Electron Configuration of Germanium
If you’ve ever wondered what makes germanium tick at the atomic level, understanding its electron configuration is the key. Knowing how electrons are arranged in germanium’s atoms not only reveals its chemical behavior but also helps you predict how it interacts with other elements.
Whether you’re a student trying to master chemistry or simply curious about this fascinating metalloid, this article will guide you step-by-step through germanium’s electron setup. By the end, you’ll be confident in writing its electron configuration and grasping why germanium holds such an important place in science and technology.
Ready to unlock the secrets of germanium’s electrons? Let’s dive in!

Credit: www.youtube.com
Germanium Basics
The atomic structure of germanium reveals how its electrons are arranged around the nucleus. This arrangement affects its chemical behavior and physical properties. Understanding this structure helps explain germanium's role in electronics and semiconductors.
Germanium has an atomic number of 32. This means it has 32 protons in its nucleus and 32 electrons orbiting it. The electrons fill different energy levels or shells, each with specific sublevels.
Electron Shells And Subshells
Electrons in germanium occupy shells around the nucleus. These shells are labeled 1, 2, 3, 4, and so on. Each shell contains one or more subshells: s, p, d, and f. The subshells hold electrons in specific patterns.
For germanium, electrons fill the first three shells fully and partially fill the fourth shell. The subshells fill in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p.
Electron Configuration Of Germanium
The electron configuration shows how electrons distribute in shells and subshells. Germanium’s configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p². This means the first shell has 2 electrons in the s subshell. The second shell has 8 electrons in s and p subshells combined.
The third shell has 18 electrons, filling s, p, and d subshells. The fourth shell contains 4 electrons in s and p subshells. This arrangement gives germanium its unique characteristics.
Importance Of Atomic Structure
Germanium’s atomic structure controls its chemical reactions and bonding. The electrons in the outer shell, especially in 4s and 4p subshells, are important. These outer electrons interact with other atoms and form bonds.
This makes germanium useful in semiconductors and electronics. Its atomic structure allows it to conduct electricity under certain conditions. This property is key to its use in technology.

Credit: www.youtube.com
Atomic Structure
Germanium has 32 electrons arranged in energy levels around its nucleus. Its electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p². This setup shows how electrons fill shells and orbitals in the atom.
Understanding The Orbital Filling Order
The orbital filling order explains how electrons arrange themselves in an atom. Electrons fill orbitals starting from the lowest energy level. This order follows specific rules to keep the atom stable. The order affects the chemical behavior of elements like germanium.
Germanium has 32 electrons. These electrons fill orbitals in a sequence that follows the Aufbau principle. This principle states that electrons occupy the lowest energy orbitals first before moving to higher ones.
How Electrons Fill The Orbitals In Germanium
First, electrons fill the 1s orbital. Then they move to the 2s and 2p orbitals. Next, the 3s and 3p orbitals fill with electrons. After these, the 4s orbital fills before the 3d orbitals.
For germanium, the 3d orbitals fill after 4s is fully occupied. The 3d orbitals can hold up to 10 electrons. Finally, electrons fill the 4p orbitals, which are the outermost orbitals in germanium.
Energy Levels And Subshells In Germanium
Electrons in germanium occupy four main energy levels. These are levels 1 through 4. Each energy level contains one or more subshells: s, p, and d. The s subshell holds 2 electrons, p holds 6, and d holds 10.
The order of filling goes from 4s to 3d, then to 4p. This happens because 4s has lower energy than 3d. Understanding this helps explain germanium’s position in the periodic table.
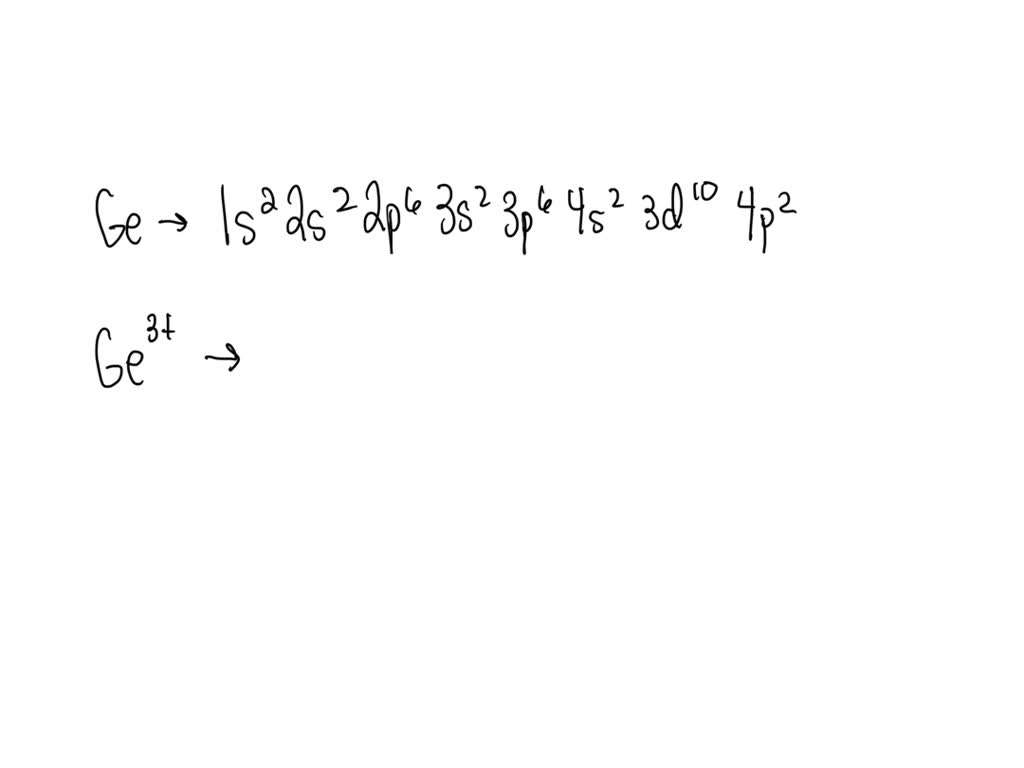
Credit: www.numerade.com
Frequently Asked Questions
How Do You Write The Electron Configuration For Germanium?
Write germanium’s electron configuration as 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p². This shows all electrons in their orbitals by increasing energy levels.
What Element Has An Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 is Rubidium (Rb). It has atomic number 37.
What Is The 2 8 8 18 18 32 Rule?
The 2 8 8 18 18 32 rule describes electron distribution in atomic shells. It shows maximum electrons each shell can hold. This rule helps predict element electron configurations in chemistry and physics.
Which Atom Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 belongs to the chromium (Cr) atom. This configuration shows chromium's unique stability with a half-filled 3d subshell.
Conclusion
Understanding the electron configuration of germanium helps explain its chemical behavior. Germanium has 32 electrons arranged in specific shells and subshells. This arrangement influences its position in the periodic table and its properties. Knowing this structure makes it easier to study germanium’s role in electronics and materials science.
Clear and simple electron patterns reveal how atoms bond and interact. Keep these basics in mind when exploring germanium or similar elements. This knowledge builds a strong foundation for learning chemistry concepts.



No comments