Electron Configuration of Bohrium
Are you curious about the mysterious element bohrium and how its electrons arrange themselves? Understanding the electron configuration of bohrium is key to unlocking its chemical behavior and place in the periodic table.
Whether you’re a student, a science enthusiast, or just someone fascinated by the building blocks of matter, knowing bohrium’s electron setup will give you a clearer picture of this rare, synthetic element. You’ll discover exactly how bohrium’s electrons are organized, why this matters, and how it connects to other elements you already know.
Keep reading to master the electron configuration of bohrium and deepen your grasp of chemistry’s hidden patterns.

Credit: commons.wikimedia.org
Bohrium Basics
Bohrium is a synthetic element with unique features. It belongs to the group of heavy elements created in labs. Studying its electron configuration helps us understand its nature.
This section covers the basics of bohrium. You will learn about its atomic number, symbol, position in the periodic table, and metallic properties.
Atomic Number And Symbol
Bohrium has the atomic number 107. Its chemical symbol is Bh. This number tells the count of protons in bohrium’s nucleus. It also equals the number of electrons in a neutral atom.
Position In The Periodic Table
Bohrium is placed in period 7 of the periodic table. It belongs to group 7, which includes other transition metals. Its position shows it is a d-block element. This means its electrons fill the d sublevel.
Metallic Properties
Bohrium is expected to have metallic properties like other heavy transition metals. It likely conducts electricity and heat well. Its appearance might be shiny and solid under normal conditions. These traits help categorize bohrium as a metal.

Credit: pilgaardelements.com
Electron Shells And Energy Levels
Bohrium, a synthetic element, has a complex electron arrangement. Understanding its electron shells and energy levels helps explain its chemical behavior. Electrons occupy spaces called shells around the nucleus. Each shell holds a specific number of electrons based on energy rules.
The arrangement of these electrons follows fixed principles. These rules determine how electrons fill shells and subshells in order. This structure influences bohrium’s properties and reactions.
Electron Shell Capacity
Each electron shell has a maximum number of electrons it can hold. This number is given by the formula 2n², where n is the shell number. For example, the first shell holds 2 electrons, the second holds 8, and the third holds 18. Bohrium’s electrons fill shells following these limits, starting from the lowest energy level.
Energy Level Principles
Electrons fill the lowest energy levels first. This is known as the Aufbau principle. Electrons also follow Pauli’s exclusion rule, meaning no two electrons share the same quantum state. Hund’s rule says electrons fill orbitals singly before pairing. These principles guide the order of electron filling in bohrium.
Valence Electrons Role
Valence electrons are the outermost electrons in an atom. They determine how bohrium reacts with other elements. These electrons participate in chemical bonding and influence the element’s stability. Bohrium’s valence electrons reside in the 7th shell, affecting its placement in the periodic table.
Bohrium Electron Configuration
Bohrium is a synthetic element with atomic number 107. Its electron configuration helps us understand its chemical properties. Electrons fill orbitals in a specific order based on energy levels. Bohrium’s configuration reflects its place in the periodic table as a transition metal. This section explains its electron arrangement clearly and simply.
Long-form Notation
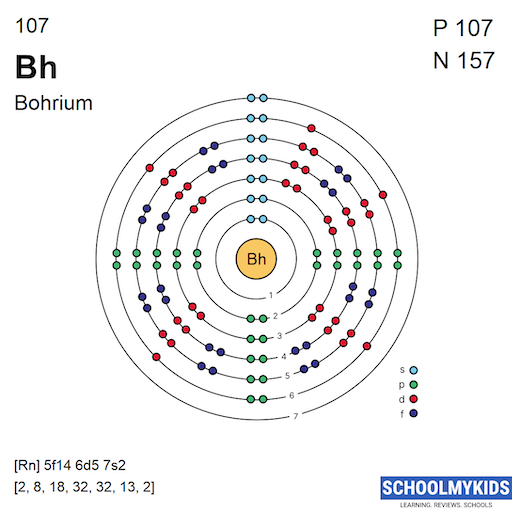
The full electron configuration of bohrium is: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 4f¹⁴ 5s² 5p⁶ 5d⁵ 6s² 6p⁶ 6d⁵ 7s².
This notation shows all occupied orbitals in order. It includes the inner shells and the outermost electrons. The 6d and 7s electrons are important for bohrium's chemical behavior.
Noble Gas Shortcut
Using a noble gas to shorten the configuration saves space. For bohrium, the shorthand is [Rn] 5f¹⁴ 6d⁵ 7s².
Here, [Rn] stands for radon, which covers the first 86 electrons. The rest shows only bohrium’s valence and near-valence electrons. This form is easier to write and remember.
Comparison With Lighter Homologues
Bohrium is in group 7, below manganese and technetium. Its electron configuration is similar but has more filled inner shells.
Manganese ends with 3d⁵ 4s². Technetium ends with 4d⁵ 5s². Bohrium’s 6d⁵ 7s² mirrors these patterns at a higher level.
This pattern helps predict bohrium’s chemical traits by comparing it with lighter elements.
Credit: www.schoolmykids.com
Quantum Numbers And Orbitals
Understanding the electron configuration of bohrium requires knowledge about quantum numbers and orbitals. Quantum numbers describe the position and energy of electrons in an atom. Orbitals are regions where electrons are most likely to be found. These concepts help explain bohrium's chemical and physical properties.
Principal Quantum Number
The principal quantum number, symbolized as n, shows the main energy level of an electron. It is a positive integer starting from 1. Higher n values mean electrons are farther from the nucleus. For bohrium, electrons fill up to the 7th energy level, reflecting its position in the periodic table.
Azimuthal Quantum Number
The azimuthal quantum number, l, defines the shape of the orbital. It ranges from 0 to n−1. Each number corresponds to a type of orbital: 0 for s, 1 for p, 2 for d, and 3 for f. Bohrium has electrons in s, d, and f orbitals, with d orbitals playing a key role in its chemistry.
Magnetic And Spin Quantum Numbers
The magnetic quantum number, ml, indicates the orientation of the orbital in space. It ranges from −l to +l. The spin quantum number, ms, shows the electron’s spin direction. It can be either +½ or −½. Together, these quantum numbers define the exact state of an electron in bohrium.
Filling Order And Exceptions
The electron configuration of bohrium follows a specific filling order of atomic orbitals. This order is guided by fundamental principles in quantum chemistry. These rules explain how electrons populate orbitals and why some exceptions occur. Understanding these concepts helps clarify bohrium’s complex electron arrangement.
Aufbau Principle
The Aufbau principle states electrons fill orbitals starting with the lowest energy. Electrons occupy the 1s orbital first, then move to higher levels. For bohrium, electrons fill inner shells before outer ones. This order minimizes the atom’s total energy. The principle helps predict the expected electron configuration.
Pauli Exclusion Principle
The Pauli exclusion principle limits electrons in an orbital. Each orbital can hold two electrons with opposite spins only. This rule prevents electrons from having identical quantum states. In bohrium, this principle ensures proper filling of orbitals. It controls electron pairing within each orbital.
Hund's Rule
Hund’s rule states electrons fill degenerate orbitals singly before pairing. This reduces electron repulsion and stabilizes the atom. In bohrium, electrons spread out in orbitals of the same energy first. Only after all orbitals have one electron do pairs form. This rule explains the distribution in d and f subshells.
Experimental Challenges
Studying bohrium’s electron configuration faces major experimental challenges. This element is synthetic and highly unstable. Scientists must overcome difficulties in production and measurement. These obstacles limit detailed understanding of its atomic structure.
Synthesis Of Bohrium
Bohrium is created in particle accelerators. Scientists collide lighter nuclei at high speeds. Only a few atoms form in each reaction. This low yield makes research slow and difficult. Producing enough bohrium for experiments remains a key challenge.
Short Half-life Impact
Bohrium atoms decay very quickly. Their half-life lasts seconds to minutes. Experiments must occur rapidly after creation. This short lifespan hinders detailed study. Researchers must use fast detection tools to capture data.
Electron Configuration Verification
Directly observing bohrium’s electron arrangement is nearly impossible. Researchers rely on theoretical models and indirect methods. Comparing chemical behavior with lighter elements helps. Advanced spectroscopy techniques assist but have limits. These factors complicate confirming bohrium’s exact electron configuration.
Chemical Behavior And Electron Configuration
The chemical behavior of bohrium is closely linked to its electron configuration. This element sits in the seventh period and group 7 of the periodic table. Its electron arrangement helps predict how it reacts with other substances. Understanding its electron setup sheds light on its properties and possible uses.
Bohrium’s electron configuration influences its valence electrons. These outer electrons determine how bohrium bonds and interacts chemically. Studying bohrium alongside its group 7 neighbors reveals patterns in behavior and reactivity.
Predicted Reactivity
Bohrium is expected to behave like other group 7 elements, such as manganese and rhenium. It likely forms compounds with oxidation states of +7 and +5. These states suggest strong oxidizing abilities. Reactivity may be slower due to relativistic effects on its electrons. Still, it should form stable oxides and halides under certain conditions.
Valence Electron Influence
Bohrium’s valence electrons mainly occupy the 7s and 6d orbitals. These electrons are key to its chemical reactions. Their arrangement controls how bohrium shares or loses electrons. The outer electrons affect bond strength and type. This makes bohrium similar to other transition metals in group 7.
Comparison With Other Group 7 Elements
Bohrium’s electron configuration resembles that of rhenium and manganese. Like them, it has five d-electrons and two s-electrons in its outer shells. This similarity suggests comparable chemical properties. Differences arise due to bohrium’s heavier atomic mass and relativistic electron effects. These factors may alter bond lengths and reactivity compared to lighter group members.
Frequently Asked Questions
What Is The Electron Configuration Of Bohrium?
The electron configuration of bohrium (Bh, atomic number 107) is [Rn] 5f¹⁴ 6d⁵ 7s². It follows the Aufbau principle for transition metals.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2 3d10?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 corresponds to the element zinc (Zn). It has 30 electrons filling shells and subshells.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration is typically 2, 8, 8 for lighter elements. For heavier elements, it can be 2, 8, 18. Shell capacity depends on the element’s atomic number and energy levels.
How To Write Bohr Electron Configuration?
Write Bohr electron configuration by placing electrons in shells around the nucleus. Use the 2n² rule for maximum electrons per shell. Fill inner shells first, then outer shells. Count total electrons equal to the element’s atomic number. Show electrons as dots or numbers per shell.
Conclusion
Understanding bohrium’s electron configuration reveals its place in the periodic table. It follows the typical filling of d-block elements. Electrons fill orbitals in a specific order to maintain stability. This arrangement affects bohrium’s chemical properties and behavior. Though rare and synthetic, studying its configuration helps scientists predict reactions.
Such knowledge supports research in chemistry and physics. Bohrium’s electron setup is complex but follows known principles. Exploring these patterns enriches our grasp of heavy elements. The study of bohrium connects atomic theory with experimental science.



No comments