Electron Configuration of Copernicium
Are you curious about what makes copernicium so unique on the periodic table? Understanding its electron configuration unlocks the secrets of this mysterious superheavy element.
Whether you’re a student, a science enthusiast, or just someone eager to learn, grasping how copernicium’s electrons are arranged will deepen your knowledge of chemistry and the forces that shape matter. You’ll discover the basics of copernicium’s electron setup, why it’s important, and how it influences the element’s rare properties.
Keep reading, and you’ll find out exactly what sets copernicium apart from all the other elements you know.
Credit: www.schoolmykids.com
Basic Properties
Understanding the basic properties of Copernicium helps us grasp its unique nature. This synthetic element has interesting characteristics shaped by its atomic structure and position in the periodic table. These properties are essential for studying its electron configuration and chemical behavior.
Copernicium is a superheavy element, and much about it is based on predictions and limited experiments. Its rarity and instability make it a subject of ongoing research in chemistry and physics.
Atomic Number And Mass
Copernicium has the atomic number 112. This means it has 112 protons in its nucleus. Its most stable isotope has an atomic mass of about 286 atomic mass units. Because it is synthetic, its atoms are produced in laboratories using particle accelerators. The element is highly radioactive and decays quickly into lighter elements.
Discovery And Naming
Scientists first created Copernicium in 1996. It was made by colliding zinc ions with lead atoms. The name Copernicium honors Nicolaus Copernicus, the famous astronomer. This name was officially accepted in 2010. The discovery represents a major step in exploring the limits of the periodic table.
Position In Periodic Table
Copernicium belongs to group 12 of the periodic table. It sits below mercury and cadmium. Its place suggests it shares some properties with these elements. It is part of the 7th period, marking it as a superheavy element. Its electron configuration is predicted to end with 7s², similar to other group 12 metals.
Electron Configuration Details
Understanding the electron configuration of copernicium reveals its atomic structure. This detail helps explain its chemical behavior and placement in the periodic table. Copernicium is a superheavy, synthetic element with atomic number 112. Its electron arrangement is mostly theoretical due to its instability and rarity.
Ground State Configuration
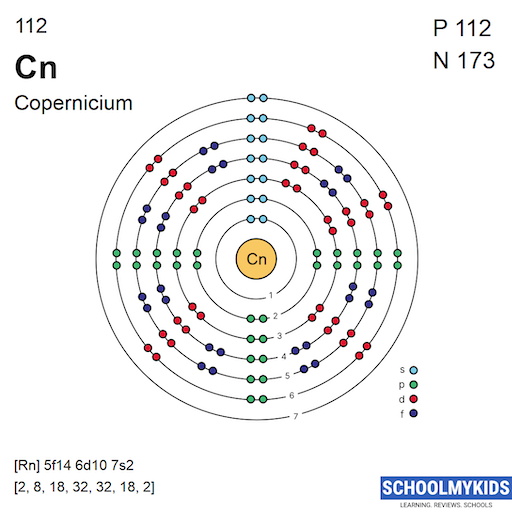
The ground state electron configuration of copernicium is predicted as [Rn] 5f14 6d10 7s2. This means it has a full 5f and 6d subshell, with two electrons in the 7s orbital. These electrons occupy the lowest energy levels in a stable state.
The 7p and 7d orbitals remain empty in the ground state. This configuration aligns copernicium with group 12 elements like mercury.
Noble Gas Core Notation
Using noble gas core notation simplifies copernicium’s electron configuration. It begins with radon (Rn), which represents the first 86 electrons. Then, the remaining electrons fill the 5f, 6d, and 7s orbitals.
This notation looks like: [Rn] 5f14 6d10 7s2. It shows the element’s inner shell electrons are stable and complete.
Valence Electrons
Copernicium has two valence electrons in the 7s orbital. These electrons influence its chemical reactions and bonding. The filled 5f and 6d orbitals do not usually participate in bonding.
Its valence electrons make it chemically similar to zinc, cadmium, and mercury. These two outer electrons are crucial for its properties and reactivity.
Theoretical Predictions
Copernicium is a superheavy element with atomic number 112. Scientists cannot study it easily because it exists only for a short time. Most information about its electron configuration comes from theory and computer models. These models help predict how electrons behave around its nucleus. Understanding these predictions reveals how copernicium compares with lighter elements.
Theoretical studies focus on how copernicium's electrons arrange themselves. This arrangement affects its chemical properties and reactions. Due to its high atomic number, special effects influence its electron cloud. These effects make copernicium different from elements in the same group.
Relativistic Effects
Relativistic effects play a big role in copernicium's electron configuration. Electrons near the nucleus move very fast, close to the speed of light. This changes their mass and energy levels. As a result, the 7s electrons become more stable and closer to the nucleus.
These effects cause the 6d and 7p orbitals to shift in energy. The shifts alter how electrons fill these orbitals. This change affects copernicium’s chemical behavior. It makes copernicium less reactive than expected for its group.
Comparison With Mercury
Copernicium is below mercury in the periodic table. They share some similarities in electron arrangement. Mercury’s electron configuration ends with 5d10 6s2. Copernicium’s predicted configuration ends with 6d10 7s2.
Relativistic effects are stronger in copernicium, changing its properties. Mercury is a liquid metal at room temperature. Copernicium may be a gas or a volatile metal. The changes in electron configuration explain these differences.
Predicted Oxidation States
Copernicium is expected to show oxidation states of +2 and 0. The +2 state comes from losing the two 7s electrons. The 6d electrons are less likely to participate in bonding. This prediction differs from lighter group 12 elements.
Oxidation state +1 may also be possible but is less stable. The unique electron configuration influences these states. Scientists rely on theory to understand copernicium’s chemistry due to limited experiments.
Synthesis Challenges
Creating copernicium is a tough task for scientists. This element does not exist naturally and must be made in labs. The process to produce it faces many hurdles. Its electron configuration is influenced by these challenges. Understanding these difficulties helps us appreciate the work behind this element’s discovery.
Production Methods
Copernicium is made by smashing lighter atoms together. Particle accelerators fire ions at target atoms at high speeds. These collisions sometimes merge nuclei, forming copernicium atoms. The process requires exact conditions and precise timing. It takes a lot of energy to achieve these reactions. Even then, only a few atoms form at a time.
Stability And Half-life
Copernicium atoms are very unstable. They decay quickly into other elements. The half-life of its isotopes is just seconds or less. This short lifespan limits the study of its properties. Scientists must act fast to detect and analyze copernicium. Its instability also affects the electron configuration studies.
Detection Difficulties
Detecting copernicium atoms is extremely hard. The few atoms produced vanish quickly. Instruments must be highly sensitive and fast. Researchers use special detectors to spot decay patterns. These patterns confirm copernicium’s presence. The rarity and fleeting nature of the atoms make detection a big challenge.
Chemical Behavior
Copernicium is a superheavy element with unique chemical traits. Its electron configuration influences how it behaves chemically. Scientists study its properties through theory and experiments with similar elements. This helps predict its nature and reactions.
Understanding copernicium's chemical behavior reveals insights into its place in the periodic table. It shows how it might interact with other elements and form compounds. These aspects are crucial for research in chemistry and material science.
Metallic Or Non-metallic Nature
Copernicium is expected to show metallic properties. It belongs to group 12, like zinc, cadmium, and mercury. Yet, relativistic effects may change its character. It could behave like a noble metal with low reactivity. Its surface may resist oxidation and corrosion.
Reactivity Trends
Reactivity of copernicium is predicted to be very low. It might be less reactive than mercury. This is due to strong relativistic effects on its electrons. These effects stabilize the outer electrons, reducing chemical activity. Copernicium may not easily form ions or bonds.
Potential Compounds
Few compounds of copernicium are known or expected. It may form simple halides such as CnF2 or CnCl2. These compounds would likely be volatile and unstable. Scientists predict copernicium could behave similarly to mercury in bonding. Research continues to confirm these potential compounds.
Credit: stock.adobe.com
Applications And Research
Copernicium, a superheavy element, has unique electron configurations. These configurations influence its chemical behavior and research value. Scientists study copernicium to understand the limits of the periodic table. Research also explores its potential applications in science and technology. This section covers current studies, future prospects, and its role in superheavy element research.
Current Scientific Studies
Researchers focus on copernicium’s electron configuration to predict its properties. Experiments test its stability and chemical reactions. Particle accelerators create tiny amounts of copernicium atoms. Scientists observe how it interacts with other elements. These studies help refine theoretical models. They also improve understanding of superheavy elements’ behavior.
Future Prospects
Future research may reveal new uses for copernicium. Its electron configuration suggests unusual chemical traits. These traits could lead to novel materials or catalysts. Advances in technology might allow larger quantities for testing. More stable isotopes may be discovered or synthesized. This would expand possibilities for practical applications.
Use In Superheavy Element Research
Copernicium acts as a key element in superheavy research. It helps scientists explore the "island of stability." This concept predicts longer-lived superheavy nuclei. Studying copernicium guides synthesis of heavier elements. It also informs nuclear physics and atomic structure theories. Research on copernicium shapes the future of element discovery.

Credit: www.shutterstock.com
Frequently Asked Questions
What Is The Electronic Configuration Of Copernicium?
Copernicium's electron configuration is [Rn] 5f14 6d10 7s2. It follows radon's noble gas core.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 represents the element calcium (Ca). It has 20 electrons.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ is Ruthenium (Ru).
Is Element 119 Theoretically Possible?
Element 119 is theoretically possible as a superheavy element beyond the current periodic table. Scientists predict it could be created in particle accelerators, but it remains highly unstable and difficult to detect due to rapid decay and synthesis challenges. Research continues to explore its existence.
Conclusion
Understanding copernicium’s electron configuration helps explain its chemical behavior. This element’s electrons fill orbitals following the predicted pattern based on its atomic number. Its configuration shows similarities with other group 12 elements. Although copernicium is synthetic and unstable, studying its electrons gives insight into superheavy elements.
This knowledge aids scientists in exploring the periodic table’s limits. Exploring such elements expands our grasp of chemistry and atomic structure.



No comments