Electron Configuration of Californium
Are you curious about what makes Californium so unique at the atomic level? Understanding its electron configuration reveals the secrets behind its chemical behavior and uses.
By exploring how electrons are arranged in Californium, you’ll gain a clearer picture of this rare element’s properties and why it matters in science and industry. Keep reading to uncover the fascinating details that connect tiny particles to big discoveries.

Credit: www.webelements.com
Basic Properties Of Californium
Californium is a rare and radioactive element. It belongs to the actinide series. Its chemical symbol is Cf, and its atomic number is 98. This metal is synthetic, meaning it does not occur naturally. Scientists produce it in laboratories through nuclear reactions.
Californium has unique physical and chemical properties. It is silvery and shiny but quickly tarnishes in air. The metal is soft and malleable, making it easy to shape. Californium’s most notable property is its strong radioactivity, which limits its everyday use.
Atomic Structure And Electron Configuration
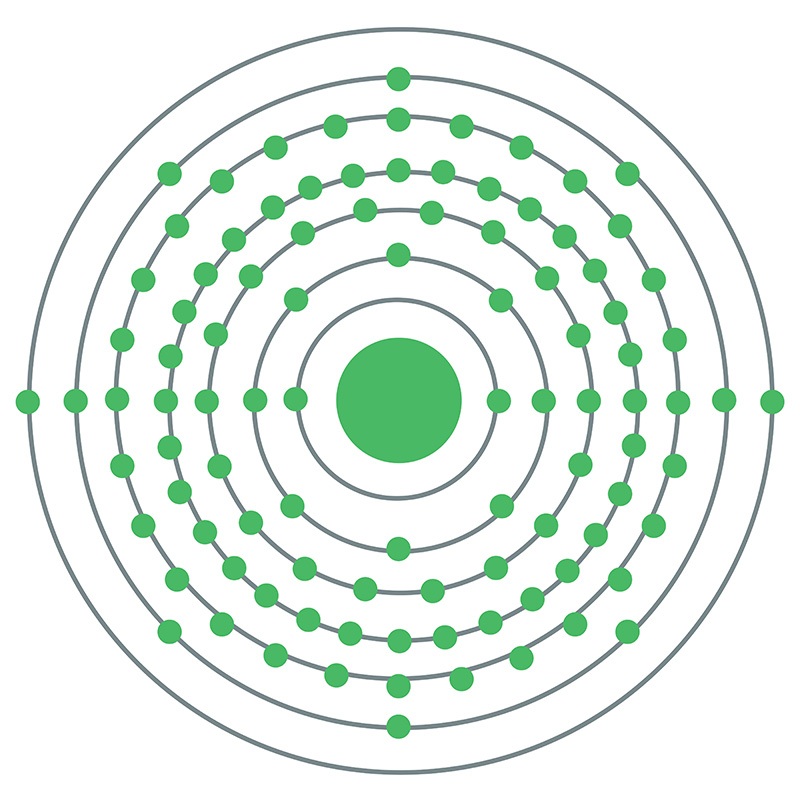
Californium has 98 electrons orbiting its nucleus. Its electron configuration is [Rn] 5f10 7s2. This means it has 10 electrons in the 5f orbital and 2 in the 7s orbital. The f-orbitals play a key role in its chemical behavior.
Physical Characteristics
The element appears as a soft, silvery metal. It melts at about 900 degrees Celsius. Californium is dense, weighing nearly 15 grams per cubic centimeter. It reacts slowly with oxygen and water, forming a thin oxide layer.
Chemical Behavior
Californium shows multiple oxidation states, commonly +3 and +4. It forms compounds with halogens and oxygen. Its chemistry resembles other actinides like curium and berkelium. The element’s radioactivity influences its chemical reactions.
Atomic Structure Overview
Californium is a rare, radioactive element found in the actinide series. Its atomic structure shows how electrons are arranged around the nucleus. This arrangement affects the element’s chemical and physical properties.
Understanding californium’s electron configuration helps explain its behavior in reactions and its place in the periodic table. The atomic number of californium is 98, meaning it has 98 protons and usually 98 electrons.
The electrons in californium fill several energy levels or shells. Each shell contains smaller parts called subshells: s, p, d, and f. These subshells hold electrons in specific patterns.
Californium’s electrons mainly fill the 5f subshell. This gives it unique characteristics compared to other elements. The 5f electrons are crucial in its chemical activity.
Ground State Electron Configuration
The ground state shows the lowest energy arrangement of electrons. For californium, this is written as [Rn] 5f10 7s2. It means californium has ten electrons in the 5f subshell and two in the 7s subshell.
This configuration reflects the balance of energy and stability in the atom. The electrons fill the orbitals to minimize energy.
Role Of Valence Electrons
Valence electrons are the outermost electrons involved in bonding. Californium has four valence electrons: two in 7s and two in 5f. These electrons decide how californium bonds with other elements.
The 5f electrons make californium’s chemistry complex and interesting. Their position influences the element’s magnetic and electrical properties.
Electron Shells And Subshells
Electron shells and subshells describe how electrons are arranged around an atom's nucleus. Each shell holds a certain number of electrons in specific energy levels. These shells are divided into smaller parts called subshells, which determine the shape and energy of electron clouds.
Understanding the electron shells and subshells of californium helps explain its chemical behavior. Californium is a heavy element with many electrons filling various shells and subshells.
Electron Shells In Californium
Californium has electrons arranged in seven main shells. These shells are numbered 1 through 7. Each shell contains electrons with increasing energy levels as the number grows. The outer shells have electrons that influence the atom’s reactivity.
Subshells In Californium
Each shell splits into subshells labeled s, p, d, and f. Californium’s electrons fill all these subshells up to the 5f subshell. The 5f subshell is important for its unique properties. It contains electrons that affect californium’s magnetism and radioactivity.
Electron Distribution Pattern
Californium’s electrons fill shells and subshells in a specific order. The filling follows the Aufbau principle, starting from low to high energy. This pattern creates a stable electron arrangement. The last electrons enter the 5f subshell, characterizing californium as an actinide.

Credit: www.youtube.com
Californium Electron Arrangement
Californium is a rare and radioactive element. Its electron arrangement reveals much about its chemical properties. Understanding this arrangement helps explain how californium behaves in different reactions.
The electrons in californium fill specific energy levels and orbitals. These patterns follow the rules of quantum mechanics. Each electron occupies the lowest energy state available.
Basic Electron Configuration Of Californium
Californium has 98 electrons. These electrons fill shells in a particular order. The general configuration is based on the Aufbau principle.
The main configuration is: [Rn] 5f10 7s2. This means it follows the radon core and adds electrons in the 5f and 7s orbitals.
Role Of 5f Electrons In Californium
The 5f electrons define many properties of californium. They are located in the f-orbital, which can hold up to 14 electrons. Californium has 10 electrons in the 5f orbital.
These electrons influence magnetism and bonding. They are less shielded and more reactive than electrons in outer shells.
Energy Levels And Electron Distribution
Electrons fill the 7s orbital after the 5f. The 7s orbital holds 2 electrons in californium. This arrangement stabilizes the atom.
Other orbitals such as 6d are not occupied in the ground state. This specific distribution impacts californium’s chemical activity.
Role Of 5f Orbitals
Californium’s electron configuration involves filling 5f orbitals. These orbitals hold electrons that influence its chemical properties. The 5f orbitals help explain how californium behaves in reactions and bonding.
Understanding The 5f Orbitals In Californium
The 5f orbitals play a key role in defining californium’s chemical properties. These orbitals hold electrons that influence how californium atoms interact. They are part of the actinide series and differ from other orbitals by their shape and energy levels.
Electrons in the 5f orbitals are less shielded from the nucleus. This causes stronger attraction between electrons and the nucleus. As a result, californium shows unique magnetic and chemical behavior.
Electron Distribution In 5f Orbitals
Californium has electrons filling its 5f orbitals before moving to higher energy levels. This filling pattern affects its stability and reactivity. The exact number of electrons in these orbitals changes with the element’s ionization state.
The 5f orbitals can hold up to 14 electrons. Californium usually has around 10 electrons in these orbitals. This partial filling causes complex bonding and different oxidation states.
Impact On Californium’s Chemical Bonds
Electrons in the 5f orbitals help form bonds with other atoms. These bonds are often stronger due to the orbital’s shape and electron distribution. Californium tends to form covalent bonds using 5f electrons.
The involvement of 5f orbitals leads to multiple oxidation states. This variety gives californium flexibility in chemical reactions. It can combine with many elements, showing diverse chemistry.
Credit: www.americanelements.com
Comparison With Other Actinides
The electron configuration of californium offers unique insights among the actinide series. Comparing it with other actinides helps understand its chemical and physical properties. Each actinide element fills its electrons in a specific order within 5f, 6d, and 7s orbitals.
These patterns affect the element's reactivity, magnetism, and stability. Californium’s electron arrangement shows subtle differences that influence its behavior. This section compares californium’s electron configuration with selected actinides to highlight these variations.
Electron Configuration Trends In Actinides
Actinides generally fill electrons in the 5f orbital first. The 7s orbital fills before 5f and 6d. Early actinides like thorium and uranium show more 6d electron involvement. Later actinides, including californium, primarily fill the 5f orbital.
This trend affects their chemical bonding and oxidation states. Californium's configuration reflects a near-complete 5f subshell. This influences its position and properties within the series.
Californium Vs. Uranium Electron Configuration
Uranium has the electron configuration [Rn] 5f3 6d1 7s2. It has more electrons in 6d orbitals than californium. Californium’s configuration is [Rn] 5f10 7s2, with no electrons in 6d orbitals.
This difference causes uranium to be more chemically reactive. Californium shows greater stability in its electron arrangement. The filled 5f orbitals in californium reduce its tendency to lose electrons.
Impact Of Electron Configuration On Californium’s Properties
Californium’s electron setup leads to strong magnetic and radioactive properties. It has a higher number of unpaired 5f electrons. These unpaired electrons contribute to its magnetic behavior.
Other actinides with fewer 5f electrons show weaker magnetic effects. Californium also has unique oxidation states due to its electron arrangement. This sets it apart in chemical reactions and industrial use.
Impact On Chemical Behavior
The electron configuration of californium shapes how it acts in chemical reactions.
This element's unique arrangement of electrons affects its bonding and stability.
Understanding this helps explain californium’s rare and complex chemistry.
Electron Arrangement And Reactivity
Californium has electrons in the 5f, 6d, and 7s orbitals.
These electrons influence how easily it loses or gains electrons.
This controls californium’s ability to form compounds with other elements.
Oxidation States And Stability
Californium commonly shows +3 and +4 oxidation states.
These states depend on which electrons are lost during reactions.
The +3 state is more stable and often found in compounds.
Covalent Bonding Characteristics
The 5f electrons can participate in bonding, unlike many metals.
This leads to covalent bonds with some nonmetals and ligands.
Such bonding influences californium’s chemical properties and uses.
Electron Configuration And Radioactivity
Californium is a rare and radioactive element found in the actinide series. Its electron configuration plays a key role in its radioactivity and chemical properties. Understanding this configuration helps explain why californium behaves the way it does.
The way electrons arrange themselves around the nucleus affects the atom’s stability. Californium’s electrons fill specific orbitals, influencing its radioactive nature. This section breaks down the electron configuration and links it to the element’s radioactivity.
Electron Configuration Of Californium
Californium’s atomic number is 98, meaning it has 98 electrons. These electrons fill the orbitals in order of increasing energy. The full electron configuration is: [Rn] 5f10 7s2. The 5f orbitals hold most of the valence electrons. These orbitals are less stable and contribute to the element’s unique properties.
Connection Between Electron Configuration And Radioactivity
Electrons in the 5f orbitals cause instability in the nucleus. This instability leads to radioactive decay. Californium emits alpha particles as it seeks a more stable state. The incomplete filling of the 5f shell makes the nucleus prone to breaking down.
The electron arrangement affects californium’s chemical reactions. Its valence electrons in the 5f and 7s orbitals allow it to form bonds with other elements. Californium often shows a +3 oxidation state in compounds. This state relates directly to its electron configuration and radioactivity.
Techniques To Determine Configuration
Understanding the electron configuration of californium requires precise techniques. Scientists use different methods to find out how electrons arrange themselves in this heavy element. These techniques help reveal the structure and properties of californium atoms. They also provide key insights into its chemical behavior.
Each technique has its strengths and limits. Some methods focus on measuring energy levels. Others observe the effects of magnetic fields on electrons. Together, these approaches build a clearer picture of californium’s electron layout.
Spectroscopic Analysis
Spectroscopy studies light absorbed or emitted by atoms. When californium atoms change energy states, they emit specific light wavelengths. These wavelengths show the energy differences between electron levels. By analyzing these lines, scientists determine electron arrangements accurately.
Quantum Mechanical Calculations
Computers use quantum physics equations to predict electron positions. These calculations model californium’s complex electron interactions. They help estimate the most stable electron configuration. This method complements experimental data by filling knowledge gaps.
X-ray Absorption Spectroscopy
This technique measures how X-rays interact with californium atoms. It detects changes in electron density near the nucleus. X-ray absorption reveals details about electron shells and subshells. This information refines the understanding of electron configuration.
Magnetic Susceptibility Measurements
Measuring magnetic susceptibility shows how electrons respond to magnetic fields. Californium’s unpaired electrons affect its magnetic properties. Tracking these changes reveals electron spin and arrangement. This technique provides clues about electron distribution.
Applications Influenced By Electron Structure
The electron configuration of californium shapes how it interacts with other elements and compounds. This arrangement of electrons controls its chemical behavior and physical properties. Understanding this helps us explore californium’s practical uses in science and industry.
Californium’s unique electron structure makes it valuable in several fields. It influences the element’s radioactivity and ability to emit neutrons. These traits lead to specialized applications that depend on californium’s atomic design.
Neutron Source In Nuclear Reactors
Californium’s electron arrangement enables it to emit neutrons efficiently. This makes it useful as a neutron source. It helps start and control nuclear chain reactions in reactors. Its neutron emission supports the production of other isotopes used in medicine and research.
Radiation Therapy For Cancer
Californium’s radioactive properties arise from its electron structure. It emits particles that can kill cancer cells. Doctors use californium in targeted radiation therapy. This helps treat tumors that are hard to reach with other methods.
Material Analysis And Testing
Californium’s neutrons interact with materials uniquely. This interaction reveals structural details about metals and alloys. Scientists use californium-based neutron sources to test material strength and composition. It helps ensure quality and safety in manufacturing.
Scientific Research In Nuclear Chemistry
The electron configuration of californium allows it to form various compounds. Researchers study these compounds to learn about heavy elements. Californium helps expand knowledge of atomic behavior and nuclear reactions. This research supports advances in chemistry and physics.
Frequently Asked Questions
What Is The Electron Configuration Of Californium?
Californium has the electron configuration [Rn] 5f^10 7s^2. It belongs to the actinide series. This means it has 10 electrons in the 5f subshell and 2 in the 7s subshell.
Why Is Californium's Electron Configuration Important?
Understanding Californium’s electron configuration helps explain its chemical behavior. It reveals how the element bonds and interacts. This knowledge is key in nuclear science and material research.
How Does Californium's Electron Configuration Affect Its Properties?
The 5f electrons in Californium influence its magnetic and radioactive properties. These electrons cause unique chemical reactions. This makes Californium useful in specialized scientific applications.
Where Is Californium Placed In The Periodic Table?
Californium is in the actinide series, atomic number 98. It is located in period 7. Its position reflects its electron configuration and chemical characteristics.
Conclusion
Californium’s electron configuration reveals its place in the actinide series. This pattern helps us understand its chemical behavior and properties. Each electron occupies a specific energy level, shaping how the element reacts. Knowing this arrangement supports studies in chemistry and physics.
The topic might seem complex, but breaking it down makes it clearer. Exploring californium’s electrons opens doors to learning about heavy elements. Science often starts with small details like these. Keep exploring to discover more about the elements around us.



No comments