron configuration shows how many electrons go in each subshell. Dysprosium’s configuration ends as [Xe] 4f¹⁰ 6s². This shows a complete picture of its electron arrangement.

Credit: pilgaardelements.com
Filling Order Of Orbitals
The filling order of orbitals describes how electrons arrange themselves around an atom's nucleus. Electrons fill orbitals starting from the lowest energy level to higher ones. This order helps explain the chemical and physical properties of elements like dysprosium.
Dysprosium has many electrons, so understanding its orbital filling is key. The order follows specific rules that balance energy and stability. These rules guide how electrons occupy the s, p, d, and f orbitals.
Understanding The Aufbau Principle
The Aufbau principle states electrons fill orbitals starting at the lowest energy level. Each new electron enters the next available orbital with the lowest energy. This creates a stable electron arrangement for the atom.
Order Of Orbital Filling In Dysprosium
Dysprosium's electrons fill orbitals in this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, and 5d. The 4f orbitals start filling after 6s because they have higher energy. Dysprosium has electrons in the 4f orbitals, which define its rare-earth characteristics.
Role Of The Pauli Exclusion Principle
The Pauli exclusion principle says each orbital holds up to two electrons. These electrons must have opposite spins. This rule limits how electrons fill the orbitals and affects the electron configuration of dysprosium.
Hund’s Rule And Electron Distribution
Hund’s rule states electrons fill orbitals singly before pairing up. This minimizes electron repulsion and maximizes stability. In dysprosium, electrons spread out in the 4f orbitals following this rule.
Ground State Configuration
Dysprosium’s ground state electron configuration shows how its electrons fill orbitals. It has electrons in the 4f and 6s shells. This pattern explains its chemical behavior and position in the periodic table.
The ground state configuration shows how electrons arrange themselves in an atom. Dysprosium is a rare-earth metal with atomic number 66. It has 66 electrons that fill its orbitals in a specific order to reach stability.
This configuration follows the Aufbau principle, where electrons fill lower energy levels first. Understanding this helps explain many of dysprosium’s chemical properties and its place in the periodic table.
Electron Arrangement In Dysprosium
Dysprosium’s electrons fill shells from the innermost to the outermost. The first electrons fill the 1s orbital, then move to higher orbitals such as 2s, 2p, and so on. This process continues until all 66 electrons occupy their places.
The unique feature of dysprosium is the filling of the 4f orbitals, which belong to the f-block elements. These orbitals hold electrons that influence the element’s magnetic and spectral properties.
Detailed Orbital Filling Order
The full electron configuration of dysprosium is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁰. This shows the electrons filling from the 1s orbital up to the 4f orbital.
The 4f orbitals start filling after the 6s orbital is occupied. Dysprosium has ten electrons in the 4f sublevel. This arrangement is key to its chemical behavior and magnetic strength.
Significance Of The Ground State Configuration
The ground state configuration determines how dysprosium bonds with other elements. The electrons in the outermost shells, especially 4f and 6s, are involved in chemical reactions. They also impact dysprosium’s magnetic and optical properties.
Scientists use this configuration to predict dysprosium’s reactions and its role in materials like magnets and lasers. Knowing the ground state helps in understanding its applications clearly.
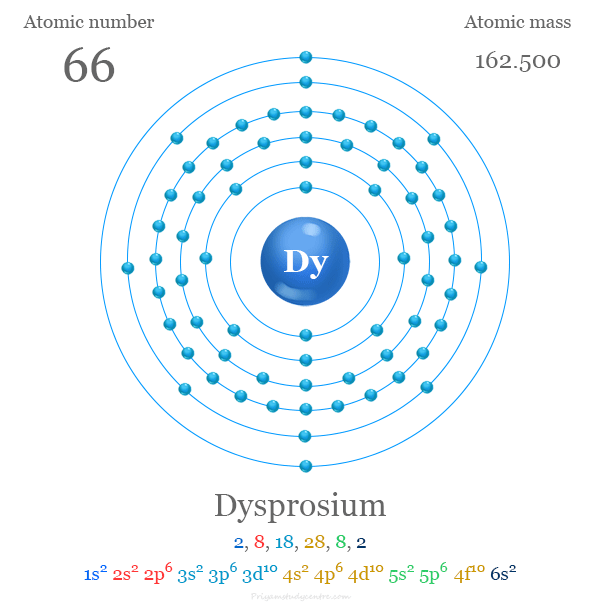
Credit: www.priyamstudycentre.com
Abbreviated Electron Configuration
The abbreviated electron configuration simplifies the full electron arrangement. It uses noble gas notation to shorten the sequence. This method helps focus on the electrons beyond core shells. It shows the valence electrons clearly, which are important in chemical behavior.
For dysprosium, abbreviated configuration starts with the nearest noble gas. This saves space and makes reading easier. It highlights the electrons added after the noble gas core. This is useful for students and scientists alike.
What Is Abbreviated Electron Configuration?
Abbreviated electron configuration uses the symbol of the last noble gas. It replaces all the electrons up to that noble gas. This leaves only the outer electrons visible. It helps understand an element’s reactivity and bonding.
Abbreviated Electron Configuration Of Dysprosium
Dysprosium’s atomic number is 66. The nearest noble gas before dysprosium is xenon (Xe), with 54 electrons. The abbreviated configuration starts as [Xe].
After xenon, dysprosium has electrons in the 4f and 6s orbitals. The full abbreviated configuration is [Xe] 4f10 6s2. This shows dysprosium’s unique electron arrangement in the f-block.
Why Use Abbreviated Configuration For Dysprosium?
This format is simpler and clearer. It avoids writing all 66 electrons individually. Scientists can quickly identify the valence electrons. These electrons influence dysprosium’s magnetic and chemical properties.
Abbreviated configuration also helps compare dysprosium with other lanthanides. It shows how electrons fill the 4f sublevel. This is key to understanding rare earth metals.
Role Of 4f And 6s Orbitals
Dysprosium’s electron configuration reveals the key roles of the 4f and 6s orbitals. These orbitals shape the element’s chemical and physical behavior. Understanding their function helps explain why dysprosium belongs to the lanthanide series.
The 4f orbitals are deeply buried and hold electrons that contribute to dysprosium’s magnetic properties. The 6s orbitals, on the other hand, are more exposed and influence its reactivity. Both orbitals work together to define dysprosium’s unique characteristics.
4f Orbitals And Their Electron Capacity
The 4f orbitals can hold up to 14 electrons in total. Dysprosium has 10 electrons occupying these 4f orbitals. These electrons are shielded by outer electrons, making their behavior less affected by the environment. This shielding leads to stable and complex magnetic properties.
Role Of 6s Orbitals In Chemical Reactions
The 6s orbitals contain two electrons in dysprosium’s ground state. These electrons are loosely held and can be lost or shared during chemical reactions. This makes the 6s orbitals crucial for bonding and the element’s typical +3 oxidation state.
Interaction Between 4f And 6s Orbitals
The 4f and 6s orbitals influence each other despite their different locations. The 6s electrons help stabilize the 4f electron cloud. This interaction affects dysprosium’s atomic size and ionization energies. Together, they determine how dysprosium behaves in compounds.
Electron Configuration And Chemical Behavior
Dysprosium is a rare-earth metal with unique electron arrangement. This arrangement shapes its chemical properties and reactions. Understanding its electron configuration reveals why it behaves a certain way in compounds and materials.
The way electrons fill its orbitals impacts its magnetism and bonding ability. Dysprosium’s electrons fill the 4f and 6s orbitals, influencing its stability and reactivity. This pattern helps explain its role in technology and industry.
Basic Electron Configuration Of Dysprosium
Dysprosium has an atomic number of 66. Its full electron configuration is [Xe] 4f10 6s2. This means it has ten electrons in the 4f sublevel and two in the 6s sublevel. The inner shells are like xenon’s, a noble gas, which provides a stable core.
Impact On Chemical Reactivity
The 4f electrons are shielded by outer electrons, so they react less easily. The two 6s electrons are more available for chemical bonding. Dysprosium usually forms +3 oxidation states by losing these outer electrons. This behavior defines its participation in chemical reactions.
Magnetic And Physical Properties Linked To Electron Setup
The large number of unpaired 4f electrons gives dysprosium strong magnetic properties. It is used in making magnets that work at high temperatures. These properties come directly from its electron configuration and affect its industrial use.
Comparison With Neighboring Elements
Dysprosium’s electron configuration closely resembles its neighbors terbium and holmium. Differences appear mainly in the filling of 4f orbitals. This pattern reflects gradual changes across the lanthanide series.
Electron Configuration Of Dysprosium
Dysprosium is a rare-earth element found in the f-block of the periodic table. Its electron configuration shows how electrons fill its atomic orbitals. This configuration affects its chemical and physical properties.
Dysprosium’s configuration is unique but shares similarities with nearby elements. Examining these similarities helps us understand trends in the lanthanide series.
Comparison With Terbium
Terbium comes just before dysprosium in the periodic table. Both elements have electrons filling the 4f orbitals. Terbium's electron configuration ends in 4f9 6s2, while dysprosium has 4f10 6s2.
The extra electron in dysprosium slightly changes its magnetic and optical properties. This difference influences their behavior in alloys and magnets.
Comparison With Holmium
Holmium follows dysprosium and has one more electron in the 4f orbital. Its configuration is 4f11 6s2. This gradual increase affects atomic size and ionization energy.
Holmium’s properties reflect the addition of this electron, showing changes in reactivity and magnetic strength compared to dysprosium.
Trends Across The Lanthanide Series
The lanthanides fill the 4f orbitals in a stepwise manner. Dysprosium fits into this pattern with ten electrons in 4f orbitals. This filling causes a steady change in atomic radius and chemical behavior.
Each neighboring element’s electron configuration helps explain their place in the series. Dysprosium’s configuration marks a midpoint with distinct magnetic and chemical traits.
Applications Linked To Electron Structure
The electron structure of dysprosium defines many of its practical uses. Electrons in dysprosium’s outer shells shape its magnetic and chemical traits. These traits make dysprosium valuable in specific industries and technologies.
Understanding dysprosium’s electron arrangement helps explain its role in magnets, lasers, and nuclear reactors. Each electron shell and subshell affects how dysprosium interacts with other elements and fields.
Magnetic Applications
Dysprosium has unpaired electrons in its 4f subshell. These electrons create strong magnetic moments. This property makes dysprosium useful in high-performance magnets. Dysprosium improves magnet stability at high temperatures. It is often added to neodymium magnets to boost their strength and heat resistance.
Laser Technology
The electron configuration allows dysprosium ions to emit specific light wavelengths. This feature is vital for solid-state lasers. Dysprosium-doped lasers are used in medicine and communication. The electron transitions in dysprosium ions produce clear and precise laser beams.
Nuclear Reactor Use
Dysprosium’s electron structure enables it to absorb neutrons efficiently. It acts as a control material in nuclear reactors. Dysprosium helps manage the reactor’s chain reactions safely. Its electron setup supports these neutron capture properties.
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² represents calcium (Ca). It has 20 electrons filling up to the 4s orbital.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ belongs to the element Chromium (Cr).
Which Atom Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 belongs to the chromium (Cr) atom. Chromium has atomic number 24 with a unique, stable half-filled 3d subshell.
What Is The 2 8 8 18 18 Rule?
The 2 8 8 18 18 rule describes electron distribution in atomic shells. It shows electrons filling shells with these maximum numbers: 2, 8, 8, 18, and 18. This helps explain element electron configurations and chemical behavior.
Conclusion
Understanding dysprosium’s electron configuration helps explain its chemical traits. Its electrons fill shells following specific rules and orders. This pattern affects dysprosium’s behavior in reactions and materials. Knowing this setup aids learning about rare-earth elements. Dysprosium’s place in the f-block shows its unique properties.
Simple electron patterns reveal complex atomic features. This knowledge supports studies in chemistry and physics fields. Exploring electron configurations makes elements easier to understand and use.



No comments