Atomic Structure in Physics
Have you ever wondered what makes up everything around you—from the air you breathe to the device in your hand? The answer lies in the tiny building blocks called atoms.
Understanding the atomic structure in physics is key to unlocking how matter behaves and interacts. You’ll discover how atoms are arranged, what particles form them, and why these details matter to everything in your daily life. By the end, you’ll see the invisible world inside atoms in a whole new light—and why grasping this concept is essential if you want to truly understand the physical world.
Ready to dive into the fascinating world of atomic structure? Let’s get started!
Atomic Particles
The atom is the smallest unit of matter. It consists of tiny particles called atomic particles. These particles include protons, neutrons, and electrons. Each plays a unique role in forming the atom’s structure and properties. Understanding these particles helps explain how atoms behave and interact.
Protons And Neutrons
Protons carry a positive electrical charge. They reside in the center of the atom, known as the nucleus. Neutrons are neutral; they have no charge. Neutrons also sit in the nucleus alongside protons. Together, they make up almost all the atom’s mass. The number of protons defines the element.
Electron Properties
Electrons are tiny particles with a negative charge. They orbit the nucleus in layers called shells. Electrons are much lighter than protons or neutrons. Their movement creates an electron cloud around the nucleus. Electrons determine how atoms bond and react with others.
Atomic Number And Mass Number
The atomic number equals the number of protons in an atom. It identifies the chemical element. The mass number is the total count of protons and neutrons. This number shows the atom’s overall mass. Different atoms can share the same mass number but have different atomic numbers.
Ions And Isotopes
Ions form when atoms gain or lose electrons. This gives them a positive or negative charge. Isotopes are atoms of the same element with different neutron counts. Isotopes have the same atomic number but different mass numbers. Both ions and isotopes affect the atom’s behavior and stability.
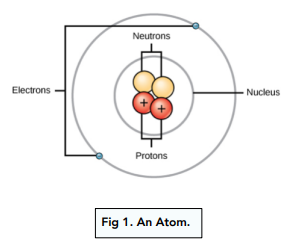
Credit: studymind.co.uk
Electron Arrangement
Electrons are tiny particles that orbit the center of an atom. Their arrangement around the nucleus shapes how atoms behave. Understanding electron placement helps explain many physical and chemical properties of matter. Electrons do not float randomly; they follow specific paths called shells or energy levels. These shells hold electrons in a structured way.
Each shell has a certain energy and capacity. Electrons fill the lower energy shells first before moving to higher ones. This arrangement is key to how atoms interact and form bonds with other atoms.
Energy Levels And Shells
Electrons orbit the nucleus in layers called energy levels or shells. Each shell is a fixed distance from the nucleus and holds electrons with similar energy. The first shell is closest to the nucleus and has the lowest energy. Outer shells have higher energy and are farther away. Electrons in higher energy levels are less tightly held by the nucleus.
Electron Capacity Per Shell
Each shell can hold a limited number of electrons. The maximum number is given by the formula 2n2, where n is the shell number. For example, the first shell (n=1) can hold 2 electrons. The second shell (n=2) can hold 8 electrons, and so on. This limit controls how many electrons an atom can contain in each shell.
Octet Rule And Stability
Atoms tend to be most stable when their outer shell has eight electrons. This is the octet rule. Atoms will gain, lose, or share electrons to reach this stable arrangement. The octet rule explains why atoms form bonds with others. Achieving a full outer shell lowers energy and increases stability.
Atomic Models
Atomic models explain how atoms are structured and how their parts behave. These models help us understand the tiny particles inside atoms. Over time, scientists improved these models to explain new discoveries.
Each model shows a different way of viewing the atom’s parts, like electrons, protons, and neutrons. Understanding these models is key to learning about matter and chemistry.
Thomson's Model
Thomson's model is called the "plum pudding" model. It shows the atom as a sphere of positive charge. Electrons are scattered inside like plums in a pudding. This model was the first to include electrons inside atoms. It helped explain how atoms are neutral overall.
Rutherford's Model
Rutherford’s model introduced the nucleus at the center of the atom. He discovered the nucleus is very small and positively charged. Electrons move around this nucleus, like planets orbiting the sun. This model explained the results of his gold foil experiment. It showed most of the atom is empty space.
Bohr's Model
Bohr improved Rutherford’s model by adding energy levels for electrons. Electrons orbit the nucleus in fixed paths or shells. Each shell holds a set number of electrons. This model explained why atoms emit light in specific colors. It helped understand atomic spectra and chemical behavior.
Quantum Mechanical Model
The quantum mechanical model is the most accurate and modern model. It describes electrons as waves, not just particles. Electrons do not have fixed orbits but exist in areas called orbitals. These orbitals show where electrons are likely to be found. This model uses math to predict electron behavior. It explains complex atomic properties and chemical reactions better than earlier models.
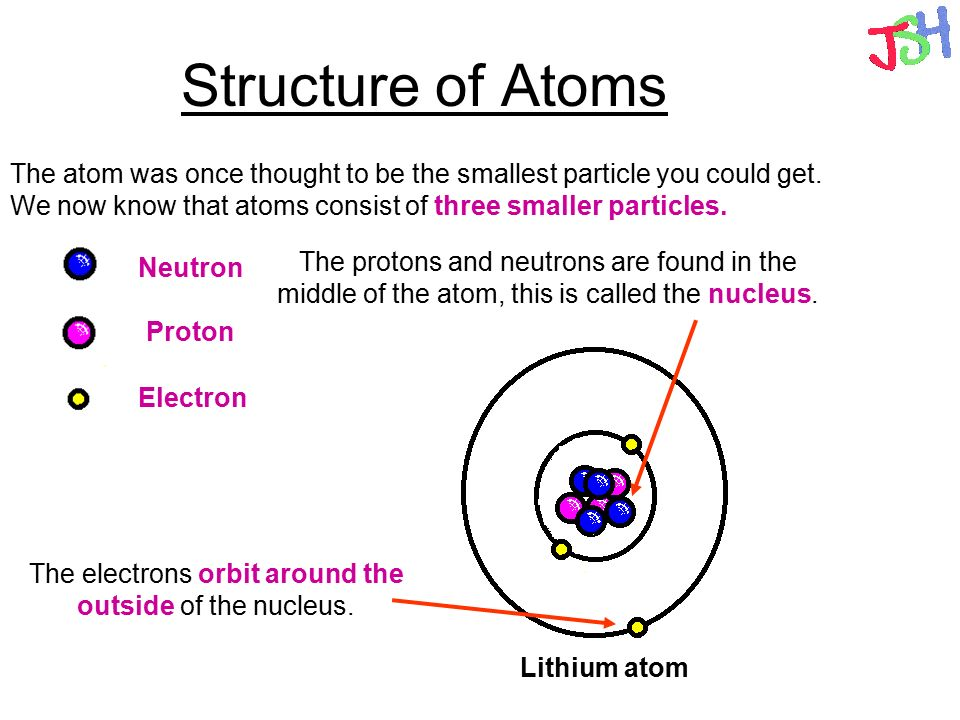
Credit: centrypump.amebaownd.com
Forces In Atoms
Atoms are held together by powerful forces that shape their structure and behavior. These forces work at tiny scales inside the atom. They keep the nucleus stable and control how electrons move around it. Understanding these forces helps explain many physical and chemical properties of matter.
Electromagnetic Force
The electromagnetic force acts between charged particles. It pulls negatively charged electrons toward the positively charged nucleus. This attraction keeps electrons orbiting the nucleus in defined paths. At the same time, electrons repel each other because they have the same negative charge. The balance of these forces determines the atom’s shape and size.
This force also causes atoms to interact and bond with each other. It is responsible for electricity, magnetism, and light. Without electromagnetic force, atoms could not form molecules or solids.
Nuclear Forces
Nuclear forces hold the protons and neutrons tightly inside the nucleus. Protons have positive charges and should repel each other strongly. But nuclear forces are stronger than this repulsion at very short distances. They bind protons and neutrons together to form a stable nucleus.
These forces are very strong but act only within the tiny nucleus. They do not affect electrons directly. Nuclear forces explain why atoms have a dense center. They also play a key role in nuclear reactions and energy release.
Applications In Physics
The atomic structure is central to many areas of physics. It helps explain how atoms behave and interact. Understanding this structure has led to advances in technology and science. The applications stretch across various fields, from studying light to predicting chemical reactions.
Spectral Lines
Spectral lines are unique colors of light emitted or absorbed by atoms. Each element produces a specific pattern of these lines. This happens because electrons jump between energy levels. Scientists use spectral lines to identify elements in stars and gases. They also help in developing lasers and other light-based technologies.
Chemical Behavior
The arrangement of electrons in an atom defines its chemical properties. Atoms with similar structures react in similar ways. This explains why elements form certain compounds. Understanding atomic structure helps predict reactions and design new materials. It also plays a role in medicine and environmental science.
Atomic Interactions
Atoms interact through forces between their electrons and nuclei. These interactions form bonds that create molecules. Studying atomic interactions reveals the nature of solids, liquids, and gases. It also helps explain electrical conductivity and magnetism. This knowledge supports advances in electronics and nanotechnology.

Credit: www.atomicarchive.com
Frequently Asked Questions
What Is The Definition Of Atomic Structure In Physics?
Atomic structure in physics describes an atom's composition: a central nucleus with protons and neutrons, surrounded by electrons orbiting in energy levels. Protons define the element, while electrons influence chemical behavior through electromagnetic attraction to the nucleus.
Is The Atomic Structure Physics?
Yes, atomic structure is a key concept in physics. It studies the arrangement of protons, neutrons, and electrons in an atom. This field explains how electrons orbit the nucleus and influence atomic behavior and interactions.
What Is The 2 8 8 18 18 Rule?
The 2 8 8 18 18 rule describes electron shell capacities in atoms. It shows maximum electrons per shell: 2, 8, 8, 18, then 18. This pattern helps explain atomic structure and electron arrangement in physics.
Is Atomic Structure In Physics Paper 1 Or 2?
Atomic structure is typically covered in Physics Paper 1. It focuses on the atom's nucleus, protons, neutrons, and electron arrangement.
Conclusion
Understanding atomic structure helps explain how atoms form matter around us. Atoms consist of a nucleus with protons and neutrons. Electrons orbit this nucleus in defined energy levels or shells. These arrangements influence chemical reactions and properties of elements. Scientists built models over time to better describe this tiny world.
Exploring atomic structure connects physics to everyday life and technology. It shows the building blocks of everything we see and use. A clear grasp of atoms opens doors to learning more about science.



No comments