Electron Configuration of Gallium
Are you curious about how the electrons arrange themselves in gallium? Understanding the electron configuration of gallium is key to unlocking its chemical behavior and unique properties.
Whether you're a student trying to master chemistry or just someone fascinated by the building blocks of elements, this guide will walk you through gallium’s electron setup step-by-step. By the end, you’ll see how its electrons fill different shells and subshells, making gallium the special element it is.
Ready to discover the hidden pattern behind gallium’s electrons? Let’s dive in and make it simple for you!
Credit: homework.study.com
Gallium Basics
Gallium is a soft, silvery metal that melts near room temperature. It belongs to the group 13 elements on the periodic table. Its atomic number is 31, which means it has 31 protons and electrons.
Understanding gallium’s electron configuration helps explain its chemical behavior. Electrons arrange themselves in energy levels around the nucleus. These arrangements follow specific rules and patterns.
Electron shells are the main energy levels where electrons reside. Gallium’s electrons fill shells starting from the closest to the nucleus. The first shell holds 2 electrons, the second shell holds 8, and the third shell holds 18 electrons.
Subshell Filling Order
Within each shell, electrons occupy smaller regions called subshells. These are labeled as s, p, d, and f. Gallium’s electrons fill subshells in a specific order based on energy. The 4s subshell fills before the 3d subshell.
The Aufbau principle guides how electrons fill orbitals. Electrons fill lower energy orbitals first before moving to higher ones. This principle helps predict the electron configuration of gallium accurately.
Bohr Model And Its Limits
The Bohr model shows electrons in fixed orbits around the nucleus. It works well for lighter elements but is less accurate for gallium. Gallium’s electrons behave more like clouds than fixed paths.
Atomic Structure
The atomic structure of gallium explains how its electrons are arranged around the nucleus. This arrangement affects the element's chemical behavior and properties. Understanding gallium's atomic structure helps in studying its reactions and uses.
Gallium has 31 protons in its nucleus, which defines its atomic number. It also has 31 electrons orbiting the nucleus. These electrons fill specific energy levels called shells and subshells.
Electron shells are layers around the nucleus where electrons reside. Gallium’s electrons fill these shells in order of increasing energy. The first shell holds up to 2 electrons, the second up to 8, and the third up to 18.
Gallium’s electron distribution in shells is 2, 8, 18, and then 3 electrons in the outermost shell. This outer shell determines gallium’s chemical properties.
Subshell Filling Order
Each shell contains subshells labeled s, p, d, and f. Electrons fill these subshells in a specific order based on their energy levels. For gallium, the order is 1s, 2s, 2p, 3s, 3p, 4s, 3d, then 4p subshells.
Gallium’s last electrons enter the 4p subshell, giving it unique chemical traits compared to other elements.
The Aufbau principle guides electron placement in atoms. Electrons fill the lowest energy subshells first before moving to higher levels. This rule helps predict gallium’s electron configuration accurately.
Following the Aufbau principle, gallium’s electrons occupy the 4s subshell before filling 3d and then 4p subshells.
Bohr Model Of Gallium
The Bohr model shows electrons orbiting the nucleus in fixed paths. Though simple, it helps visualize gallium’s electron arrangement in shells. This model works well for lighter atoms but is a basic view for gallium.
Using the Bohr model, gallium’s electrons are placed in four energy levels, matching its shell structure.
Limitations Of The Bohr Model
The Bohr model cannot explain all properties of gallium. It ignores electron interactions and quantum mechanics. Modern atomic theory uses complex shapes of orbitals to describe electron positions more accurately.
Despite its limits, the Bohr model is a good starting point to understand gallium’s atomic structure.
Electron Shells
Gallium’s electron shells hold 31 electrons arranged by energy levels. Its configuration ends with 4p¹, showing how electrons fill shells and subshells. This pattern helps explain gallium’s chemical behavior.
Electron Shells Of Gallium
Gallium has electrons arranged in shells around its nucleus. These shells hold electrons at different energy levels. Each shell can contain one or more subshells.
Gallium's electron shells follow a specific order based on energy. The shells are labeled as 1, 2, 3, 4, and so on. Each shell holds a set number of electrons.
Shell 1 And 2: The Inner Shells
The first shell of gallium holds 2 electrons. It is the closest shell to the nucleus. The second shell holds 8 electrons. These two shells are full and stable.
Electrons in these shells have lower energy. They stay close to the nucleus and do not react much in chemical processes.
Shell 3: The Third Shell
The third shell of gallium holds 18 electrons. This shell has three subshells: s, p, and d. Gallium fills these subshells before moving to the next shell.
This shell contains 3s², 3p⁶, and 3d¹⁰ electrons. The 3d subshell fills after the 4s subshell but belongs to the third shell.
Shell 4: The Outer Shell
The fourth shell of gallium holds electrons that define its chemical properties. It contains 4s² and 4p¹ electrons. These are the valence electrons.
Valence electrons in shell 4 participate in bonding and reactions. The presence of one electron in 4p makes gallium reactive.
Subshells And Orbitals
The electron configuration of gallium reveals how electrons are arranged in its atoms. This arrangement depends on subshells and orbitals, which are key to understanding atomic structure. Subshells group orbitals with similar energy levels. Orbitals are regions where electrons are most likely found.
Gallium has 31 electrons. These electrons fill subshells in a specific order. Each subshell contains one or more orbitals. The shape and energy of orbitals vary by type.
What Are Subshells?
Subshells are divisions within an electron shell. They are labeled as s, p, d, and f. Each letter represents a different shape and energy level. The s subshell has one orbital, p has three, d has five, and f has seven. Electrons fill these subshells in a defined sequence.
Orbitals In Gallium
Orbitals hold electrons in pairs with opposite spins. Gallium’s electron configuration ends in the 4p subshell. It fills orbitals starting from 1s to 4p. The 4p orbitals are important as they hold the valence electrons. These electrons influence gallium’s chemical behavior.
Filling Order Of Gallium’s Subshells
Electrons fill subshells by increasing energy levels. The filling order follows the Aufbau principle. For gallium, electrons fill 1s, 2s, 2p, 3s, 3p, 4s, 3d, and finally 4p subshells. The 3d subshell fills after 4s because of energy differences. This order explains gallium’s unique properties.
Aufbau Principle
The Aufbau Principle guides how electrons fill the atomic orbitals. It states that electrons occupy the lowest energy orbitals first. This helps build the electron configuration of elements step by step. Using this rule, we can predict the arrangement of electrons in Gallium.
Gallium has 31 electrons. These electrons fill orbitals starting from the lowest energy level. The Aufbau Principle ensures a stable and accurate electron layout for Gallium.
What Is The Aufbau Principle?
The Aufbau Principle means "building up" in German. It tells us electrons fill orbitals from low to high energy. This order helps avoid confusion in electron arrangements. It forms the base for writing electron configurations.
How Aufbau Principle Applies To Gallium
Gallium’s electrons fill orbitals starting with 1s, then 2s, and so on. Each orbital is filled according to its energy level. Following this rule, Gallium’s electron configuration becomes clear and systematic.
Order Of Subshell Filling
The filling order follows this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p. Gallium’s electrons fill up to the 4p orbital. This order respects the Aufbau Principle and creates a stable electronic structure.

Credit: www.chegg.com
Gallium Electron Configuration
Gallium is a chemical element with atomic number 31. It has 31 electrons that fill its atomic orbitals in a specific order. Understanding gallium’s electron configuration helps explain its chemical behavior and bonding properties.
The electron configuration shows how electrons are distributed in shells and subshells around the nucleus. This arrangement follows rules based on quantum mechanics and energy levels.
Gallium Electron Shells
Gallium’s electrons occupy several shells, starting from the innermost. The shells are numbered 1, 2, 3, and so on, moving outward from the nucleus.
The first shell holds 2 electrons, the second holds 8, and the third shell holds more as it fills. Gallium’s electrons fill these shells in order of increasing energy.
Subshell Filling Order
Electrons fill subshells in a specific sequence: s, p, d, and f. Each subshell can hold a certain number of electrons. The s subshell holds 2, p holds 6, d holds 10, and f holds 14.
For gallium, the 3d subshell fills before the 4p subshell because it has lower energy. This order affects how the electrons arrange themselves.
Aufbau Principle Applied To Gallium
The Aufbau principle states electrons fill the lowest energy orbitals first. Gallium’s configuration follows this rule strictly. It fills the 1s, 2s, 2p, 3s, 3p, and 3d orbitals before the 4s and 4p orbitals.
This principle helps predict gallium’s electron configuration accurately and explains its chemical properties.
Gallium Electron Configuration Notation
The full electron configuration of gallium is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p¹. This notation lists all occupied orbitals and their electron counts.
In shorthand using the noble gas preceding gallium, it is written as [Ar] 3d¹⁰ 4s² 4p¹. This form is simpler and often used in chemistry.
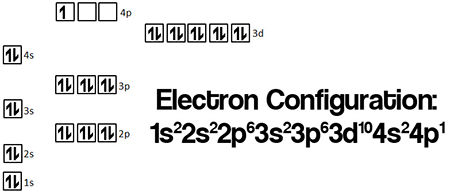
Orbital Diagram
The orbital diagram of gallium shows how its electrons fill different orbitals. It helps visualize electron arrangement in shells and subshells. This diagram follows the Aufbau principle and Pauli exclusion rule for correct electron placement.
Understanding The Orbital Diagram
The orbital diagram shows how electrons fill the orbitals in an atom. It uses arrows to represent electrons and boxes for orbitals. Each box stands for an orbital that can hold up to two electrons. The arrows point up or down, indicating electron spin.
This diagram helps visualize electron arrangement beyond the simple electron configuration. It follows specific rules to show the most stable form of the atom. These rules include filling lower energy orbitals first and pairing electrons only when necessary.
Orbital Diagram Of Gallium
Gallium has 31 electrons. Its orbitals fill in a specific order based on energy levels. The diagram starts with the 1s orbital and moves to higher orbitals step by step. Each orbital is shown with arrows to mark electron spins.
The order for gallium is: 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p. The 4p orbital contains only one electron for gallium. This shows that gallium’s outer shell is not fully filled, affecting its chemical properties.
Rules Followed In The Orbital Diagram
The Aufbau principle guides the filling order of orbitals. Electrons enter the lowest energy orbitals first. Hund's rule states that electrons fill empty orbitals singly before pairing. The Pauli exclusion principle allows only two electrons per orbital with opposite spins.
These rules ensure the diagram accurately represents gallium’s electron distribution. The orbital diagram helps understand gallium’s reactivity and bonding behavior. It gives a clear picture of where electrons are located within the atom.
Valence Electrons
Valence electrons are the outermost electrons in an atom. They play a key role in chemical reactions and bonding. Understanding these electrons helps explain gallium's properties and behavior.
Number Of Valence Electrons In Gallium
Gallium has three valence electrons. These electrons are found in the 4th energy level. Specifically, they occupy the 4s and 4p orbitals. This makes gallium part of group 13 in the periodic table.
Electron Configuration Of Gallium's Valence Shell
The valence shell of gallium is written as 4s² 4p¹. This means two electrons fill the 4s orbital. One electron occupies the 4p orbital. The 3d orbitals are filled but belong to an inner shell, not the valence shell.
Importance Of Valence Electrons In Gallium
Valence electrons determine how gallium bonds with other elements. Gallium tends to lose three electrons to form a +3 charge. This behavior influences its use in semiconductors and alloys. Knowing valence electrons helps predict gallium's chemical reactions.
Comparison With Neighbors
Understanding the electron configuration of gallium becomes clearer when comparing it to its neighboring elements. This comparison highlights subtle differences in electron arrangement that affect chemical behavior.
Gallium sits in group 13 of the periodic table. Its neighbors include zinc to the left and germanium to the right. Each element’s electron configuration reveals unique patterns in how electrons fill their shells.
Electron Configuration Of Zinc And Gallium
Zinc has the electron configuration [Ar] 3d10 4s2. Gallium follows with [Ar] 3d10 4s2 4p1. The key difference is gallium’s extra 4p electron. This single electron in the 4p orbital gives gallium distinct chemical properties compared to zinc.
Comparison With Germanium
Germanium’s configuration is [Ar] 3d10 4s2 4p2. It has one more 4p electron than gallium. This small change affects its reactivity and bonding. Germanium behaves more like a metalloid, while gallium acts as a metal.
Impact On Chemical Properties
Gallium’s electron arrangement influences its softness and melting point. Its neighbors show trends in metallic character due to their electron filling. Comparing their configurations helps explain these differences.
Applications And Importance
The electron configuration of gallium plays a key role in its various uses and significance. This element's unique arrangement of electrons influences its chemical behavior and physical properties.
Understanding gallium's electron configuration helps explain why it works well in certain technologies and industries. Its position in the periodic table and electron structure make it valuable in electronics and materials science.
Applications In Electronics
Gallium is essential in making semiconductors. Its electron configuration allows it to combine with other elements like arsenic. This forms gallium arsenide, a material widely used in LEDs and solar cells. Gallium's ability to conduct electricity efficiently makes it ideal for high-speed devices.
Role In Material Science
The electron configuration affects gallium's melting point and bonding. Gallium can melt just above room temperature, which is unusual for metals. This property allows it to be used in alloys and cooling systems. Its structure helps create materials with special electrical and thermal properties.
Importance In Chemistry Education
Gallium's electron configuration serves as a good example for teaching atomic structure. It shows how electrons fill different shells and subshells. Students learn about the Aufbau principle and electron distribution by studying gallium. This strengthens their understanding of chemical elements and reactions.

Credit: www.youtube.com
Frequently Asked Questions
How To Write Electron Configuration For Gallium?
Write gallium’s electron configuration as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p¹. Follow the Aufbau principle and fill orbitals by increasing energy levels.
What Element Has An Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1?
The element with the electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 is Rubidium (Rb). It belongs to Group 1, Period 5 on the periodic table.
Is The 2 8 8 18 Rule Correct?
The 2-8-8-18 rule simplifies electron distribution in shells but is not fully accurate for heavier elements. It works mainly for lighter atoms.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ belongs to the element Ruthenium (Ru).
Conclusion
Understanding the electron configuration of gallium helps explain its chemical behavior. Gallium’s unique arrangement of electrons influences its bonding and reactivity. This knowledge is important for studying metals and their properties. Remember, gallium has electrons filling the 4p orbital after the 3d and 4s orbitals.
This pattern follows the Aufbau principle and affects gallium’s place in the periodic table. Knowing this can make learning chemistry easier and more interesting. Keep exploring how electron configurations shape elements and their uses.



No comments