Electron Configuration of Cadmium
Are you curious about how cadmium’s electrons are arranged and why this matters? Understanding the electron configuration of cadmium isn’t just for chemistry experts—it’s a key to unlocking how this element behaves in reactions and materials.
Whether you’re a student, a science enthusiast, or someone working with metals, knowing cadmium’s electron layout can sharpen your grasp of its properties and uses. You’ll discover exactly how cadmium’s electrons fill its orbitals, what that means for its chemical behavior, and why this knowledge can make a difference in your studies or work.
Keep reading to get clear, simple explanations that will boost your confidence in mastering this essential concept.
Credit: chem.libretexts.org
Basic Properties Of Cadmium
Cadmium is a soft, bluish-white metal found in the periodic table. It belongs to the group 12 elements along with zinc and mercury. This metal is known for its unique physical and chemical properties. These properties make cadmium useful in various industrial applications.
Cadmium has a relatively low melting point and is quite malleable. It does not react quickly with air at room temperature, which helps it resist corrosion. However, cadmium can be toxic, so handling it requires care and safety measures.
Atomic Number And Symbol
The atomic number of cadmium is 48, meaning it has 48 protons in its nucleus. Its chemical symbol is Cd. This number places cadmium in the d-block of the periodic table, specifically in period 5 and group 12.
Electron Configuration
Cadmium’s electron configuration is [Kr] 4d10 5s2. This shows that cadmium has a full 4d subshell and two electrons in the 5s subshell. The full d subshell gives cadmium stability and affects its chemical behavior.
Physical Characteristics
Cadmium is soft and can be cut easily with a knife. It has a melting point of 321°C and a boiling point of 767°C. The metal is dense but lighter than many transition metals. It appears shiny and silvery when freshly cut.
Chemical Properties
Cadmium resists corrosion in air but slowly oxidizes when heated. It reacts with acids to form cadmium salts. Cadmium compounds often show toxicity, so they need careful handling. The metal can form bonds with other elements, especially sulfur and oxygen.

Credit: www.youtube.com
Atomic Number And Electrons
The atomic number defines the number of protons in an atom. For cadmium, this number is 48. This means cadmium has 48 protons in its nucleus. In a neutral atom, the number of electrons equals the number of protons. Therefore, cadmium also contains 48 electrons.
Electrons arrange themselves in energy levels or shells around the nucleus. The way electrons are distributed affects the element’s chemical behavior. Understanding the atomic number and electrons is key to learning cadmium’s electron configuration.
What Is Cadmium’s Atomic Number?
Cadmium’s atomic number is 48. This number places it in the periodic table as a transition metal. It tells us the total positive charge in the nucleus. The atomic number is unique to each element.
How Many Electrons Does Cadmium Have?
Cadmium has 48 electrons in its neutral state. These electrons balance the positive charge of the protons. They fill the shells and subshells in order of increasing energy. This arrangement determines the element’s properties.
Why Atomic Number And Electrons Matter
The atomic number and electron count define cadmium’s identity. They guide how electrons fill orbitals around the nucleus. This influences cadmium’s chemical bonds and reactions. Knowing these helps us understand its place in chemistry.
Electron Shells And Subshells
Understanding the electron shells and subshells of cadmium helps explain its chemical behavior. Electrons are arranged in shells around the nucleus. Each shell contains one or more subshells. These subshells hold electrons in different energy levels and shapes. This arrangement affects how cadmium bonds and reacts with other elements.
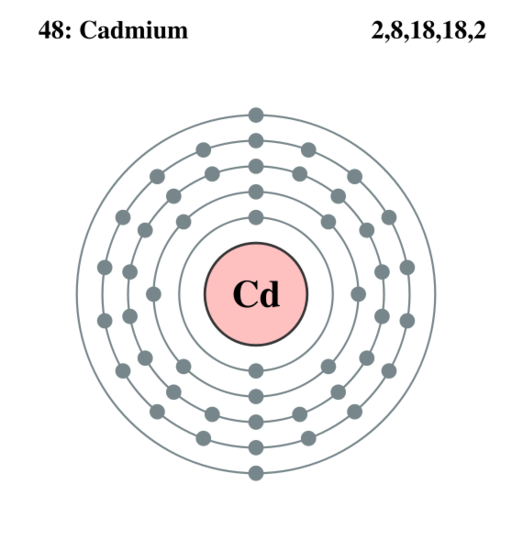
Electron Shells Of Cadmium
Cadmium has five electron shells. These shells are labeled from 1 to 5, starting from the nucleus outward. Each shell can hold a specific number of electrons. The outermost shell determines the element’s reactivity. For cadmium, the fifth shell contains the valence electrons that interact with other atoms.
Subshells And Their Types
Within each shell, electrons occupy subshells named s, p, d, and f. Each subshell has a different shape and capacity for electrons. The s subshell holds 2 electrons, p holds 6, d holds 10, and f holds 14. Cadmium’s electron configuration includes s and d subshells in its outer shells. These subshells play key roles in its chemical properties.
Cadmium’s Electron Distribution
Cadmium’s electrons fill the shells and subshells in a specific order. The first two shells fill completely with s and p electrons. The third and fourth shells fill with s, p, and d electrons. The fifth shell contains the 5s electrons. The 4d subshell is fully filled, which influences cadmium’s stability and behavior in compounds.
Credit: chem.libretexts.org
Ground State Electron Configuration
The ground state electron configuration shows how electrons fill orbitals in an atom.
This arrangement is the lowest energy state for the electrons.
Understanding the ground state helps explain chemical behavior and bonding.
Basic Electron Structure Of Cadmium
Cadmium has 48 electrons, matching its atomic number.
These electrons occupy specific orbitals in order of increasing energy.
Step-by-step Electron Filling
Electrons fill the 1s orbital first, then move to higher levels.
The sequence follows the Aufbau principle, filling lower energy orbitals first.
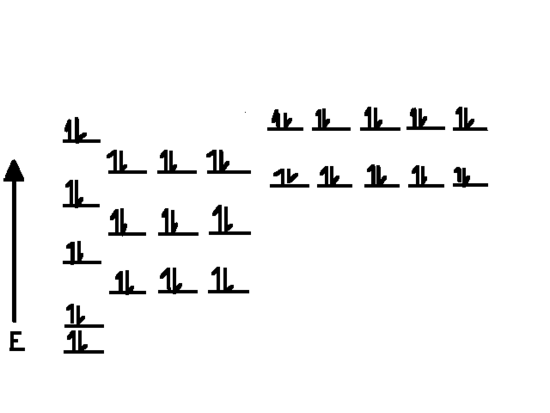
Cadmium’s Complete Ground State Configuration
The full electron configuration for cadmium is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰
This shows filled 4d and 5s orbitals, typical for a transition metal.
Orbital Filling Order
The orbital filling order explains how electrons occupy the orbitals of an atom. Electrons fill orbitals starting from the lowest energy level. This order follows specific rules to minimize the atom's energy.
Understanding cadmium's orbital filling helps reveal its chemical behavior. Cadmium has 48 electrons to place in orbitals. The order of filling determines its electron configuration.
Aufbau Principle
The Aufbau principle states electrons fill orbitals from lowest to highest energy. Electrons occupy the 1s orbital first, then move outward. This principle guides the sequence for cadmium’s electrons.
Pauli Exclusion Principle
This principle says each orbital holds two electrons with opposite spins. It prevents electrons from having the same quantum state. Cadmium’s electrons pair up in orbitals following this rule.
Hund’s Rule
Hund’s rule requires electrons to fill degenerate orbitals singly first. Electrons spread out to minimize repulsion. In cadmium, this applies especially in the d and p orbitals.
Energy Levels And Sublevels For Cadmium
Cadmium’s electrons fill 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, and 4d orbitals. The 5s orbital fills before 4d due to lower energy. The 4d sublevel fills last with ten electrons.
Electron Configuration Of Cadmium Ions
Cadmium ions form when cadmium atoms lose electrons. Understanding their electron configuration helps explain their chemical behavior. Cadmium usually forms a +2 ion by losing two electrons. These changes alter the electron arrangement around the nucleus.
Electron Configuration Of Neutral Cadmium Atom
The neutral cadmium atom has 48 electrons. Its configuration ends in the 4d subshell, which is fully filled. The full configuration is [Kr] 4d10 5s2. This means the 5s orbital holds two electrons, and the 4d orbital holds ten.
Formation of Cd2+ IonWhen cadmium becomes Cd2+, it loses two electrons. These electrons come from the 5s orbital first. The 4d orbitals remain fully occupied. So, the 5s2 electrons are removed, leaving only the 4d electrons.
Electron Configuration of Cd2+ IonThe Cd2+ ion has the configuration [Kr] 4d10. Losing the 5s electrons makes the ion more stable. This configuration explains Cd2+'s common chemical properties and bonding patterns.
Magnetic Properties Of Cadmium Ions
Cd2+ is diamagnetic because all electrons are paired. The filled 4d orbitals have no unpaired electrons. This affects how cadmium ions interact with magnetic fields and other atoms.
Magnetic Properties Related To Configuration
The magnetic properties of cadmium are directly linked to its electron configuration. Understanding this connection helps explain why cadmium behaves the way it does in magnetic fields. The arrangement of electrons in different orbitals influences whether the element is magnetic or not.
Cadmium has a unique electron structure that affects its magnetism. The electrons fill certain orbitals completely, which plays a big role in its magnetic response. This section explores how cadmium’s configuration shapes its magnetic traits.
Electron Configuration Of Cadmium
Cadmium’s electron configuration is [Kr] 4d10 5s2. This means its 4d and 5s orbitals are fully filled. Complete filling of these orbitals results in paired electrons. Paired electrons have opposite spins, canceling out magnetic effects.
Diamagnetism In Cadmium
Because all electrons are paired, cadmium is diamagnetic. Diamagnetic materials create a weak magnetic field opposite to an applied magnetic field. This causes cadmium to be slightly repelled by magnets. It has no unpaired electrons to generate a strong magnetic moment.
Effect Of Ionization On Magnetism
When cadmium loses electrons to form Cd2+, it loses the two 5s electrons. The 4d orbitals remain full with ten electrons. As a result, Cd2+ is also diamagnetic. Its electron pairing stays intact, preventing magnetic attraction.
Comparison With Neighboring Elements
Comparing the electron configuration of cadmium with its neighboring elements helps us understand its unique chemical behavior. Cadmium sits in the d-block of the periodic table, and its electron arrangement influences its properties and reactions.
Examining elements around cadmium reveals patterns in electron filling and stability. These patterns explain differences in reactivity and bonding among these metals.
Electron Configuration Of Cadmium
Cadmium has the electron configuration [Kr] 4d10 5s2. This means its 4d subshell is fully filled with 10 electrons, while the 5s subshell contains 2 electrons. The filled d-subshell adds to cadmium’s chemical stability.
Comparison With Zinc (zn)
Zinc, located two places before cadmium, has the configuration [Ar] 3d10 4s2. Like cadmium, zinc has a fully filled d-subshell and two s-electrons. Both elements show similar low reactivity due to filled d orbitals. Zinc’s electrons are in lower energy levels, making it lighter and less dense than cadmium.
Comparison With Mercury (hg)
Mercury follows cadmium and has the configuration [Xe] 4f14 5d10 6s2. It also has a filled d-subshell and two s-electrons but includes a filled 4f subshell. Mercury’s electron arrangement leads to its liquid state at room temperature, unlike solid cadmium. This difference results from relativistic effects affecting mercury’s electrons.
Differences With Silver (ag)
Silver has the configuration [Kr] 4d10 5s1. It has one less s-electron compared to cadmium. This single s-electron allows silver to form more varied chemical bonds. Cadmium’s fully paired s-electrons limit its bonding types, making it less reactive than silver.
Role Of D-orbitals In Cadmium
Cadmium’s electron configuration ends with filled 4d orbitals, giving it stable chemical properties. The d-orbitals hold ten electrons, influencing its bonding and reactivity. These orbitals play a key role in cadmium’s behavior as a transition metal.
Electron Configuration And The D-orbitals In Cadmium
Cadmium has an atomic number of 48. Its electron configuration ends in the 4d orbitals. These d-orbitals play a key role in its chemical behavior.
The 4d orbitals fill after the 5s orbital. This filling impacts cadmium's stability and reactivity. The fully filled 4d sublevel makes cadmium less reactive compared to other transition metals.
Impact Of Filled 4d Orbitals On Chemical Properties
Cadmium's 4d orbitals are completely filled with 10 electrons. This full d-subshell leads to a stable electron arrangement. It affects cadmium’s bonding and its limited ability to form complex ions.
The filled d-orbitals reduce the number of unpaired electrons. This results in cadmium being mostly diamagnetic. The chemical reactions of cadmium often involve the 5s electrons instead.
Role Of D-orbitals In Cadmium’s Oxidation States
Cadmium typically shows a +2 oxidation state. The 4d electrons remain paired and do not usually participate in bonding. This is because the 4d sublevel is fully occupied and stable.
The 5s electrons are lost in ion formation. The d-orbitals help maintain the structure of the atom during this process. This explains why cadmium’s chemistry is different from other transition metals.
Influence Of D-orbitals On Cadmium’s Physical Characteristics
The filled d-orbitals contribute to cadmium’s metallic properties. They affect the element’s color, density, and conductivity. These orbitals help cadmium maintain its shiny, silver-white appearance.
Cadmium has a relatively low melting point among transition metals. The electron configuration, especially the d-orbitals, influences this physical trait. It also affects how cadmium interacts with other metals in alloys.
Applications Influenced By Electron Setup
The electron configuration of cadmium plays a crucial role in determining its applications. Cadmium’s unique electron setup affects its chemical and physical properties. These properties influence how cadmium behaves in various industries. Understanding this link helps explain cadmium’s wide range of uses.
Cadmium has the electron configuration [Kr] 4d10 5s2. This full d-subshell provides stability and specific reactivity. The filled 4d orbitals make cadmium less reactive than some other transition metals. This stability is key to its role in batteries, pigments, and coatings.
Battery Technology And Electron Stability
Cadmium’s electron arrangement allows it to store and release energy efficiently. Nickel-cadmium (NiCd) batteries use cadmium for this reason. The stable 4d electrons help maintain consistent voltage output. These batteries are durable and can handle many charge cycles. This makes them useful in portable electronics and power tools.
Corrosion-resistant Coatings
The electron configuration also impacts cadmium’s corrosion resistance. Cadmium coatings protect metals from rust and wear. Its electrons form a protective barrier on steel and iron surfaces. This barrier slows down oxidation and extends the metal’s life. Cadmium plating is common in aerospace and automotive parts.
Pigments And Color Stability
Cadmium’s electron setup affects the colors it produces in pigments. Cadmium compounds create bright, stable colors in paints and plastics. The filled d-orbitals contribute to light absorption and color intensity. These pigments resist fading under sunlight. Artists and manufacturers value cadmium-based pigments for their durability.
Frequently Asked Questions
What Element Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5 belongs to Bromine (Br). It has 35 electrons.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 corresponds to the element Cobalt (Co), atomic number 27.
Which Element Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s1 3d5 is Chromium (Cr). It has 24 electrons.
Is Cd Always +2?
Cadmium (Cd) commonly forms a +2 oxidation state by losing two 5s electrons. It is not always +2 but most stable.
Conclusion
Understanding cadmium’s electron configuration helps explain its chemical behavior. Cadmium has 48 electrons arranged in specific orbitals. This arrangement impacts its bonding and reactivity. Knowing the electron structure supports studies in chemistry and materials science. It also aids learning about related elements in the periodic table.
This knowledge is useful for students and professionals alike. Keep exploring electron configurations to better grasp element properties. Simple steps can clarify complex atomic details.



No comments