Electron Configuration of Rubidium
Are you curious about how rubidium’s electrons are arranged and why it matters? Understanding the electron configuration of rubidium can unlock insights into its chemical behavior, reactivity, and place in the periodic table.
Whether you’re a student tackling chemistry homework or someone eager to deepen your knowledge, getting a clear grasp on rubidium’s electron setup will make complex concepts easier to follow. You’ll discover a simple, step-by-step explanation of rubidium’s electron configuration, how to write it correctly, and what it reveals about this fascinating element.
Ready to master this key topic? Let’s dive in and explore rubidium’s electrons like never before!
Credit: www.schoolmykids.com
Atomic Structure Of Rubidium
Rubidium is a soft, silvery metal found in the alkali metal group. Its atomic structure defines many of its chemical properties. Understanding this structure helps explain its behavior in reactions and its placement in the periodic table.
The atom consists of a nucleus surrounded by electrons arranged in energy levels. These electrons fill specific orbitals according to strict rules. The arrangement influences how rubidium interacts with other elements.
Electron Shells Of Rubidium
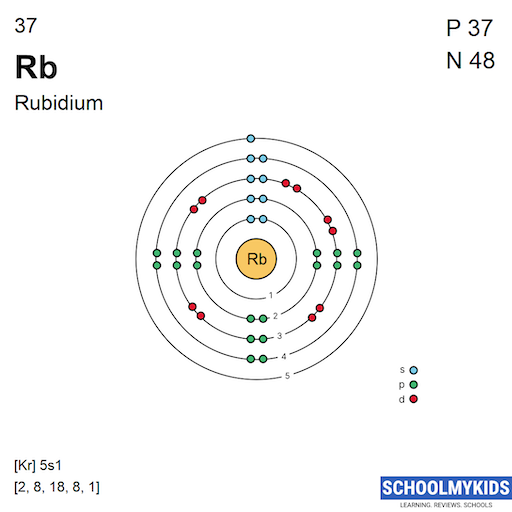
Rubidium has 37 electrons distributed in shells around its nucleus. These shells are numbered from 1 to 5, with each holding a certain number of electrons. The first shell holds 2 electrons, the second holds 8, and the third holds 18.
The fourth shell contains 8 electrons, and the fifth shell has only 1 electron. This single electron in the outermost shell plays a key role in rubidium’s chemical activity.
The electron configuration of rubidium is written as [Kr] 5s¹. This means it has the same electron arrangement as krypton, plus one extra electron in the 5s orbital. This notation simplifies the full configuration and shows its position on the periodic table.
The 5s¹ electron makes rubidium highly reactive and eager to lose this electron to form positive ions. This is typical of alkali metals, which have a single electron in their outermost shell.
Nuclear Composition And Stability
The nucleus of rubidium contains 37 protons and usually 48 neutrons. The number of protons defines the element as rubidium. Neutrons add to the mass and contribute to nuclear stability.
This balance of protons and neutrons creates a stable atom under normal conditions. The nucleus holds the positive charge and attracts the negatively charged electrons, keeping the atom intact.
Electron Shells And Subshells
Electron shells and subshells show how electrons arrange around an atom's nucleus. This arrangement helps explain an element's chemical behavior.
Rubidium has a unique electron configuration that follows specific rules. Understanding its shells and subshells helps us learn about its properties.
Electron Shells In Rubidium
Rubidium has five electron shells. Each shell holds a certain number of electrons. These shells are labeled from 1 to 5, starting from the closest to the nucleus.
The outermost shell in rubidium is the fifth shell. It contains the electron that mainly affects rubidium's reactions.
Subshells And Their Types
Each shell contains one or more subshells. Subshells are groups of orbitals where electrons live.
There are four types of subshells: s, p, d, and f. These subshells fill in a specific order as electrons add up.
Rubidium's Electron Subshell Filling
Rubidium's electrons fill subshells up to 5s. The full configuration ends with one electron in the 5s subshell.
This single 5s electron makes rubidium very reactive. It easily loses this electron to form positive ions.
Ground State Configuration
The ground state configuration of rubidium reveals the arrangement of its electrons in the lowest energy state. This configuration helps us understand the chemical behavior of rubidium. It shows how electrons fill the atomic orbitals in sequence.
Rubidium has an atomic number of 37, meaning it has 37 electrons. These electrons are distributed among different shells and subshells according to energy levels. The configuration follows the Aufbau principle, which states that electrons occupy the lowest energy orbitals first.
Basic Electron Structure Of Rubidium
Rubidium’s electrons fill the shells starting from the innermost to the outermost. The first 36 electrons fill orbitals up to the krypton core, a noble gas with a stable arrangement. This core is represented as [Kr].
Valence Electron In The 5s Orbital
The 37th electron enters the 5s subshell after the krypton core. This electron is the only one in the 5s orbital, making rubidium an alkali metal. The valence electron defines many properties, including reactivity and bonding.
Notation Of Ground State Configuration
The full ground state electron configuration of rubidium is written as [Kr] 5s¹. This notation shows the filled inner shells as [Kr] and the outermost single electron in the 5s subshell. It is simple and clear for understanding rubidium’s electron arrangement.
Using The Noble Gas Core
Using the noble gas core simplifies writing rubidium's electron configuration. This method replaces the inner electrons with the symbol of the nearest noble gas. It makes the configuration shorter and easier to read.
For rubidium, the noble gas core is krypton, which has a full set of electrons in its shells. After krypton, only the outer electrons of rubidium need to be shown. This highlights the electrons that are involved in chemical reactions.
What Is The Noble Gas Core?
The noble gas core is a shorthand way to write electron configurations. It uses the symbol of the last noble gas before the element. This represents all the inner, filled electron shells. It helps avoid writing long, complex configurations.
Rubidium's Noble Gas Core Configuration
Rubidium's atomic number is 37. The noble gas before rubidium is krypton, with atomic number 36. The configuration starts with [Kr], representing all electrons in krypton. Then, rubidium has one extra electron in the 5s orbital. So, the full configuration is written as [Kr] 5s¹.
Benefits Of Using Noble Gas Core
This method saves time and space in writing configurations. It focuses on the valence electrons, which control chemical behavior. It also makes comparing elements easier. Students and chemists use it to quickly understand electron structure.
Valence Electron Of Rubidium
Rubidium is a soft, silvery metal in the alkali metal group. Its chemical behavior depends on its valence electron. This electron is the outermost electron that participates in chemical reactions. Understanding rubidium’s valence electron helps explain its reactivity and bonding.
Rubidium has one valence electron. This single electron is in the 5s orbital. It sits outside the inner core electrons, making it easy to lose during reactions. This lone valence electron gives rubidium its characteristic properties.
Location Of Rubidium’s Valence Electron
The valence electron of rubidium is in the fifth shell. More precisely, it occupies the 5s orbital. This orbital is the outermost energy level for rubidium. The electron is loosely held because it is far from the nucleus.
Role Of The Valence Electron In Chemical Reactions
Rubidium’s valence electron is highly reactive. It easily loses this electron to form positive ions. This loss leads to the formation of Rb⁺ ions. These ions help rubidium bond with other elements, especially nonmetals.
Like other alkali metals, rubidium has one valence electron. Sodium has its valence electron in the 3s orbital. Potassium’s valence electron is in the 4s orbital. Rubidium’s 5s electron is further from the nucleus, making it more reactive.
Electron Configuration Notation
Electron configuration notation shows how electrons are arranged in an atom. It uses numbers and letters to describe the location of electrons. This helps us understand chemical behavior and properties of elements.
Rubidium has 37 electrons. Writing its electron configuration means placing these electrons in order. The notation follows energy levels and sublevels, from lowest to highest.
What Is Electron Configuration Notation?
Electron configuration notation uses numbers, letters, and superscripts. Numbers show the energy level or shell, such as 1, 2, 3, and so on. Letters represent sublevels: s, p, d, and f. Superscripts tell how many electrons are in each sublevel.
Electron Configuration Of Rubidium Explained
Rubidium’s electron configuration starts with filling lower shells. It follows the pattern: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s. Rubidium ends with 5s¹, meaning one electron in the 5s sublevel. This makes its full notation: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s¹.
Using Noble Gas Shorthand For Rubidium
To simplify, noble gas shorthand uses the symbol of the previous noble gas in brackets. For rubidium, krypton (Kr) is the noble gas before it. The shorthand notation is [Kr] 5s¹. This means rubidium has the same configuration as krypton, plus one electron in 5s.
Rubidium Ion Configuration
Rubidium forms ions by losing electrons to achieve a stable state. The most common ion is Rb+, which has a positive charge.
This ion forms by losing one electron from the outer shell. It results in a different electron configuration than neutral rubidium.
What Is The Electron Configuration Of Neutral Rubidium?
Neutral rubidium has 37 electrons. Its electron configuration ends with 5s1. This means it has one electron in the 5s orbital.
The full configuration is [Kr] 5s1. Here, [Kr] stands for the electron configuration of krypton, the previous noble gas.
How Does Rubidium Form Its Ion?
Rubidium loses its single 5s electron. This loss creates the Rb+ ion. The ion now has 36 electrons instead of 37.
Losing this electron gives rubidium a full octet, making the ion more stable.
Electron Configuration of the Rubidium Ion (Rb+)The Rb+ ion has the electron configuration [Kr]. This means it has the same electron setup as krypton.
It has a complete set of electrons in the 4p orbital, with no electrons in the 5s orbital.
This stable configuration explains why rubidium forms ions easily and participates in many chemical reactions.

Credit: www.youtube.com
Comparison With Other Alkali Metals
Rubidium shares many properties with other alkali metals. These elements all have one electron in their outermost s-orbital. Their electron configurations end with ns¹, where n increases down the group. This pattern affects their chemical behavior and physical traits.
Studying rubidium’s electron configuration alongside lithium, sodium, potassium, and cesium helps us understand trends in the group. These trends include atomic size, ionization energy, and reactivity. Each alkali metal adds a new electron shell, changing its properties step by step.
Electron Configuration Patterns In Alkali Metals
Alkali metals have a simple pattern in electron arrangements. Lithium is 1s² 2s¹. Sodium is [Ne] 3s¹. Potassium is [Ar] 4s¹. Rubidium continues this with [Kr] 5s¹. Cesium is [Xe] 6s¹. This pattern shows the filling of the outermost s-orbital with one electron.
Atomic Size And Electron Configuration
As you move down the alkali metals, atomic size grows. The new electron shell adds distance between the nucleus and outer electron. Rubidium has a larger atomic radius than potassium because it adds the fifth shell. This larger size influences how strongly the atom holds its valence electron.
Ionization Energy Trends
Ionization energy decreases down the group. Rubidium has a lower ionization energy than potassium. This is because its outer electron is farther from the nucleus and easier to remove. The electron configuration helps explain why rubidium loses its single valence electron more readily.
Chemical Reactivity Differences
Rubidium is more reactive than lithium, sodium, and potassium. The ease of losing the valence electron increases down the group. Rubidium’s [Kr] 5s¹ electron can be lost easily, making it highly reactive. This reactivity trend is tied directly to the electron configuration and atomic structure.
Role In Chemical Reactivity
Rubidium’s electron configuration plays a key role in its chemical behavior. Its outermost electron is in the 5s orbital, making it highly reactive. This single valence electron is easily lost during chemical reactions.
Because rubidium readily loses its 5s electron, it forms positive ions (Rb⁺) quickly. This tendency defines its place among alkali metals and explains its strong reactivity with nonmetals.
Electron Configuration And Valence Electrons
Rubidium’s electron arrangement is [Kr] 5s¹. The single 5s electron is loosely held. It is this electron that rubidium tends to lose, leading to chemical reactions. The loss forms a stable Rb⁺ ion with a noble gas configuration.
Formation Of Ions And Reactivity
Rubidium easily loses its outer electron to form a positive ion. This ion formation drives its chemical reactions. Rubidium reacts quickly with oxygen, water, and halogens. These reactions release energy and produce compounds.
Rubidium is more reactive than potassium but less than cesium. Its electron is easier to remove than potassium’s. This trend increases down the group due to atomic size and electron shielding.
Impact On Compound Formation
The single valence electron allows rubidium to form ionic bonds. It commonly bonds with halogens to create rubidium halides. These compounds are stable and have high melting points.

Credit: www.chegg.com
Orbital Diagram Representation
Electron configuration of rubidium shows how electrons fill orbitals around its nucleus. The orbital diagram represents these electrons with arrows in boxes, illustrating their arrangement and spin. This visual helps understand rubidium’s chemical behavior and placement in the periodic table.
Introduction To Orbital Diagram Representation
The orbital diagram shows how electrons fill the orbitals in an atom. It uses arrows to represent electrons and boxes for orbitals. Each arrow points up or down, indicating the electron's spin.
This diagram helps visualize the electron arrangement more clearly than just writing numbers. It reveals the distribution of electrons in each sublevel and orbital.
Orbital Diagram For Rubidium
Rubidium has 37 electrons to place in orbitals. Its electron configuration ends with 5s¹, meaning one electron is in the 5s orbital.
The diagram fills orbitals starting from the lowest energy level. The first 36 electrons fill the orbitals up to the 4p sublevel, matching the noble gas krypton (Kr).
The last electron occupies the 5s orbital alone, shown by a single arrow in the 5s box.
How To Draw The Orbital Diagram For Rubidium
Begin with the 1s orbital and add two arrows pointing in opposite directions. Continue filling orbitals in order: 2s, 2p, 3s, 3p, 4s, 3d, 4p.
Once the 36 electrons fill these orbitals, add one arrow in the 5s orbital. This completes rubidium’s electron arrangement.
Significance Of The Orbital Diagram
The diagram shows the lone electron in the 5s orbital. This electron determines rubidium’s chemical behavior. It is easily lost, making rubidium highly reactive.
Understanding the orbital diagram helps explain rubidium’s place in the periodic table. It clarifies why rubidium behaves like other alkali metals.
Frequently Asked Questions
How To Write The Electronic Configuration Of Rubidium?
Write rubidium’s electron configuration by starting with krypton’s core: [Kr] 5s¹. It has one electron in the 5s orbital.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 represents the element Zinc (Zn) with atomic number 30.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 represents the element Cobalt (Co). It has 27 electrons and is a transition metal in the d-block.
Which Atom Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 belongs to the chromium (Cr) atom. Chromium has an atomic number of 24 and exhibits this unique configuration due to electron stability in its d-orbital.
Conclusion
Understanding rubidium’s electron configuration reveals its place in the periodic table. Rubidium has one electron in its 5s orbital beyond the krypton core. This simple arrangement explains many of its chemical behaviors. Knowing this helps predict how rubidium reacts with other elements.
It also shows why rubidium is part of the alkali metals group. The electron setup guides scientists and students alike. It makes learning about rubidium easier and more interesting. Remember, each element’s electrons tell a unique story. Rubidium’s story is straightforward but important in chemistry.



No comments