Electron Configuration of Cesium
If you want to understand cesium at a deeper level, knowing its electron configuration is key. This simple arrangement of electrons reveals why cesium behaves the way it does in chemical reactions and helps you predict its properties.
Whether you’re a student, a chemistry enthusiast, or just curious, mastering cesium’s electron configuration will give you a clearer picture of this fascinating element. Keep reading, and you’ll discover exactly how cesium’s electrons are arranged and why that matters to your studies or projects.
Atomic Structure Of Cesium
Cesium is a soft, silvery metal found in the alkali metal group. Its atomic structure defines its chemical behavior. Understanding this structure helps explain its reactivity and placement on the periodic table.
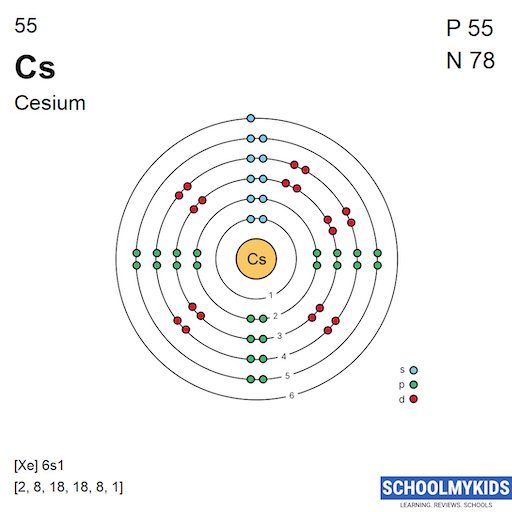
The atomic number of cesium is 55. This means it has 55 protons in its nucleus. To balance the positive charge, it also has 55 electrons orbiting the nucleus. These electrons are arranged in specific shells and subshells.
Electron Shells Of Cesium
Cesium's electrons fill shells in a specific order. The shells are labeled from 1 to 6, moving outward from the nucleus. Each shell holds a certain number of electrons. Cesium’s outermost shell is the sixth one.
The first shell holds 2 electrons, the second 8, the third 18, the fourth 18, and the fifth 8. Finally, the sixth shell contains 1 electron. This single electron in the outer shell is important for cesium’s chemical reactions.
Subshell Arrangement In Cesium
Each electron shell contains subshells named s, p, d, and f. These subshells have different shapes and electron capacities. Cesium's electrons fill these subshells in order.
For cesium, the subshell filling goes up to 6s. The 6s subshell holds the single outer electron. This electron is loosely bound, making cesium highly reactive. The inner subshells, such as 5p and 4d, are fully occupied.
Electron Configuration Notation
The full electron configuration of cesium is written as: [Xe] 6s1. The [Xe] represents the electron configuration of xenon, the noble gas before cesium. This shorthand shows that cesium’s electrons fill all shells up to xenon’s configuration plus one more in 6s.
This configuration explains why cesium is in group 1 of the periodic table. The single electron in the 6s subshell is the valence electron. It participates in bonding and gives cesium its characteristic properties.
Credit: www.schoolmykids.com
Cesium’s Electron Orbitals
Cesium is a chemical element with atomic number 55. Its electron orbitals show how electrons are arranged around its nucleus. This arrangement helps explain many of cesium's physical and chemical properties.
Understanding cesium's electron orbitals can clarify its behavior in reactions. It also reveals why cesium is highly reactive and where it fits in the periodic table.
Electron Shells And Energy Levels
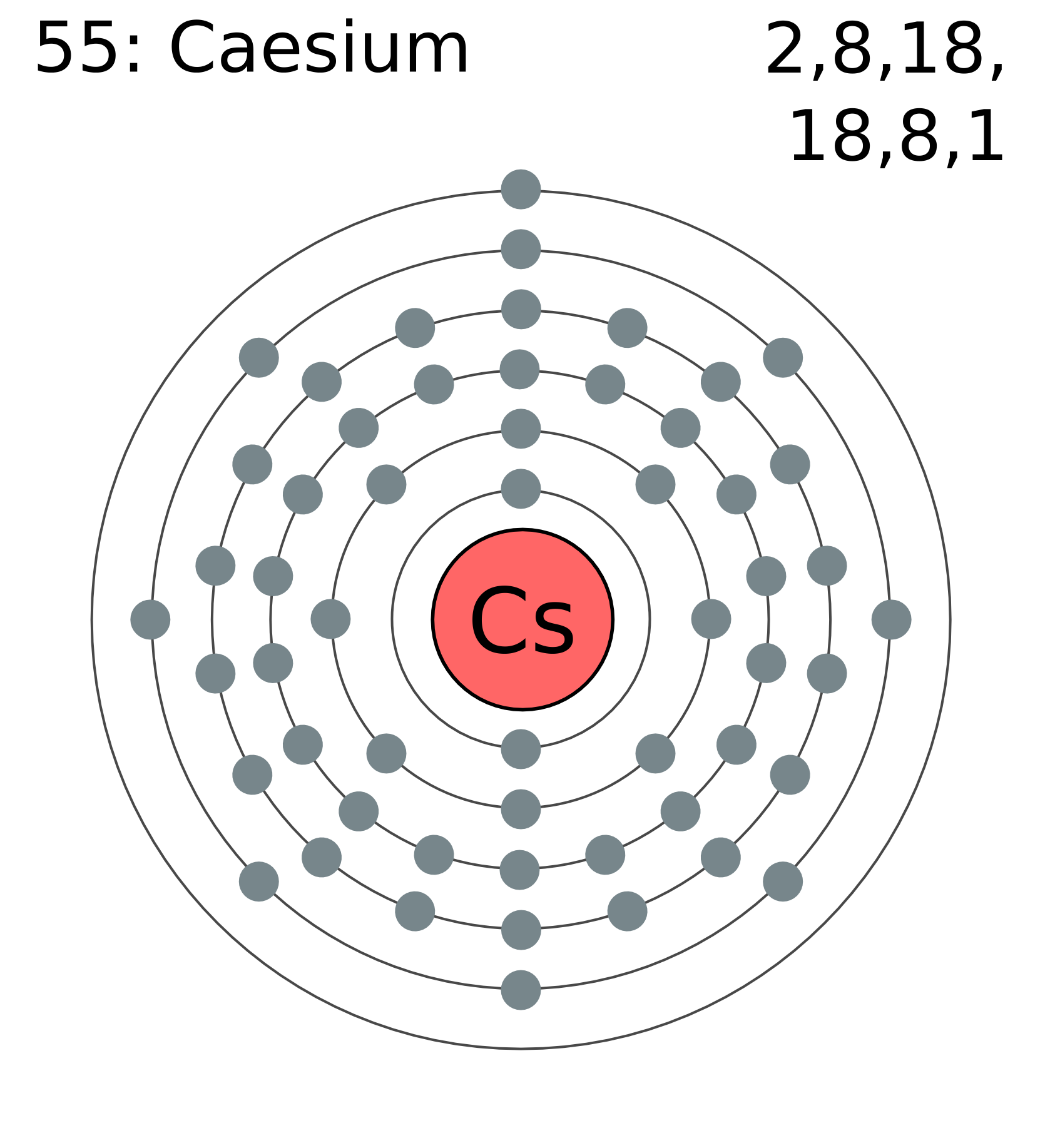
Cesium has electrons in six main shells. Each shell holds electrons at different energy levels. The outermost shell is the sixth shell, which contains only one electron. This single electron is important for cesium's reactivity.
Electron Configuration Notation
The full electron configuration of cesium is written as [Xe] 6s1. The symbol [Xe] represents the electron configuration of xenon, a noble gas before cesium. The "6s1" means one electron is in the 6s orbital.
Valence Electron And Reactivity
The lone electron in the 6s orbital is called the valence electron. This electron is loosely held, making cesium very reactive. Cesium easily loses this electron to form positive ions.
Full Electron Configuration
The full electron configuration shows how electrons fill the orbitals in an atom. It gives a detailed map of electron arrangement for cesium.
Cesium has 55 electrons. These electrons occupy different energy levels and sublevels. The full configuration helps us understand cesium’s chemical behavior.
Electron Configuration Notation
Electron configuration uses numbers and letters. Numbers indicate energy levels. Letters show sublevels: s, p, d, and f. Superscripts give the number of electrons in each sublevel.
Cesium’s Full Electron Configuration
Cesium’s electron configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶ 6s¹. This shows all electrons from the first to the sixth shell.
Significance Of The 6s¹ Electron
The last electron in cesium is in the 6s sublevel. This electron is loosely held and easily lost in reactions. It defines cesium as an alkali metal.
Abbreviated Configuration Using Noble Gas
The abbreviated electron configuration uses the nearest noble gas to shorten the full configuration. This method makes it easier to write and understand the electron arrangement of elements. For cesium, the noble gas xenon is used as a reference point. It represents the inner, fully filled electron shells.
By using the noble gas symbol in brackets, we save space and focus only on the electrons added beyond that noble gas. This approach highlights valence electrons, important for chemical reactions. It also helps students and chemists quickly identify the outer shell electrons of cesium.
What Is The Noble Gas Core For Cesium?
The noble gas core for cesium is xenon, written as [Xe]. Xenon has 54 electrons filling the inner shells. Cesium has 55 electrons total. Using [Xe] covers the first 54 electrons.
Abbreviated Electron Configuration Of Cesium
After [Xe], cesium has one electron in the 6s orbital. This is written as [Xe] 6s1. It shows cesium’s valence electron clearly. This format is simple and easy to remember.
Why Use Abbreviated Configuration?
This method reduces complexity in writing electron arrangements. It makes studying elements faster. Chemists can focus on the outer electrons that affect bonding. It also helps avoid mistakes in counting many electrons.
Valence Electrons In Cesium
Cesium has one valence electron in its 6s orbital, following the electron configuration [Xe] 6s¹. This single outer electron makes cesium highly reactive and part of the alkali metals group. Understanding this helps explain its chemical behavior and bonding.
Understanding Valence Electrons In Cesium
Cesium has a single valence electron. This electron is in the 6s orbital. Valence electrons determine how an element reacts chemically. Cesium belongs to Group 1 of the periodic table. All elements in this group have one valence electron. This makes cesium highly reactive and eager to lose that electron.
Electron Configuration And Valence Electron Location
The full electron configuration of cesium is [Xe] 6s1. The [Xe] represents the core electrons, matching the noble gas xenon. The 6s1 shows the single valence electron in the sixth shell. This electron is far from the nucleus and loosely held. This explains cesium’s tendency to form positive ions by losing this electron.
Significance Of Cesium’s Valence Electron
The single valence electron plays a key role in cesium’s chemistry. It allows cesium to form Cs+ ions easily. Losing the 6s electron gives cesium a stable electron arrangement. This stability is similar to the noble gas xenon. The ease of losing this electron makes cesium a strong reducing agent.
Cesium Ion Electron Configuration
The cesium ion forms when cesium loses one electron. This loss changes its electron arrangement. Understanding this ion's electron configuration helps explain its chemical behavior.
Cesium atoms have one electron in their outermost shell. When cesium becomes a positive ion (Cs⁺), it loses this outer electron. This loss creates a more stable electron setup.
Cesium Ion Electron Configuration Explained
Neutral cesium has the configuration: [Xe] 6s1. Removing one electron removes the 6s electron. The cesium ion’s configuration becomes [Xe]. This means it matches the noble gas xenon.
Why Cesium Ion Has A Stable Configuration
Cesium ion’s electron setup is stable. It has a full set of electrons like xenon. This full set means lower energy and less reactivity. The ion achieves a noble gas structure.
Impact On Chemical Properties
The electron loss makes cesium highly reactive. It easily forms compounds with other elements. The Cs⁺ ion is common in salts and other substances. Its electron configuration explains this common ion state.
Electron Configuration And Reactivity
Understanding the electron configuration of cesium reveals why it behaves uniquely in reactions. Cesium has a single electron in its outermost shell. This electron is loosely held and easily lost during chemical reactions.
This simple electron setup explains cesium’s high reactivity. It wants to lose its outer electron to achieve a stable state. This behavior defines its role as an alkali metal in the periodic table.
Electron Configuration Of Cesium
Cesium’s electron configuration is written as [Xe] 6s1. This means it has the same electron arrangement as xenon, plus one electron in the 6s orbital. The 6s electron is the key to its chemical properties.
Effect Of Electron Configuration On Reactivity
The single electron in the 6s orbital is far from the nucleus. This makes it easier to remove than electrons in inner shells. Because of this, cesium reacts quickly with other elements, especially nonmetals.
Cesium Ion Formation
Cesium often forms Cs+ ions by losing its 6s electron. This ion has a stable electron configuration like xenon. The ease of ion formation makes cesium highly reactive and important in many chemical reactions.

Credit: www.youtube.com
Comparison With Other Alkali Metals
Cesium shares many similarities with other alkali metals in its electron configuration. These elements all have one electron in their outermost s orbital. This single valence electron defines their chemical behavior and reactivity.
Understanding how cesium compares helps clarify trends within the alkali metal group. It also highlights differences caused by increasing atomic number and electron shells.
Electron Configuration Patterns In Alkali Metals
All alkali metals end their electron configuration with an ns1 orbital. For example, lithium ends with 2s1, sodium with 3s1, and potassium with 4s1. Cesium, being heavier, ends with 6s1. This pattern shows the gradual filling of electron shells as you move down the group.
Cesium’s Electron Configuration Compared To Sodium And Potassium
Sodium’s configuration is [Ne] 3s1, potassium’s is [Ar] 4s1. Cesium’s is [Xe] 6s1. Cesium has more inner electron shells, which shield its outer electron more. This shielding affects its size and reactivity compared to sodium and potassium.
Impact Of Electron Configuration On Reactivity
Cesium’s single electron in the 6s orbital is farther from the nucleus than sodium’s 3s electron. This distance lowers the attraction between the nucleus and the valence electron. Cesium loses its outer electron more easily, making it more reactive than lighter alkali metals.
Applications Influenced By Electron Setup
The electron configuration of cesium shapes its unique properties. These properties influence many practical uses. Understanding cesium’s electron setup helps explain why it behaves as it does in various applications.
Cesium has one electron in its outermost shell. This makes it highly reactive and useful in specific fields. Its electron setup drives its role in technology and science.
Atomic Clocks And Precision Timing
Cesium’s electron setup allows it to emit precise energy signals. These signals form the basis of atomic clocks. Atomic clocks keep time with incredible accuracy. This accuracy is critical for GPS systems and telecommunications.
Photoelectric Cells And Light Sensors
Cesium’s single outer electron easily releases under light. This property makes it perfect for photoelectric cells. These cells convert light into electrical signals. Cesium-based sensors are used in cameras and solar panels.
Medical And Scientific Instruments
Cesium’s electron configuration helps create specialized medical devices. It is used in radiation therapy for cancer treatment. Cesium isotopes also assist in scientific research and imaging.
Industrial Applications And Catalysts
Cesium’s reactivity, due to its electron setup, aids in chemical reactions. It acts as a catalyst in some industrial processes. This speeds up reactions and improves efficiency in manufacturing.
Credit: commons.wikimedia.org
Common Misconceptions About Cesium Electrons
Cesium’s electron setup often causes confusion. Many misunderstandings exist about its electron arrangement. Clearing these up helps grasp cesium’s chemical behavior better.
Common Confusion: Valence Electron Count
People often think cesium has many valence electrons. Actually, cesium has only one valence electron. This single electron in the 6s orbital makes cesium highly reactive.
Misunderstanding The Role Of Inner Electrons
Some believe all electrons affect cesium’s reactions equally. Inner electrons mainly shield the nucleus. They do not usually participate in chemical bonding or reactions.
Incorrect Assumptions About Electron Shell Filling
It is a mistake to think cesium’s outer shells fill like lighter elements. Cesium’s electron configuration follows a unique pattern due to its high atomic number. The 6s subshell fills after the 5p and 5d subshells.
Confusing Cesium’s Ion Formation
Many assume cesium forms complex ions with multiple charges. Cesium typically loses only one electron, forming a Cs+ ion. This simplicity is key to its chemistry.
Frequently Asked Questions
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element Zinc (Zn), atomic number 30.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 corresponds to the element cobalt (Co).
Which Element Is This 1s22s22p63s23p64s23d104p5?
This electron configuration 1s²2s²2p⁶3s²3p⁶4s²3d¹⁰4p⁵ corresponds to the element Bromine (Br).
Which Element Has The Electron Configuration 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁵ belongs to the element manganese (Mn).
Conclusion
Cesium’s electron configuration reveals its place in the periodic table. It has one electron in the 6s orbital beyond the xenon core. This simple arrangement explains cesium’s chemical behavior and reactivity. Knowing this helps understand how cesium forms ions and bonds.
The 6s¹ electron is easily lost, making cesium highly reactive. This knowledge is useful for students and chemistry enthusiasts alike. Understanding cesium’s electron setup builds a foundation for learning other elements. Clear and simple, cesium’s electron configuration is key to its chemistry.



No comments