Electron Configuration of Bismuth
Have you ever wondered how the tiny particles inside atoms shape the world around you? Understanding the electron configuration of elements like bismuth can unlock secrets about their behavior and uses.
If you want to grasp how bismuth’s electrons are arranged and why that matters, you’re in the right place. By the end of this article, you’ll see bismuth in a whole new light—and you’ll be able to explain its unique properties with confidence.
Let’s dive into the fascinating world of bismuth’s electron configuration and discover what makes this element so special.

Credit: www.researchgate.net
Basic Properties Of Bismuth
Bismuth is a heavy metal with unique features. It belongs to the group of post-transition metals. This element stands out for its low toxicity compared to others in its group.
Bismuth has a silvery-white color with a slight pink tint. Its density is high, making it one of the heaviest stable elements. Bismuth also has a low melting point for a metal.
Atomic Number And Symbol
Bismuth’s atomic number is 83. Its chemical symbol is Bi. This number tells us how many protons are in the atom’s nucleus.
Physical State And Appearance
At room temperature, bismuth is solid. It has a shiny, metallic look with a pinkish hue. The metal is brittle and breaks easily.
Electron Configuration
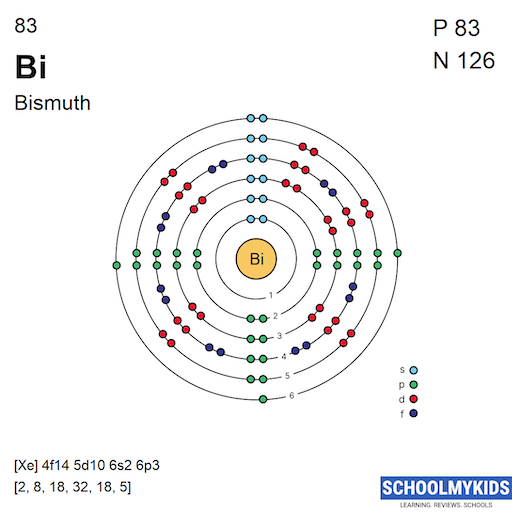
The electron configuration of bismuth is key to its properties. It is written as [Xe] 4f14 5d10 6s2 6p3. This shows the arrangement of electrons in its shells.
Melting And Boiling Points
Bismuth melts at 271.5°C and boils at 1,564°C. Its melting point is low compared to other metals. This makes it useful in low-temperature applications.
Density And Atomic Mass
The density of bismuth is about 9.78 g/cm³. Its atomic mass is 208.98 u. Both values are high, which reflects its heavy metal nature.
Atomic Structure Of Bismuth
Bismuth is a heavy metal with unique atomic properties. Its atomic structure reveals how its electrons are arranged. This arrangement affects many of its chemical and physical traits.
Understanding the atomic structure of bismuth helps explain its behavior in reactions and compounds. The electron arrangement also influences its place in the periodic table.
Electron Shells And Energy Levels
Bismuth has five electron shells. These shells hold electrons at different distances from the nucleus. Each shell corresponds to a specific energy level. The outermost shell contains electrons that interact during chemical reactions.
Number Of Protons, Neutrons, And Electrons
Bismuth has 83 protons in its nucleus. It also contains about 126 neutrons. The number of electrons equals the number of protons, so 83 electrons orbit the nucleus. This balance keeps the atom electrically neutral.
The electron configuration shows the exact order of electron filling. For bismuth, it is written as 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p³. This notation helps predict how bismuth bonds with other elements.
Electron Shells And Subshells
Electron shells and subshells describe how electrons arrange around an atom's nucleus. They help explain the chemical behavior of elements. Each shell holds a certain number of electrons. Inside each shell, electrons fill smaller groups called subshells.
Bismuth is a heavy element with many electrons. Its electron arrangement shows how shells and subshells fill up step by step. This setup affects its properties and reactions.
Understanding Electron Shells In Bismuth
Bismuth has five main electron shells. These shells are numbered from 1 to 6, but some shells are not fully filled. The first shell holds up to 2 electrons. The next shells hold more, following specific rules. Bismuth’s electrons fill shells up to the sixth one.
Role Of Subshells In Electron Configuration
Each shell contains subshells named s, p, d, and f. These subshells hold different numbers of electrons. The s subshell holds 2 electrons, p holds 6, d holds 10, and f holds 14. Bismuth’s electron configuration includes all these subshells.
How Electrons Fill Bismuth’s Subshells
Electrons fill subshells from lower to higher energy. Bismuth’s electrons fill the 1s subshell first. Then they fill 2s, 2p, 3s, 3p, and so on. The pattern follows the Aufbau principle, which guides electron filling order. Bismuth’s last electrons enter the 6p subshell.
Credit: www.schoolmykids.com
Bismuth’s Electron Configuration
Bismuth is a heavy metal with interesting properties. Its electron configuration reveals how its electrons are arranged in shells and subshells. This arrangement explains many of its chemical behaviors.
Understanding bismuth’s electron configuration helps us see why it reacts the way it does. It also shows its place in the periodic table among other elements.
Bismuth’s Atomic Number And Electrons
Bismuth has the atomic number 83. This means it has 83 electrons. These electrons fill the energy levels around the nucleus in a specific order.
Electron Shells And Subshells Of Bismuth
The electrons in bismuth are arranged in shells. Each shell contains subshells named s, p, d, and f. These subshells hold electrons in a certain sequence.
Full Electron Configuration Of Bismuth
The full electron configuration of bismuth is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 4f¹⁴ 5s² 5p⁶ 5d¹⁰ 6s² 6p³. This shows how electrons fill the orbitals step by step.
Valence Electrons And Chemical Properties
Bismuth’s valence electrons are in the 6s and 6p subshells. These five electrons mainly decide bismuth’s chemical reactions. Valence electrons are key for bonding with other atoms.
Orbital Filling Order
Bismuth’s electrons fill orbitals in a specific order based on energy levels. This sequence helps explain its unique electron configuration. Orbital filling follows simple rules to place electrons correctly.
Understanding Orbital Filling Order
The orbital filling order shows how electrons arrange in an atom. Electrons fill orbitals from lower to higher energy levels. This pattern follows specific rules to keep the atom stable.
Each orbital can hold a set number of electrons. Electrons fill orbitals in the order of increasing energy. This order helps predict the chemical behavior of elements like bismuth.
Electron Configuration Of Bismuth
Bismuth has 83 electrons. These electrons fill orbitals following the Aufbau principle. It means they occupy the lowest energy orbitals first before moving to higher ones.
The electron configuration of bismuth is written by listing filled orbitals in order. This shows which orbitals contain electrons and how many.
Orbital Filling For Bismuth: Step By Step
First, electrons fill the 1s orbital. Next, they fill 2s and then 2p orbitals. The process continues through 3s, 3p, and 4s orbitals.
After these, electrons fill 3d and 4p orbitals. Then come 5s and 4d orbitals, followed by 5p. The 6s orbital fills next, then 4f and 5d orbitals.
Finally, electrons fill the 6p orbitals, where bismuth’s outer electrons reside. This filling order explains bismuth’s placement in the periodic table.

Credit: www.youtube.com
Electron Configuration Notation
Bismuth’s electron configuration shows how its electrons fill energy levels. It follows the order of orbitals with 83 electrons arranged step by step. This notation helps understand bismuth’s chemical behavior.
What Is Electron Configuration Notation?
Electron configuration notation shows how electrons are arranged in an atom. It uses numbers, letters, and superscripts. These symbols represent energy levels, sublevels, and electron count.
This notation helps us understand an element’s chemical behavior. It explains how atoms bond and react with others.
Electron Configuration Notation Of Bismuth
Bismuth has 83 electrons. Its electron configuration notation shows how these electrons fill orbitals. The notation for bismuth is:
1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p⁶ 6s² 4f¹⁴ 5d¹⁰ 6p³
This sequence follows the order of increasing energy levels. It fills lower-energy orbitals first.
How To Read Electron Configuration Notation
Each part of the notation has meaning. The number shows the energy level or shell. The letter shows the type of orbital: s, p, d, or f. The superscript shows how many electrons are in that orbital.
For example, "6p³" means three electrons in the p orbital of the sixth shell.
Why Electron Configuration Notation Matters
This notation reveals an element’s chemical properties. It predicts how atoms will bond and interact. Understanding bismuth’s electron configuration explains its place in the periodic table.
It also helps in studying its physical and chemical traits.
Role Of Electrons In Chemical Behavior
Electrons determine how an atom reacts with others. They are the main players in chemical bonds. The way electrons arrange themselves affects an element’s properties. Bismuth is no different.
Understanding the electrons in bismuth helps explain its behavior in reactions. These electrons control how bismuth forms compounds. They also influence its stability and reactivity.
Electron Shells And Their Influence
Bismuth has electrons arranged in different shells. Each shell holds a certain number of electrons. The outermost shell, called the valence shell, is most important. These valence electrons take part in chemical bonds.
Because bismuth’s valence shell has few electrons, it tends to lose them. This loss forms positive ions. These ions help bismuth connect with other atoms.
Valence Electrons And Reactivity
Bismuth has five valence electrons. These electrons decide how easily it reacts. More valence electrons usually mean stronger bonds. But bismuth’s electrons are held loosely, so it reacts moderately.
This moderate reactivity makes bismuth useful in many applications. It forms stable compounds without reacting too fast or too slow.
Electron Configuration And Bond Formation
Bismuth’s electron configuration is unique. It ends with 6p3 electrons in the outer shell. These electrons help create covalent and ionic bonds. The way they share or transfer electrons defines the bond type.
This configuration also affects the shape and strength of molecules. It helps chemists predict how bismuth will behave in different reactions.
Comparison With Neighboring Elements
Bismuth sits in the periodic table near several interesting elements. Comparing its electron configuration with neighbors helps us understand its unique traits. These comparisons reveal patterns and differences in chemical behavior.
Neighboring elements include lead (Pb) and polonium (Po). Each has a distinct arrangement of electrons in their shells. This arrangement affects how they react and bond with other elements.
Electron Configuration Of Lead (pb)
Lead has the atomic number 82. Its electron configuration ends in 6p2. This means lead has two electrons in the outermost p orbital. Compared to bismuth, lead has fewer electrons in the outer shell. This difference causes lead to have different chemical properties.
Electron Configuration Of Polonium (po)
Polonium has the atomic number 84. Its electron configuration ends in 6p4. Polonium has four electrons in its outer p orbital. Bismuth, with 6p3, has one less outer p electron. This small change affects how each element bonds and reacts.
How Bismuth Differs From Its Neighbors
Bismuth has the electron configuration 6p3. It has three electrons in the outer p orbital. This gives bismuth a balance between lead and polonium. Bismuth is less reactive than polonium but more than lead. Its unique electron setup explains this behavior.
Impact On Physical Properties
Bismuth’s electron configuration affects its density and melting point. This arrangement also influences its electrical conductivity and hardness. These physical traits come from how electrons fill the atom’s outer shells.
Electron Configuration And Density
Bismuth's electron arrangement affects its density. Its electrons fill inner shells tightly. This creates strong attraction between atoms. As a result, bismuth is heavier and denser than many metals.
Influence On Electrical Conductivity
The electron setup controls how electrons flow. Bismuth has fewer free electrons for conduction. This limits its electrical conductivity compared to metals like copper. It acts more like a semiconductor in some cases.
Effect On Thermal Properties
Bismuth's electrons impact heat transfer. The structure slows down heat movement. This makes bismuth a poor conductor of heat. It is useful in applications needing heat insulation.
Relation To Magnetic Behavior
The electron configuration defines bismuth's magnetic traits. Its electrons pair up, canceling magnetic effects. Bismuth shows weak diamagnetism. It repels magnetic fields slightly but is not magnetic itself.
Applications Linked To Electron Structure
The electron configuration of bismuth shapes many of its uses. This element’s electrons fill energy levels in a specific pattern. These patterns influence bismuth’s chemical behavior and physical properties. Understanding these details helps explain why bismuth works well in certain applications.
Bismuth’s electron arrangement leads to unique traits. It is a heavy metal but less toxic than others. Its electron structure also supports strong diamagnetism. These features make bismuth valuable in science and industry.
Electron Configuration and Medical UsesBismuth’s electron setup allows it to form stable compounds. Many of these compounds have antibacterial properties. Doctors use bismuth compounds to treat stomach ulcers and infections. The electron structure helps bismuth interact safely with human cells. This makes it useful in medicine.
Role in Electronics and SemiconductorsThe electron configuration of bismuth affects its electrical conductivity. Bismuth can act as a semiconductor material. It is used in devices that need precise temperature control. Its electron structure helps maintain stability in electronic circuits.
Use in Alloys and Safety DevicesBismuth’s electrons give it a low melting point. This property is important in making alloys. These alloys are used in fire detectors and safety devices. The electron arrangement allows bismuth to melt quickly and safely in emergencies.
Frequently Asked Questions
What Is The Electron Configuration Of Bismuth?
Bismuth's electron configuration is [Xe] 4f14 5d10 6s2 6p3. It shows the distribution of electrons in shells and subshells. This configuration reveals Bismuth’s position in the periodic table and its chemical properties.
Why Is Bismuth’s Electron Configuration Important?
It helps predict Bismuth’s chemical reactivity and bonding behavior. Understanding its configuration aids in studying its role in alloys and pharmaceuticals. It also explains Bismuth’s oxidation states and magnetic properties.
How Does Bismuth's Electron Configuration Affect Its Properties?
The 6p3 electrons influence Bismuth's metallic and chemical traits. Its unique configuration leads to low toxicity and high stability. This makes Bismuth useful in cosmetics, medicines, and industrial applications.
What Is The Valence Electron Configuration Of Bismuth?
Bismuth’s valence electron configuration is 6s2 6p3. These electrons participate in chemical bonding and reactions. Valence electrons determine Bismuth’s ability to form compounds and its oxidation states.
Conclusion
Bismuth’s electron configuration reveals its unique chemical nature. It helps explain how bismuth bonds with other elements. Understanding this pattern makes studying its properties easier. This knowledge supports learning about heavier elements too. Bismuth plays an important role in many scientific fields.
Knowing its electrons’ arrangement adds to basic chemistry skills. Keep exploring elements to see how electron configurations vary. This simple structure holds key information about element behavior.



No comments