Electron Configuration of Tungsten
Have you ever wondered what makes tungsten such a powerful and unique element? Understanding its electron configuration is the key to unlocking its special properties.
You’ll discover how the arrangement of tungsten’s electrons shapes its behavior and uses. By the end, you’ll have a clear and simple grasp of this concept, making tungsten’s role in science and technology easier to appreciate. Keep reading—you’re about to uncover something fascinating about one of the most important elements on the periodic table!
Credit: www.priyamstudycentre.com
Basic Properties Of Tungsten
Tungsten is a unique metal known for its strength and heat resistance. It has special properties that make it useful in many industries. Understanding these properties helps explain why tungsten is so valuable.
This metal has a very high melting point. It can withstand extreme heat without melting or breaking down. These traits make tungsten ideal for use in light bulbs and rocket engines.
Atomic Number And Symbol
Tungsten has the atomic number 74. Its chemical symbol is W, from the German word "Wolfram." This symbol is used in science and industry worldwide.
Density And Hardness
Tungsten is very dense, heavier than many other metals. It is also very hard and strong. These qualities allow it to resist scratching and wear in tough conditions.
Melting And Boiling Points
Tungsten has the highest melting point of all metals. It melts at 3422°C (6192°F). Its boiling point is even higher, at 5555°C (10031°F). This makes it perfect for high-temperature tools.
Electron Configuration
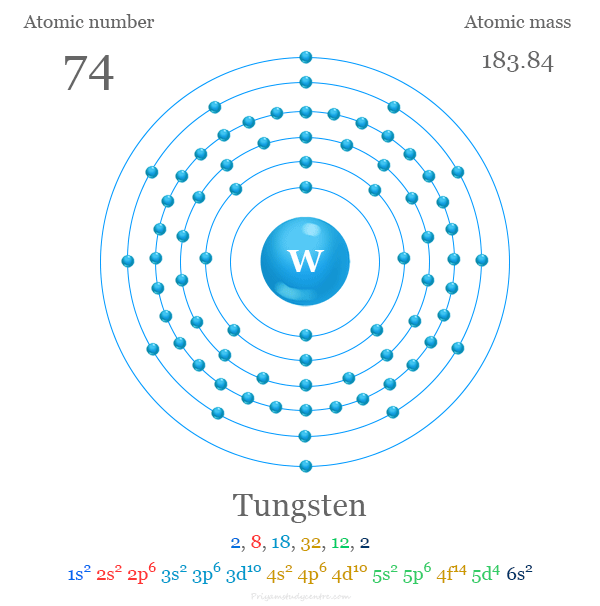
The electron configuration of tungsten is complex. It is written as [Xe] 4f14 5d4 6s2. This arrangement affects how tungsten bonds with other elements.
Atomic Structure Of Tungsten
Tungsten is a heavy metal with unique atomic properties. Its atomic structure defines its behavior and uses. Understanding this structure helps explain its strength and high melting point.
The atomic structure of tungsten includes protons, neutrons, and electrons. These particles arrange in specific ways. This arrangement determines chemical properties and physical traits.
Electron Configuration Of Tungsten
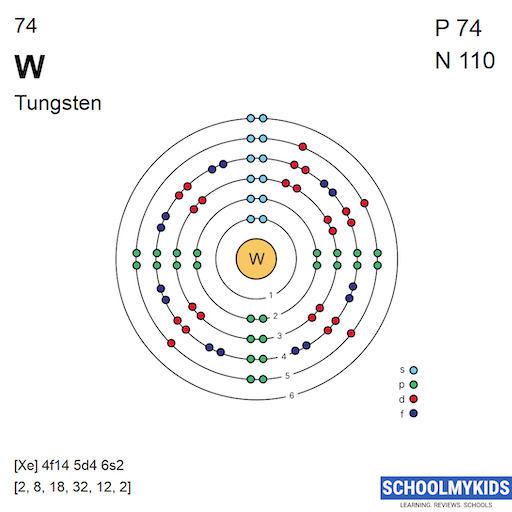
Tungsten has 74 electrons. These electrons fill orbitals in a defined order. The electron configuration is [Xe] 4f14 5d4 6s2. This shows the distribution of electrons in shells and subshells.
Protons And Neutrons In The Nucleus
The nucleus contains 74 protons. It also holds about 110 neutrons. These particles give tungsten its atomic mass of around 184.
Orbital Structure And Energy Levels
Electrons in tungsten occupy different orbitals. These orbitals have specific shapes and energy levels. The 5d and 6s orbitals play a key role in tungsten’s chemistry.
Electron Configuration Basics
Understanding electron configuration is key to learning how atoms behave. It shows how electrons arrange around an atom's nucleus. This arrangement affects an element's chemical properties and reactions.
Each atom has energy levels called shells. These shells hold electrons in smaller groups known as subshells. The subshells fill in a specific order based on energy levels.
What Is Electron Configuration?
Electron configuration is the order of electrons in shells and subshells. It uses numbers and letters to show electron positions. The numbers tell the shell, and letters show the subshell type.
Shells And Subshells Explained
Shells are numbered 1, 2, 3, and so on. Each shell has subshells named s, p, d, and f. Subshells hold different numbers of electrons. For example, s holds 2 electrons, p holds 6.
Rules For Filling Electrons
Electrons fill the lowest energy level first. They follow the Aufbau principle, filling shells step by step. Each subshell fills completely before electrons enter the next one.
Importance Of Electron Configuration
Electron configuration helps predict how elements react chemically. It also explains an element’s place in the periodic table. Scientists use it to understand bonding and material properties.
Ground State Configuration
Tungsten’s ground state configuration shows how its electrons fill energy levels. It follows a unique pattern due to its atomic structure. This arrangement helps explain tungsten’s chemical behavior.
What Is Ground State Configuration?
The ground state configuration shows how electrons are arranged in an atom.
It describes the lowest energy state of an atom. Electrons fill orbitals from low to high energy.
This arrangement helps explain many properties of elements, like chemical behavior and bonding.
Ground State Configuration Of Tungsten
Tungsten has 74 electrons. These electrons fill the orbitals in a specific order.
The ground state electron configuration of tungsten is [Xe] 4f14 5d4 6s2.
This means tungsten's electrons fill up to the xenon core, then add electrons to 4f, 5d, and 6s orbitals.
Why Tungsten’s Configuration Is Unique
Tungsten's 5d electrons have a special pattern due to electron interactions.
The 5d orbitals fill after the 6s, which is unusual but stable for tungsten.
This configuration affects tungsten's strength and high melting point.
Orbital Filling Order
The orbital filling order explains how electrons arrange themselves in an atom. It follows specific rules to fill orbitals from lower to higher energy levels. Understanding this order helps us know the behavior and properties of elements like tungsten.
Electrons fill orbitals to create the most stable arrangement. This pattern follows the Aufbau principle, Pauli exclusion principle, and Hund's rule. These rules guide electron placement in each orbital.
Aufbau Principle
The Aufbau principle states that electrons occupy the lowest energy orbitals first. This means orbitals closer to the nucleus fill before outer ones. For tungsten, this principle helps predict its electron configuration step by step.
Pauli Exclusion Principle
The Pauli exclusion principle says no two electrons can have the same set of quantum numbers. Each orbital can hold two electrons with opposite spins. This rule limits how many electrons fit in each orbital.
Hund’s Rule
Hund’s rule requires that electrons fill degenerate orbitals singly before pairing. This reduces electron repulsion and increases stability. For tungsten, this means electrons spread out in orbitals within the same sublevel.
Exceptions In Electron Configuration
Electron configuration usually follows a clear pattern based on energy levels. Tungsten, a transition metal, shows some exceptions to these patterns. These exceptions help explain its unique chemical behavior.
Understanding these exceptions is key to grasping tungsten’s role in chemistry. Let’s explore the main exceptions in tungsten’s electron configuration.
What Causes Exceptions In Electron Configuration?
Electrons fill orbitals to reach the lowest energy state. Sometimes, half-filled or fully filled subshells offer extra stability. This stability causes electrons to rearrange differently than expected. Tungsten shows this effect in its d and s orbitals.
Electron Configuration Of Tungsten
Expected configuration: [Xe] 4f14 5d4 6s2. Actual configuration: [Xe] 4f14 5d5 6s1. One electron moves from 6s to 5d for stability. This creates a half-filled d subshell, which is more stable.
Why Tungsten Prefers A Half-filled D Subshell
Half-filled subshells reduce electron repulsion and increase stability. Tungsten’s 5d orbitals gain stability with five electrons. The single electron in the 6s orbital balances the overall energy. This arrangement lowers tungsten’s total energy.
Electron Configuration And Chemical Properties
Tungsten’s electron configuration plays a key role in its chemical behavior. It defines how tungsten atoms interact with other elements. This interaction shapes tungsten’s unique properties.
The arrangement of electrons affects tungsten’s stability and reactivity. Understanding this arrangement helps explain tungsten’s common uses.
Basic Electron Configuration Of Tungsten
Tungsten has 74 electrons. Its full electron configuration is [Xe] 4f14 5d4 6s2. This means tungsten fills the 4f and 5d orbitals before the 6s orbital. The electrons in the 5d and 6s orbitals influence its chemical properties most.
Effect On Chemical Reactivity
The 5d electrons make tungsten less reactive than many metals. Tungsten forms strong bonds with other elements. It resists corrosion and oxidation. This resistance makes it useful in harsh environments.
Role In Bond Formation
Tungsten can share or lose its 6s and 5d electrons during reactions. This ability allows it to form various compounds. It often forms complex ions with other metals and nonmetals.
Impact On Physical Properties
The electron structure contributes to tungsten’s high melting point. Strong metallic bonds result from its electron configuration. This gives tungsten great strength and durability.
Credit: www.schoolmykids.com
Tungsten Ions And Their Configurations
Tungsten forms several ions by losing electrons from its neutral atom. Each ion has a unique electron configuration. These configurations explain tungsten’s chemical behavior and properties.
Tungsten ions mainly lose electrons from the 6s and 5d orbitals. The changes affect their stability and reactivity. Understanding these helps in fields like chemistry and materials science.
W+ Ion Electron ConfigurationThe W+ ion loses one electron from the 6s orbital. Its configuration is [Xe] 4f14 5d4 6s1. This ion is less common but important in some reactions.
W2+ Ion Electron ConfigurationThe W2+ ion loses two electrons, both from the 6s orbital. Its configuration is [Xe] 4f14 5d4. This ion shows higher stability than W+.
W6+ Ion Electron ConfigurationThe W6+ ion loses six electrons. The configuration becomes [Xe] 4f14. This ion is very stable and common in tungsten compounds.
Spectroscopic Notation For Tungsten
Spectroscopic notation offers a clear way to write the electron configuration of tungsten. It uses numbers and letters to show how electrons fill different orbitals. This method helps scientists and students understand the structure of atoms quickly.
Tungsten, with the atomic number 74, has a complex electron setup. Spectroscopic notation simplifies this by grouping electrons into energy levels and orbital types. This notation also shows the number of electrons in each orbital.
Spectroscopic Notation Explained
In spectroscopic notation, numbers represent energy levels. Letters stand for orbital types: s, p, d, and f. Superscript numbers show how many electrons fill each orbital. For example, 1s2 means two electrons in the first energy level’s s orbital.
Electron Configuration Of Tungsten In Spectroscopic Notation
Tungsten’s configuration is written as 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d4. This shows tungsten’s electrons filling orbitals from low to high energy.
Importance Of Spectroscopic Notation For Tungsten
This notation helps in studying tungsten’s chemical behavior. It shows electron distribution clearly. Scientists use it to predict how tungsten reacts with other elements. It also helps in understanding tungsten’s physical properties.

Credit: www.researchgate.net
Applications Influenced By Electron Configuration
The electron configuration of tungsten plays a key role in its many uses. Its unique arrangement of electrons makes it special. This affects how it reacts with other elements and how it conducts electricity. Understanding these details helps explain why tungsten is important in many fields.
Each electron in tungsten fills specific energy levels. These levels influence the metal’s strength, melting point, and electrical behavior. These traits lead to important practical applications in industry and technology.
Electron Configuration And Electrical Conductivity
Tungsten has high electrical conductivity. This comes from its electron arrangement. The electrons can move freely, carrying electric current. This makes tungsten useful in electronics and lighting. It is often used in wires and contacts where good conductivity is needed.
Impact On Thermal Resistance
The electron configuration contributes to tungsten’s high melting point. This makes it very heat resistant. Industries use tungsten in parts that must handle extreme heat. Examples include furnace components and rocket engine parts. Its ability to resist heat comes from how its electrons hold the atoms together.
Role In Chemical Stability
Tungsten’s electron layout gives it chemical stability. It does not easily react with many chemicals. This makes tungsten useful in harsh environments. It can resist corrosion and wear. This property is important for tools and machinery exposed to tough conditions.
Frequently Asked Questions
What Is The Electron Configuration Of Tungsten?
Tungsten's electron configuration is [Xe] 4f14 5d4 6s2. It shows the arrangement of electrons in its atomic orbitals.
Why Is Tungsten's Electron Configuration Important?
Understanding tungsten's electron configuration helps explain its chemical properties and reactivity. It also aids in material science studies.
How Does Tungsten's Electron Configuration Affect Its Properties?
The 5d and 6s electrons contribute to tungsten's high melting point and strength. These electrons influence its conductivity and durability.
What Does [xe] Mean In Tungsten's Configuration?
[Xe] represents the electron configuration of xenon, a noble gas. It simplifies tungsten's full electron configuration notation.
Conclusion
Tungsten’s electron configuration helps explain its unique traits. It shows how electrons fill different energy levels. This pattern affects tungsten’s strength and conductivity. Understanding this can aid studies in chemistry and physics. It also helps with learning about other transition metals.
Remember, each element has its own electron setup. This detail makes the study of elements interesting and useful. Keep exploring to see how electrons shape the world around us.



No comments