Electron Configuration of Erbium
If you want to truly understand the unique properties of erbium, getting to know its electron configuration is essential. The arrangement of electrons around an atom's nucleus determines how it behaves, reacts, and interacts with other elements.
By exploring the electron configuration of erbium, you’ll unlock insights into its position on the periodic table, its chemical characteristics, and why it’s valuable in technology and industry. Keep reading to discover the step-by-step breakdown of erbium’s electrons and how this knowledge can deepen your grasp of chemistry.
Credit: www.schoolmykids.com
Erbium Basics
Erbium is a chemical element with the symbol Er and atomic number 68. It belongs to the lanthanide series, also known as rare earth elements. These elements are known for their unique electronic properties and complex electron configurations.
Erbium is a silvery-white metal that is stable in air. It has a hexagonal crystal structure and reacts slowly with oxygen. This element finds uses in lasers, fiber optics, and nuclear technology.
Atomic Number And Symbol
Erbium's atomic number is 68. This means it has 68 protons in its nucleus. The symbol "Er" identifies it on the periodic table. The atomic number helps determine its electron arrangement.
Position In The Periodic Table
Erbium is part of the lanthanide series, located in period 6. It sits between holmium and thulium. This position influences its chemical behavior and electron configuration.
Physical And Chemical Properties
Erbium is a soft metal with a high melting point. It has a hexagonal close-packed crystal structure. The element is moderately reactive, especially with acids and oxygen.
Electron Configuration Overview
Erbium’s electrons fill orbitals in a specific order. Its configuration ends in the 4f subshell, which is typical for lanthanides. This arrangement affects its magnetic and optical properties.
Atomic Number And Mass
The atomic number and mass are fundamental properties of erbium. They define its identity and position in the periodic table.
The atomic number tells how many protons are in the nucleus. For erbium, this number is 68.
The mass number represents the total count of protons and neutrons. Erbium has a mass number around 167.
Atomic Number Of Erbium
Erbium’s atomic number is 68. This means each erbium atom has 68 protons. The number of electrons is also 68 in a neutral atom. This balance keeps the atom stable.
Mass Number Of Erbium
The mass number varies slightly due to isotopes. The most common isotope has a mass of 167. It has 99 neutrons and 68 protons together.
Importance Of Atomic Number And Mass
The atomic number defines chemical behavior and element type. The mass influences the element’s stability and physical properties. Both numbers are key in understanding erbium’s role in science.
Protons, Neutrons, And Electrons
Understanding the particles inside an erbium atom helps explain its electron configuration. Erbium is a chemical element with unique properties. These depend on three main particles: protons, neutrons, and electrons. Each particle plays a specific role in the atom's structure and behavior.
Protons carry a positive charge and define the element's identity. Neutrons have no charge but add to the atom's mass. Electrons carry a negative charge and fill different energy levels around the nucleus. Their arrangement forms the electron configuration.
Protons In Erbium
Erbium has 68 protons in its nucleus. This number is called the atomic number. It determines the element's position in the periodic table. The positive charge of protons balances the negative charge of electrons. This balance keeps the atom stable.
Neutrons In Erbium
The number of neutrons in erbium varies by isotope. The most common isotope has 99 neutrons. Neutrons add mass without changing the charge. They help hold the nucleus together. This strengthens the atom’s core.
Electrons In Erbium
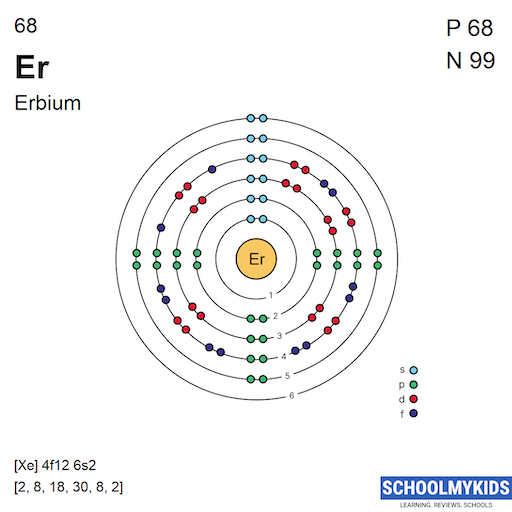
Erbium has 68 electrons orbiting the nucleus. These electrons fill specific orbitals in a set order. The arrangement follows the Aufbau principle. Electrons fill lower energy levels first. Erbium’s electron configuration ends in 4f and 6s orbitals. This pattern affects its chemical properties and reactivity.

Credit: www.youtube.com
Full Electron Configuration
The full electron configuration reveals how electrons are arranged in an atom. It shows each energy level and sublevel occupied by electrons.
For Erbium, this configuration explains its chemical behavior and position in the periodic table. Erbium belongs to the lanthanide series.
Understanding The Electron Shells Of Erbium
Erbium has an atomic number of 68, meaning it has 68 electrons. These electrons fill shells starting from the lowest energy level.
The shells are labeled as 1s, 2s, 2p, 3s, and so on. Electrons fill these shells in a specific order guided by energy rules.
Step-by-step Electron Filling For Erbium
First, the 1s orbital holds 2 electrons. Next, the 2s and 2p orbitals fill with 8 electrons combined.
Continuing this order, the 3s, 3p, 4s, 3d, 4p, 5s, 4d, and 5p orbitals fill with their respective electrons. This accounts for 46 electrons.
The remaining 22 electrons occupy the 6s and 4f orbitals, completing the configuration for Erbium.
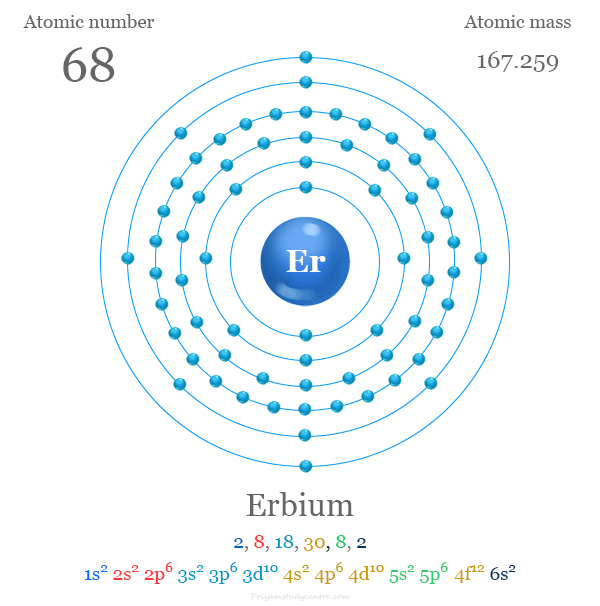
Complete Electron Configuration Of Erbium
The full electron configuration of Erbium is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s² 4f¹²
This layout shows that Erbium has 12 electrons in the 4f sublevel and 2 in the 6s sublevel.
The 4f electrons give Erbium its unique magnetic and optical properties.
Abbreviated Configuration
The abbreviated electron configuration simplifies the full electron arrangement of an element. It uses the nearest noble gas to represent core electrons. This method makes complex configurations easier to read and understand.
For erbium, the abbreviated configuration highlights its position among the lanthanides. It shows valence electrons clearly, which helps explain chemical behavior. This format is useful for students and scientists alike.
What Is Abbreviated Electron Configuration?
Abbreviated configuration starts with a noble gas symbol in brackets. This represents all inner-shell electrons. Then, the remaining electrons are listed to complete the element’s configuration.
Abbreviated Configuration Of Erbium
Erbium’s atomic number is 68. Its abbreviated configuration begins with [Xe], the noble gas with 54 electrons. Then it continues with 4f and 6s orbitals: [Xe] 4f12 6s2.
Why Use The Abbreviated Configuration?
It shortens long electron listings. It focuses on outer electrons that affect chemical properties. It also helps in comparing elements quickly. This style reduces confusion for learners and professionals.
Orbital Diagram
The orbital diagram provides a clear visual of how electrons fill the orbitals in an atom. It shows the arrangement of electrons in each subshell using arrows to represent their spins. This diagram helps understand the electron distribution in erbium, a lanthanide element.
Erbium's electron configuration involves filling the 4f and 6s orbitals primarily. The orbital diagram depicts each electron’s placement and spin direction, which is crucial for grasping its chemical behavior and magnetic properties.
What Is An Orbital Diagram?
An orbital diagram uses boxes or lines to represent orbitals. Arrows inside these boxes show electrons and their spins. Each orbital can hold up to two electrons with opposite spins. This visual tool clarifies how electrons fill orbitals step-by-step.
Orbital Diagram Of Erbium (er)
Erbium has 68 electrons in total. Its orbital diagram begins with filling lower energy orbitals first. The key orbitals are 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, and finally the 4f subshell.
The 4f subshell of erbium contains 12 electrons. The orbital diagram shows these electrons occupying the seven 4f orbitals with paired and unpaired spins. The 6s orbital holds two electrons with opposite spins.
Importance Of Electron Spins In The Diagram
Electron spins in the orbital diagram follow Hund’s rule. Each orbital in the same subshell gets one electron before pairing begins. Opposite spins in paired electrons reduce repulsion and increase stability. The spin arrangement affects erbium's magnetic and chemical properties.
Valence Electrons
Valence electrons play a key role in defining an element's chemical behavior. These electrons exist in the outermost shell of an atom. They participate in bonding and chemical reactions. Understanding the valence electrons of erbium helps explain its unique properties.
Erbium is a lanthanide element with atomic number 68. It has a complex electron arrangement due to its position in the periodic table. The valence electrons determine how erbium interacts with other elements.
Valence Electron Configuration Of Erbium
Erbium’s electron configuration ends with 4f and 6s orbitals. Specifically, it has the configuration [Xe] 4f12 6s2. The 6s electrons are the outermost and most reactive. The 4f electrons are also considered valence electrons in lanthanides.
Number Of Valence Electrons In Erbium
Erbium has 2 electrons in the 6s shell. It also has 12 electrons in the 4f shell. Both sets contribute to its valence electrons. This gives erbium a total of 14 valence electrons.
Importance Of Erbium’s Valence Electrons
The valence electrons influence erbium’s magnetic and optical properties. They allow erbium to form bonds with other atoms. These electrons also affect the element’s reactivity. This makes erbium useful in lasers and other technologies.
Position In Periodic Table
Erbium holds a unique place in the periodic table. It belongs to the group of elements called lanthanides. These are also known as rare earth metals.
Lanthanides are found in the f-block of the periodic table. Erbium is specifically located in period 6. Its atomic number is 68.
This position tells us about its electron arrangement and chemical properties. Erbium shares similarities with other lanthanides, such as dysprosium and thulium. These neighbors also have electrons filling the 4f orbitals.
Group And Period Of Erbium
Erbium is in period 6, which means it has six electron shells. It is part of the lanthanide series within the f-block. Though lanthanides do not have a traditional group number, they are often placed between groups 3 and 4.
Its placement reflects the filling of the 4f subshell. This is important for its chemical behavior and bonding.
Lanthanide Series And F-block
Erbium is one of the 15 lanthanides. These elements fill their 4f orbitals with electrons. The f-block is located at the bottom of the periodic table to keep the main table compact.
Being in the f-block means erbium has complex electron configurations. This affects its magnetism and optical properties.
Neighboring Elements
Erbium sits between holmium (atomic number 67) and thulium (atomic number 69). These elements share similar properties. They all have electrons in the 4f orbitals.
Understanding erbium’s neighbors helps predict its reactivity and uses in technology.
Electron Configuration And Properties
Erbium is a rare earth element with unique electron arrangements. Its electron configuration affects its chemical behavior and physical traits. Understanding this setup helps explain how erbium interacts in various environments.
The arrangement of electrons in erbium mainly involves the 4f and 6s orbitals. These electrons define many of its magnetic and optical properties. This element's configuration also places it in the lanthanide series on the periodic table.
Basic Electron Configuration Of Erbium
Erbium has the atomic number 68. Its full electron configuration is [Xe] 4f12 6s2. This shows that it fills the 4f subshell with twelve electrons. Two electrons occupy the 6s orbital, which is the outermost shell.
Role Of 4f Electrons In Properties
The 4f electrons in erbium are shielded by outer electrons. This shielding causes unique magnetic properties. These electrons also give erbium its characteristic pink color in compounds. The 4f electrons are less reactive compared to outer electrons.
Valence Electrons And Reactivity
Erbium has two valence electrons in the 6s orbital. These electrons mainly participate in chemical reactions. Erbium tends to lose these electrons to form +3 oxidation states. This behavior is typical for lanthanides and influences erbium’s compounds.
Physical Properties Linked To Electron Configuration
The electron arrangement impacts erbium’s crystal structure. It has a hexagonal close-packed lattice. This structure relates to how electrons fill the atomic orbitals. Erbium shows moderate reactivity with air and water due to its electron setup.
Common Misconceptions
Many learners struggle with understanding the electron configuration of erbium. Some common misconceptions cause confusion about its structure and properties. Clearing these up helps grasp the element better.
Erbium belongs to the lanthanide series. Its electrons fill the 4f and 6s orbitals. Misunderstandings often arise from mixing up these orbitals or their order.
Misunderstanding The Order Of Orbital Filling
Some think 4f orbitals fill after 5d orbitals. This is incorrect for erbium. The 4f orbitals fill first, followed by 6s electrons. The correct configuration is [Xe] 4f12 6s2.
Confusing Valence Electrons With Inner Electrons
Many assume erbium’s valence electrons include 4f electrons. Actually, only the 6s electrons act as valence electrons. The 4f electrons are part of inner shells and less involved in bonding.
Incorrect Use Of Abbreviated Electron Configurations
Abbreviations like [Xe] 4f 6s often miss electron counts. This causes errors in understanding erbium’s full configuration. The full notation is necessary for accuracy and clarity.
Assuming Erbium’s Electron Configuration Is Like Transition Metals
Erbium is a lanthanide, not a typical transition metal. Its electron filling follows f-block rules, not d-block. Treating it like a transition metal leads to wrong assumptions about its chemical behavior.
Credit: www.priyamstudycentre.com
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 belongs to zinc (Zn). It shows filled 3d and 4s orbitals.
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ is manganese (Mn). It has atomic number 25.
Which Element Is This 1s22s22p63s23p64s23d104p5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5 represents the element Bromine (Br). It has atomic number 35.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration can be 2, 8, 8 or 2, 8, 18 depending on the element. Early shells fill as 2, 8, 8; heavier elements extend to 18 in the third shell. Shell capacity follows the 2n² rule.
Conclusion
Understanding the electron configuration of erbium helps explain its chemical behavior. Erbium has electrons filling the 4f and 6s orbitals. This setup places it among the lanthanides in the periodic table. Knowing this configuration reveals why erbium acts the way it does in reactions.
It also shows its role in technology and materials science. Clear knowledge of erbium’s electron arrangement makes studying its properties easier. This information supports learning about other rare earth elements too.



No comments