Electron Configuration of Rutherfordium
Have you ever wondered what makes Rutherfordium unique on the periodic table? Understanding its electron configuration unlocks the secrets of this mysterious, man-made element.
By diving into Rutherfordium’s electron arrangement, you’ll discover how its structure influences its properties and place among transition metals. Whether you’re a student, science enthusiast, or curious learner, grasping this concept will deepen your knowledge of chemistry and the elements that shape our world.
Ready to explore Rutherfordium’s electron configuration and see what sets it apart? Let’s get started!
Basic Properties
Rutherfordium is a synthetic element with unique basic properties. It exists only in laboratories and has no stable isotopes. Understanding its atomic details helps grasp its place in chemistry.
This section covers Rutherfordium’s atomic number, symbol, position in the periodic table, and its physical and chemical traits.
Atomic Number And Symbol
Rutherfordium’s atomic number is 104. Its chemical symbol is Rf. The atomic number shows it has 104 protons in its nucleus. This number defines the element’s identity.
Position In Periodic Table
Rutherfordium belongs to the d-block of the periodic table. It is part of the transition metals group. It sits in period 7, group 4. This position links it to titanium, zirconium, and hafnium.
Physical And Chemical Traits
Rutherfordium is a heavy, radioactive metal. It is unstable and decays quickly. Its chemistry likely resembles hafnium’s. Scientists predict it forms similar compounds but study is limited.
Electron Configuration Details
The electron configuration of Rutherfordium reveals how its electrons are arranged in atomic orbitals. Understanding this arrangement helps explain its chemical behavior and position in the periodic table. This section breaks down the details clearly and simply.
Ground State Configuration
Rutherfordium has the atomic number 104, meaning it has 104 electrons. Its ground state electron configuration is [Rn] 5f14 6d2 7s2. This shows that Rutherfordium fills the 5f, 6d, and 7s orbitals. The configuration reflects its place among the transactinide elements.
Noble Gas Notation
The noble gas notation for Rutherfordium uses the symbol for Radon (Rn) to simplify the configuration. Radon has 86 electrons, so Rutherfordium’s configuration starts as [Rn]. Then, the electrons in the outer shells are added: 5f14 6d2 7s2. This notation makes it easier to write and understand.
Comparison With Hafnium
Hafnium, element 72, is in the same group as Rutherfordium. Its electron configuration is [Xe] 4f14 5d2 6s2. Both have similar outer electron patterns. However, Rutherfordium’s electrons are in higher energy levels (5f, 6d, 7s) compared to Hafnium’s (4f, 5d, 6s). This difference affects their chemical and physical properties.
Electron Shell Structure
The electron shell structure of Rutherfordium reveals how its electrons arrange around its nucleus. This structure impacts its chemical behavior and physical properties. Rutherfordium has a complex electron configuration due to its high atomic number of 104.
Understanding this arrangement helps in grasping Rutherfordium’s place in the periodic table. It also explains its similarities and differences with other transition metals.
Filling Of D-block Orbitals
Rutherfordium belongs to the d-block elements. Its electrons fill the 6d orbitals after the 7s orbitals are occupied. The electron configuration ends with 6d² 7s², showing two electrons in the 6d subshell. This filling order follows the Aufbau principle, where orbitals fill from lower to higher energy levels. The d-block filling plays a key role in its chemical properties.
Valence Electrons
Valence electrons are the outermost electrons involved in bonding. For Rutherfordium, the valence shell includes the 7s and 6d electrons. It typically has four valence electrons: two in 7s and two in 6d orbitals. These electrons determine how Rutherfordium interacts with other atoms. The valence shell influences its oxidation states and bonding behavior.
Effect Of Electron Configuration On Reactivity
The electron configuration affects Rutherfordium’s reactivity significantly. The presence of 6d electrons allows it to form bonds like other transition metals. However, its heavy atomic weight and relativistic effects alter its chemical behavior. Rutherfordium tends to show similar reactivity to Hafnium, its lighter homologue. Its electron structure makes it less reactive than lighter transition metals but more reactive than some heavier elements.

Credit: commons.wikimedia.org
Radioactivity And Stability
Rutherfordium is a synthetic element known for its strong radioactivity. Its atoms are unstable and break down quickly. This instability affects how scientists study its electron arrangement. Understanding its radioactivity helps explain its chemical behavior and placement in the periodic table.
The element’s short lifespan limits the amount of data researchers can collect. Despite this, studying Rutherfordium gives insight into heavy elements and their properties. Its radioactivity plays a key role in defining its electron configuration and stability.
Isotopes And Half-lives
Rutherfordium has several isotopes, all radioactive. These isotopes have half-lives ranging from milliseconds to a few hours. The most common isotope, Rutherfordium-267, has a half-life of about 1.3 hours. Such short half-lives make it hard to study the element in detail.
The decay of these isotopes releases radiation and changes the element into other atoms. This rapid decay influences the stability of Rutherfordium’s electron shells. Scientists must use special equipment to observe these fleeting atoms.
Impact On Electron Configuration Studies
Rutherfordium’s radioactivity limits direct observation of its electron configuration. Researchers rely on theoretical models and comparisons with lighter elements. These models predict the arrangement of electrons in Rutherfordium’s outer shells.
Its unstable nature challenges experiments but also pushes advances in detection technology. Understanding electron configuration in such elements helps refine the periodic table’s structure. It also guides the search for new elements with longer lifetimes.
Synthesis And Occurrence
The synthesis and occurrence of Rutherfordium reveal its unique place among chemical elements. This element does not exist naturally on Earth. Scientists create it under controlled conditions for study. Understanding how it is made and where it is found helps explain its electron configuration and behavior.
Man-made Creation
Rutherfordium is a synthetic element created by nuclear reactions. It is made by bombarding heavy atoms with lighter ions. This process fuses the nuclei and forms new atoms. The element is highly unstable and exists only for a short time. Its creation requires advanced technology and specialized equipment.
Laboratory Production Methods
Scientists produce Rutherfordium in particle accelerators. They use targets like Californium or Plutonium. These targets are hit with ions such as Carbon or Neon. The collision causes nuclear fusion, forming Rutherfordium atoms. Researchers detect the new atoms by their radioactive decay. This method allows the study of its properties despite its short life.
Natural Occurrence Status
Rutherfordium does not occur naturally in the environment. Its atoms decay too quickly to be found in nature. It appears only in laboratory settings. Because it is synthetic, it has no practical uses outside research. This rarity makes Rutherfordium important mainly for scientific exploration.

Credit: www.webelements.com
Applications And Research
Rutherfordium’s electron configuration plays a key role in ongoing scientific studies. This element is synthetic and highly unstable. Its applications mainly focus on research rather than practical use. Scientists explore its properties to understand heavy elements better.
Research on rutherfordium helps expand knowledge of the periodic table’s transactinide series. The element’s electron arrangement affects its chemical behavior. These insights guide further experiments and theories about superheavy elements.
Scientific Importance
Rutherfordium is important in nuclear chemistry and physics. It helps scientists learn about atomic structure at extreme atomic numbers. Studying its electron configuration reveals trends in electron shells and subshells. This knowledge improves models of atomic behavior in heavy elements.
Its position in the periodic table allows comparison with lighter elements. Such comparisons clarify how electrons fill orbitals in heavy atoms. This understanding supports the development of new chemical theories.
Potential Uses
Rutherfordium’s practical uses remain limited due to its short half-life. It has no commercial or industrial applications. Scientists use it mainly for experimental purposes in laboratories. These experiments may contribute to advances in nuclear science and materials research.
Future discoveries might lead to new technologies involving transactinide elements. Current research focuses on its chemical reactions and bonding tendencies. This work could inspire innovations in synthetic element chemistry.
Challenges In Study
Rutherfordium’s instability presents major challenges for research. Its isotopes decay rapidly, limiting observation time. Producing sufficient quantities requires complex nuclear reactors or particle accelerators. This process is costly and technically demanding.
Measuring electron configuration directly is difficult because of its short existence. Scientists rely on indirect methods and theoretical calculations. These methods require advanced equipment and expertise to ensure accuracy.
Related Elements
Rutherfordium is a unique element with interesting connections to other elements. Understanding its related elements helps to see its place in the periodic table. These connections reveal trends in chemical behavior and electron structure. It also shows how Rutherfordium fits within its group and period.
Transactinide Elements
Rutherfordium is the first element in the transactinide series. These elements have atomic numbers greater than 103. They are all synthetic and radioactive. Their electron configurations often involve the filling of 5f and 6d orbitals. This makes their chemistry complex and less predictable. Transactinides share similar properties due to their position beyond the actinides. Studying them helps scientists understand the limits of the periodic table.
Comparison With Other Group 4 Elements
Rutherfordium belongs to group 4, alongside titanium, zirconium, and hafnium. These elements share similar electron configurations in their outer shells. All have two electrons in their d-orbitals, influencing their chemical reactions. However, Rutherfordium's heavy atomic mass and radioactivity affect its properties. It behaves similarly to hafnium but with subtle differences. These variations arise from relativistic effects on its electrons. Comparing these elements highlights trends in size, reactivity, and stability.
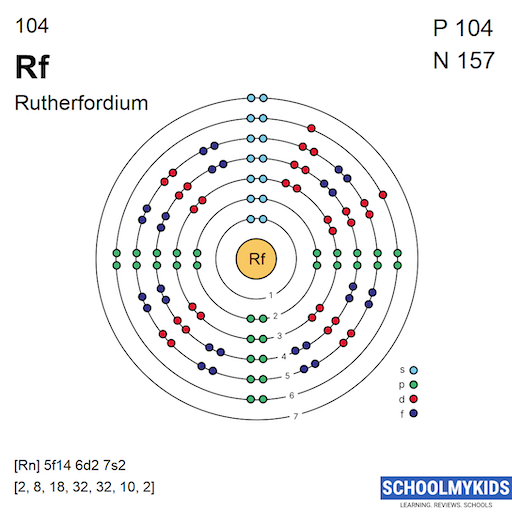
Credit: www.schoolmykids.com
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 corresponds to the element calcium (Ca). It has 20 electrons and is an alkaline earth metal. This configuration shows filled shells up to the 4s orbital, indicating calcium's position in the periodic table.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration is 2, 8, 18 for elements beyond argon. The 2, 8, 8 pattern applies only to elements up to calcium.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn). It has atomic number 25.
Which Element Is This 1s22s22p63s23p64s23d104p5?
The electron configuration 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁵ belongs to the element Bromine (Br). It has atomic number 35.
Conclusion
Understanding Rutherfordium’s electron configuration reveals its place on the periodic table. It belongs to the d-block, showing traits of transition metals. Its electrons fill shells similarly to hafnium but with more complexity. This helps scientists study its chemical behavior and properties.
Although rare and radioactive, Rutherfordium adds to our knowledge of heavy elements. Exploring its electron setup supports research in advanced chemistry. This element’s structure is a key to understanding transactinide elements better.



No comments