Electron Configuration of Astatine
Have you ever wondered what makes astatine unique on the periodic table? Understanding the electron configuration of astatine can unlock the secrets behind its chemical behavior and properties.
By diving into how its electrons are arranged, you’ll gain clearer insights into why astatine acts the way it does. If you want to grasp this mysterious element better and see how its structure influences its role in science, keep reading—you’re about to discover something truly fascinating.
Basic Properties Of Astatine
Astatine is a rare and radioactive element. It belongs to the halogen group on the periodic table. It shares traits with iodine and other halogens but is less stable. Understanding its basic properties helps explain its behavior and uses.
Astatine’s atomic number is 85, meaning it has 85 protons. It is very heavy compared to other halogens. Its radioactivity makes it difficult to study in detail. Despite this, scientists have learned key facts about its structure and properties.
Atomic Structure
Astatine has 85 electrons arranged in energy levels. The electrons fill orbitals following the electron configuration rules. Its outermost electrons determine its chemical reactions. This setup influences how it bonds with other elements.
Physical Characteristics
Astatine is a solid at room temperature. It has a metallic look unlike other halogens. Its density is higher than iodine. It can form different allotropes, but these are hard to observe. The element is rare and found in tiny amounts in nature.
Chemical Behavior
Astatine reacts similarly to iodine but is less reactive. It forms compounds with metals and non-metals. Its radioactivity affects how it interacts in chemical reactions. This limits its practical uses and study opportunities.
Credit: www.daviddarling.info
Position In The Periodic Table
Astatine holds a unique place in the periodic table. It belongs to the halogen group, which includes elements like fluorine and iodine. This group is known for its reactive nonmetals. Astatine is the heaviest member of this group.
It is found in period 6, meaning it has six electron shells. Its position helps explain its chemical behavior and physical properties. The element's atomic number is 85, showing it has 85 protons in its nucleus.
Group And Period
Astatine is in group 17, the halogen family. This group contains elements that are highly reactive. It is placed in period 6, indicating it has electrons in six energy levels.
Block In The Periodic Table
Astatine is part of the p-block. This block includes elements whose last electron enters a p orbital. The p-block elements often have varied properties, from metals to nonmetals.
Metallic Or Nonmetallic Nature
Astatine shows mixed properties. It behaves like a metalloid. It is less reactive than lighter halogens but more than metals. Its position between metals and nonmetals reflects this balance.
Atomic Structure Overview
Understanding the atomic structure of astatine helps us learn about its properties. Astatine is a rare element with unique features in its atomic makeup. Its atomic structure gives clues about how it behaves in chemical reactions.
The atomic structure consists of protons, neutrons, and electrons. These particles arrange in specific ways to form the element’s identity. The electrons follow certain patterns called electron configurations.
Electron Shells And Energy Levels
Astatine has electrons arranged in shells around its nucleus. Each shell holds electrons at different energy levels. The closer a shell is to the nucleus, the lower its energy.
Astatine’s electrons fill these shells step by step. The outermost shell determines how the element reacts with others.
Number Of Protons And Neutrons
Astatine has 85 protons in its nucleus. Protons give the element its positive charge. Neutrons are neutral and add mass to the nucleus.
The number of neutrons can vary, creating different isotopes of astatine. These isotopes have similar chemical properties but different stability.
Electron Configuration Pattern
The electron configuration of astatine shows how electrons fill orbitals. It follows a specific order based on energy rules. This pattern helps predict astatine’s chemical behavior.
Astatine’s configuration ends with electrons in the 6p orbital. This placement explains its position in the halogen group of the periodic table.
Credit: www.schoolmykids.com
Electron Shell Arrangement
Astatine’s electron configuration shows how its electrons fill different shells. Electrons arrange from inner to outer shells, following specific energy levels. This pattern helps explain astatine’s chemical behavior and properties.
Astatine is a rare element with a complex electron shell structure. Understanding its electron arrangement helps explain its chemical behavior. The electrons orbit the nucleus in different shells, each with a set capacity.
These shells fill in a specific order. The shells closer to the nucleus fill first, then the outer shells. This order affects how astatine interacts with other elements.
Principal Energy Levels In Astatine
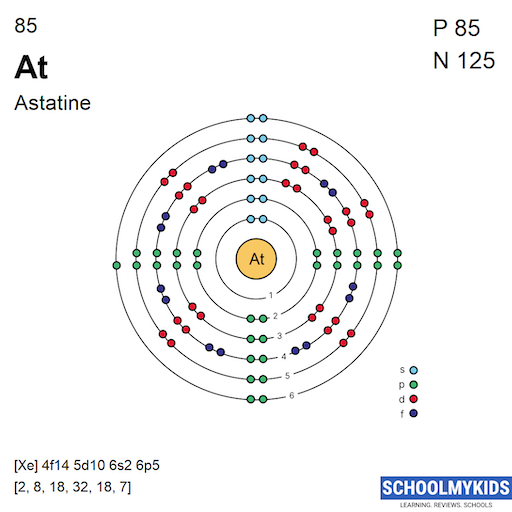
Astatine has five principal energy levels occupied by electrons. These levels are called shells and are labeled 1 to 6, skipping the empty sixth shell for this element. Each shell holds a specific number of electrons.
The first shell holds 2 electrons, the second holds 8, the third 18, the fourth 18, and the fifth shell holds 7 electrons in astatine.
Electron Distribution Across Subshells
Each shell contains subshells named s, p, d, and f. These subshells have different shapes and energy. Electrons fill these subshells in a fixed order based on energy levels.
In astatine, the filling order starts with 1s, then 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, and finally 6s and 4f subshells. This pattern defines the electron configuration precisely.
Valence Shell Configuration Of Astatine
The valence shell holds the electrons involved in chemical reactions. For astatine, the valence shell is the fifth shell. It contains 7 electrons in the 5p subshell.
These valence electrons determine astatine’s properties and how it bonds with other elements. The 7 electrons make astatine reactive, similar to other halogens.
Orbital Filling Sequence
Astatine’s electron configuration follows the orbital filling sequence, filling orbitals from lower to higher energy. Electrons occupy s, p, d, and f orbitals in a specific order. This sequence helps explain astatine’s chemical behavior and properties.
Understanding Orbital Filling Sequence
The orbital filling sequence shows how electrons fill atomic orbitals. Electrons fill orbitals from lower to higher energy levels. This order follows the Aufbau principle, which helps predict electron arrangements.
For astatine, the filling sequence determines its chemical behavior. Electrons fill orbitals in a specific pattern. This pattern affects astatine’s properties and reactivity.
Order Of Orbital Filling In Astatine
Astatine has 85 electrons. They fill orbitals in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p.
Each orbital holds a certain number of electrons. The s-orbital holds 2, p-orbitals hold 6, d-orbitals hold 10, and f-orbitals hold 14 electrons.
Electron Configuration Of Astatine Based On Orbitals
The configuration for astatine ends in the 6p orbital. It is written as [Xe] 4f14 5d10 6s2 6p5. This means astatine’s outer electrons fill the 6p orbital partially.
This partial filling in the 6p orbital is key to astatine’s chemical traits. It behaves like other halogens because of this electron arrangement.

Credit: www.youtube.com
Ground State Configuration
The ground state configuration of an element tells us how its electrons are arranged. This arrangement shows the lowest energy state of the atom. Electrons fill the orbitals in a specific order. This order follows simple rules that help predict chemical behavior.
For astatine, a rare and heavy element, understanding its ground state electron configuration is important. It helps explain its place in the periodic table and its chemical properties.
Atomic Number And Electron Count
Astatine has an atomic number of 85. This means it has 85 electrons. These electrons fill the atom’s orbitals starting from the lowest energy level.
Order Of Orbital Filling
Electrons fill orbitals in this order: s, p, d, then f. Each orbital type holds a certain number of electrons. The s orbital holds 2, p holds 6, d holds 10, and f holds 14.
Ground State Configuration Of Astatine
The electron configuration of astatine is: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 4f¹⁴ 5s² 5p⁶ 5d¹⁰ 6s² 6p⁵. This shows all electrons in their lowest energy orbitals.
Excited State Possibilities
The excited state of astatine occurs when its electrons absorb energy and move to higher orbitals. This change affects its chemical behavior and reactivity. Understanding these possibilities helps explain how astatine interacts in different environments.
Excited states are temporary. Electrons do not stay in higher orbitals for long. They quickly return to the ground state, releasing energy as light or heat. These transitions are important in fields like spectroscopy and nuclear medicine.
Electron Promotion In Astatine
In the excited state, an electron from the 6p orbital may jump to the 7s or 6d orbital. This promotion changes the atom's energy and shape. It also affects how astatine bonds with other elements.
Impact On Chemical Reactivity
Excited electrons create new bonding opportunities. Astatine can form unusual compounds under these conditions. Its reactivity increases, making it more likely to participate in chemical reactions.
Energy Levels And Transitions
Energy differences between orbitals determine excitation possibilities. The closer the orbitals, the easier the electron jumps. These transitions emit specific light wavelengths, useful for identifying astatine.
Applications Of Excited States
Excited states of astatine have uses in medicine and research. They help in imaging and cancer treatment. Studying these states also aids in understanding heavy element behavior.
Impact On Chemical Behavior
The electron configuration of astatine plays a key role in its chemical behavior. It defines how astatine atoms interact with other elements. The arrangement of electrons affects reactivity and bonding patterns.
Astatine has a unique electron setup among halogens. This influences its tendency to form compounds and its stability in reactions. Understanding this helps predict its behavior in different environments.
Valence Electrons And Reactivity
Astatine's valence electrons are in the 6p orbital. These electrons determine how easily astatine can gain, lose, or share electrons. With seven valence electrons, it tends to gain one to complete its shell.
This makes astatine reactive, but less so than lighter halogens like chlorine. The larger atomic size and lower electronegativity reduce its reactivity.
Bonding Characteristics
Astatine can form covalent bonds by sharing electrons. Its electron configuration allows bonds with metals and non-metals. These bonds are often weaker than those of other halogens.
The presence of electrons in inner shells also affects bond strength. It can form single or double bonds in various compounds, but with lower bond energy.
Oxidation States And Chemical Forms
The electron arrangement gives astatine multiple possible oxidation states. Common states include -1, +1, +3, +5, and +7. This variety allows it to form many chemical species.
Its ability to change oxidation states influences the types of reactions it undergoes. This versatility is important in its chemical and environmental behavior.
Comparison With Other Halogens
The electron configuration of astatine differs from other halogens in subtle ways. These differences affect its chemical behavior and physical properties. Comparing astatine with other halogens helps us understand its unique place in the periodic table.
Electron Configuration Patterns
All halogens have seven electrons in their outermost shell. This makes them highly reactive. Fluorine, chlorine, bromine, and iodine share similar electron configurations. Astatine, being heavier, has more inner electrons. Its outer electrons occupy the 6p orbital, unlike lighter halogens that use the 2p, 3p, 4p, or 5p orbitals.
Impact On Reactivity
Reactivity decreases down the group from fluorine to astatine. Astatine’s larger size and extra inner electrons reduce its attraction for electrons. This lowers its reactivity compared to chlorine or bromine. Its electron configuration results in weaker bonds with other elements.
Physical And Chemical Properties
Astatine’s electron setup influences its state and color. It is a rare, radioactive halogen with metallic characteristics. Other halogens are more common and non-metallic. The electron arrangement causes astatine to have higher melting and boiling points than lighter halogens.
Applications Influenced By Electron Setup
The electron configuration of astatine plays a key role in its applications. This setup shows how electrons are arranged around the atom’s nucleus. These arrangements affect how astatine reacts and bonds with other elements.
Understanding the electron setup helps explain astatine’s behavior in different fields. It guides scientists in using this rare element for specific purposes. The unique electron configuration influences astatine’s chemical properties and uses.
Radioactive Medical Uses
Astatine’s electron configuration makes it highly unstable and radioactive. This trait suits it for targeted cancer treatments. Doctors use astatine isotopes to destroy cancer cells with minimal harm to healthy cells. The electron arrangement helps control its decay and radiation emission.
Chemical Research And Bonding
The electron setup determines how astatine forms bonds. It can combine with metals and nonmetals in unique ways. Researchers study these bonds to understand astatine’s chemistry better. These studies help in developing new compounds with special properties.
Material Science Applications
Astatine’s electron arrangement influences its interaction with materials. It can alter the electrical properties of some substances. Scientists explore these effects for potential use in electronics. The electron configuration guides how astatine modifies material behavior.
Frequently Asked Questions
What Is The Electron Configuration Of Astatine?
Astatine's electron configuration is [Xe] 4f14 5d10 6s2 6p5. It shows 85 electrons arranged in shells and subshells. This configuration reveals astatine’s chemical behavior and position in the halogen group.
Why Is Astatine’s Electron Configuration Important?
Understanding astatine’s electron configuration helps predict its chemical properties. It explains reactivity and bonding patterns with other elements. This knowledge is crucial for research in chemistry and material science.
How Does Astatine’s Configuration Compare To Iodine?
Astatine’s electron configuration is similar to iodine’s, with one more electron. Both belong to the halogen group and have outer shells ending in p5. This similarity explains their related chemical characteristics.
Where Does Astatine Fit In The Periodic Table?
Astatine is in group 17, period 6 of the periodic table. Its electron configuration confirms its placement among halogens. This position helps understand its physical and chemical traits.
Conclusion
Astatine’s electron configuration reveals its place in the periodic table. It shares traits with halogens like iodine and bromine. Understanding its electrons helps explain its chemical behavior. This knowledge supports studies in chemistry and physics. Simple patterns in electron arrangement make elements easier to learn.
Astatine remains rare and less studied but important. Studying its electrons connects to real-world science and research. Keep exploring to learn more about elements and their secrets.



No comments