Electron Configuration of Silver
Have you ever wondered what makes silver so unique on the periodic table? Understanding the electron configuration of silver can unlock the secrets behind its special properties, like its stunning shine and excellent conductivity.
You’ll discover exactly how silver’s electrons are arranged, why its configuration might surprise you, and how this influences its behavior in chemistry and everyday life. If you want to grasp the core of silver’s atomic structure and why it stands out among other elements, keep reading—you’re about to get clear, simple answers that will deepen your knowledge and spark your curiosity.
Basic Electron Setup
The basic electron setup of silver is unique and interesting. Silver has 47 electrons arranged in specific energy levels around its nucleus. These electrons fill different orbitals in a defined order. Understanding this setup helps explain silver’s chemical behavior and properties.
Electrons fill orbitals starting from the lowest energy level to higher ones. The arrangement follows the Aufbau principle, which helps predict the order of filling. For silver, the electron configuration shows how electrons occupy the s, p, d, and f sublevels.
Electron Shells And Energy Levels
Silver’s electrons are organized in shells or energy levels. Each shell can hold a limited number of electrons. The first shell holds 2, the second 8, the third 18, the fourth 18, and the fifth shell starts filling next. These shells are filled to keep the atom stable.
Filling Of Orbitals In Silver
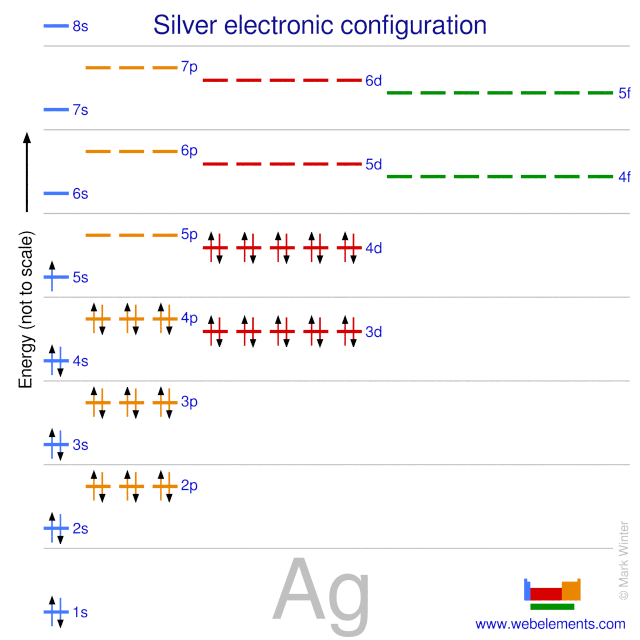
Silver’s orbitals fill in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, and 4d. The 4d orbital fills after the 5s orbital begins. But silver has an exception. It has one electron in the 5s orbital and ten electrons in the 4d orbital. This arrangement gives silver extra stability.
The electron configuration for silver is written as [Kr] 4d10 5s1. The [Kr] represents the electron configuration of krypton, a noble gas preceding silver. This shorthand shows only the electrons beyond krypton. It makes the configuration simpler and easier to read.
Credit: winter.group.shef.ac.uk
Silver’s Atomic Structure
Silver is a fascinating element with a unique atomic structure. It has an atomic number of 47, meaning it contains 47 protons in its nucleus. The electrons surrounding the nucleus fill specific energy levels and orbitals.
This arrangement of electrons is called the electron configuration. It helps explain silver’s chemical behavior and physical properties. Silver’s electron configuration differs slightly from other elements in its group.
Basic Atomic Composition Of Silver
Silver atoms have 47 electrons that orbit around the nucleus. These electrons fill energy shells in a specific order. The shells are labeled as 1, 2, 3, and so on, moving outward from the nucleus.
Each shell contains one or more subshells, called s, p, d, and f. These subshells hold a certain number of electrons. For silver, the outermost electrons play a key role in its reactions and bonding.
The electron configuration of silver is written using numbers and letters. It shows how electrons fill the shells and subshells. The full configuration for silver is [Kr] 4d10 5s1.
This means silver has a filled 4d subshell with 10 electrons and only one electron in the 5s subshell. This arrangement is unusual but stable. The [Kr] part represents the electron configuration of krypton, a noble gas before silver.
Why Silver’s Configuration Is Unique
Silver’s electron configuration differs from the expected order. Normally, the 5s subshell would fill before the 4d subshell. Silver, however, has a single electron in 5s and a full 4d subshell.
This happens because a full d subshell provides extra stability. This unique structure affects silver’s conductivity and chemical properties. It also explains why silver is a good conductor of electricity.
Role Of 4d And 5s Orbitals
Silver’s electron configuration shows one electron in the 5s orbital and a filled 4d orbital. The 4d orbitals hold ten electrons, while the 5s orbital contains a single electron, which influences silver’s chemical behavior and stability.
Understanding The 4d Orbital In Silver
The 4d orbital plays a major role in silver's electron setup. It holds the electrons that give silver its unique chemical traits. Unlike many elements, silver fills its 4d orbital almost completely, which affects its stability. This nearly full 4d orbital influences how silver bonds with other atoms. The electrons in 4d also impact silver’s color and conductivity.
The Importance Of The 5s Orbital
The 5s orbital in silver contains fewer electrons than expected. Silver has only one electron in the 5s orbital instead of two. This single 5s electron is important for silver’s chemical reactions. It is the electron that silver often loses to form ions. The 5s electron’s energy level affects how silver interacts with other elements.
How 4d And 5s Orbitals Work Together
The 4d and 5s orbitals in silver balance each other. The full 4d orbital stabilizes the atom, while the 5s orbital provides reactivity. This combination makes silver less reactive than some metals but very useful in many applications. The interplay between these orbitals explains silver's unique position in the periodic table. It also helps predict silver’s behavior in chemical reactions.

Credit: www.youtube.com
Electron Configuration Notation
Electron configuration notation describes how electrons arrange in an atom. It shows electron placement by energy levels and orbitals. This notation uses numbers, letters, and superscripts. It helps understand an element’s chemical behavior.
Silver's electron configuration notation reveals its unique structure. Silver (Ag) has 47 electrons. The notation shows how these electrons fill different orbitals.
Basics Of Electron Configuration Notation
The notation combines numbers and letters. Numbers represent energy levels, such as 1, 2, 3. Letters (s, p, d, f) indicate orbital types. Superscripts show the number of electrons in each orbital.
For example, "4d^10" means ten electrons in the 4d orbital. This system helps visualize electron distribution clearly and simply.
Electron Configuration Of Silver (ag)
Silver’s configuration is unique due to electron stability in d orbitals. Its full notation is: [Kr] 4d10 5s1.
This means silver has a full 4d subshell and one electron in 5s. The notation starts with the noble gas krypton [Kr] to shorten it.
Why Silver’s Configuration Is Different
Silver prefers 4d10 5s1 over 4d9 5s2. Fully filled d orbitals offer extra stability. This makes silver's chemistry special.
The electron configuration affects silver’s conductivity and reactivity. Understanding this helps in studying its properties and uses.
Exceptions In Silver Configuration
The electron configuration of silver does not follow the expected order. Normally, electrons fill the 5s orbital before entering the 4d orbital. Silver, however, shows an exception to this rule.
This exception occurs because a fully filled d-subshell offers extra stability. So, one electron from the 5s orbital moves to the 4d orbital. This creates a configuration that is more stable than the expected one.
Expected Electron Configuration Of Silver
The predicted electron configuration for silver is [Kr] 5s² 4d⁹. This follows the Aufbau principle where the 5s orbital fills before 4d.
However, this configuration is not the most stable for silver atoms. The energy difference between 5s and 4d orbitals is small. This allows electrons to rearrange for greater stability.
Actual Electron Configuration Of Silver
The actual electron configuration is [Kr] 5s¹ 4d¹⁰. Here, the 4d subshell is completely filled. This gives silver extra stability compared to the expected configuration.
Having a full 4d subshell lowers the overall energy of the atom. This is why silver prefers this electron arrangement.
Reason For The Exception
Electron repulsion and orbital energy levels influence this exception. A full d-subshell reduces electron repulsion inside the atom. This makes the atom more stable.
The energy gained by filling the 4d orbital outweighs the energy cost of removing one electron from 5s. This balance causes the unique electron configuration of silver.
Silver Ion (ag+) Configuration
The silver ion, symbolized as Ag+, forms when silver loses one electron. This change affects its electron arrangement.
Understanding the silver ion's electron configuration helps explain its chemical behavior. The positive charge means fewer electrons than a neutral atom.
Electron Loss In Silver Ion Formation
Silver's neutral atom has 47 electrons. Losing one electron creates Ag+ with 46 electrons. This electron is removed from the outermost shell.
The electron lost is usually from the 5s orbital, which is higher in energy than 4d orbitals. This makes the 5s electron easier to remove.
Electron Configuration Of Ag+
The neutral silver atom has the configuration [Kr] 4d10 5s1. Removing one 5s electron changes this.
The silver ion's configuration becomes [Kr] 4d10. It has a full 4d subshell and no electrons in the 5s orbital.
Stability Of The Silver Ion Configuration
A full 4d10 subshell is very stable. This stability explains why silver easily forms a +1 ion.
The filled d subshell offers lower energy and greater chemical stability for Ag+.
Comparison With Neighboring Elements
Silver’s electron configuration stands out among its neighbors in the periodic table. This uniqueness affects its chemical behavior and physical properties. Comparing silver with nearby elements reveals interesting patterns in electron arrangement and stability.
Electron Configuration Of Silver
Silver has the electron configuration [Kr] 4d10 5s1. This means it fills the 4d subshell completely and keeps one electron in the 5s orbital. This configuration is more stable than having a fully filled 5s orbital and a partially filled 4d subshell.
Electron Configuration Of Copper
Copper, just before silver, shows a similar pattern. Its configuration is [Ar] 3d10 4s1. Like silver, copper favors a full d subshell and a single s electron. This arrangement provides extra stability due to electron pairing and exchange energy.
Electron Configuration Of Cadmium
Cadmium, which comes after silver, has the configuration [Kr] 4d10 5s2. Here, the 5s orbital is fully occupied. Unlike silver, cadmium does not move an electron from the s to the d subshell. This leads to different chemical properties compared to silver.
Impact Of Electron Configuration Differences
The slight shifts in electron placement influence the elements’ reactivity. Silver and copper tend to form compounds where the d electrons play a key role. Cadmium’s filled s orbital makes it less reactive and changes its bonding characteristics.
Implications For Chemical Properties
The electron configuration of silver directly shapes its chemical behavior. Silver’s unique arrangement of electrons influences its bonding, reactivity, and physical traits. Understanding these effects helps explain why silver acts differently from other elements in the same group.
Silver has an unusual electron configuration. Instead of following the expected pattern, it fills one electron into the 5s orbital and ten into the 4d orbitals. This arrangement plays a key role in silver’s chemical properties.
Stability And Reactivity
Silver’s electron configuration creates a stable d-subshell. This stability lowers its tendency to lose electrons easily. As a result, silver shows moderate reactivity compared to other transition metals. It resists corrosion and oxidation better than many metals.
Formation Of Ions
Silver typically forms a +1 ion by losing one 5s electron. The filled 4d orbitals remain intact, which stabilizes the ion. This ion form is common in silver compounds and influences their chemical behavior and uses.
Conductivity And Metallic Properties
The electron setup allows silver to conduct electricity very well. Free electrons in the 5s orbital move easily through the metal. This explains silver’s high electrical and thermal conductivity, making it valuable in electronics and industry.
Catalytic Behavior
Silver’s d-electrons participate in catalytic reactions. The partially filled d-orbitals can interact with molecules on the surface. This ability enhances silver’s role as a catalyst in chemical processes, especially in oxidation reactions.
Electron Configuration In Periodic Trends
Electron configuration reveals how electrons fill atomic orbitals in elements. It follows specific rules that reflect the atom's energy levels and sublevels. These configurations help explain the chemical behavior of elements across the periodic table.
Periodic trends show patterns in electron arrangements as you move through periods and groups. These trends affect an element's properties like reactivity, magnetism, and conductivity. Silver's electron configuration fits into these trends but has unique traits too.
Basic Electron Configuration Of Silver
Silver has an atomic number of 47. Its electrons fill shells according to energy order. The configuration starts with the core noble gas krypton, written as [Kr]. After that, electrons occupy the 4d and 5s orbitals.
The full configuration is [Kr] 4d10 5s1. This means the 4d sublevel is completely filled with 10 electrons. The 5s sublevel holds a single electron, which is unusual for this group.
Exception In Electron Filling For Silver
Normally, the 5s orbital fills before the 4d orbital. Silver breaks this pattern. It moves one electron from 5s to 4d to achieve a full d sublevel. This arrangement gives silver extra stability.
This exception occurs because a filled d sublevel is more stable than a half-filled s sublevel. The shift lowers the atom's overall energy. This behavior is common among transition metals near silver.
Comparison With Other Periodic Elements
Elements before silver have partially filled d orbitals. For example, palladium has [Kr] 4d10 5s0. Elements after silver start filling the 5p orbitals. This trend shows how electron configuration changes across the period.
Silver’s unique configuration influences its chemical and physical traits. It has good conductivity and resistance to corrosion. These properties relate directly to its electron arrangement.

Credit: www.chegg.com
Applications In Material Science
Silver's electron configuration plays a key role in its use in material science. Understanding how electrons arrange in silver atoms helps scientists develop new materials. Silver's unique properties come from its electron structure, which affects conductivity and bonding.
Material scientists explore silver’s electron configuration to improve electronics, coatings, and catalysts. These applications depend on how silver interacts at the atomic level. Silver's single electron in the 5s orbital and filled 4d orbitals create special chemical behaviors.
Electrical Conductivity And Electron Mobility
Silver has the highest electrical conductivity among metals. This is due to its electron configuration, which allows easy flow of electrons. Electrons in the 5s orbital can move freely, making silver ideal for wires and circuits. Material scientists use this property to design efficient electrical components.
Silver Nanoparticles In Catalysis
Silver’s electron arrangement helps in catalytic reactions. Nanoparticles made from silver show high reactivity. The 4d electrons participate in bonding with other molecules. This makes silver useful in speeding up chemical reactions in industries and environmental cleanup.
Antimicrobial Coatings And Electron Interaction
Silver’s electron configuration also supports antimicrobial properties. Silver ions disrupt bacterial cells by interacting with their electrons. Coatings with silver prevent infections on medical devices and surfaces. These coatings rely on silver’s ability to release electrons to kill microbes.
Optical Properties And Plasmonics
Silver’s electrons respond to light in special ways. This leads to plasmonic effects used in sensors and photonic devices. The electron configuration enables silver to absorb and scatter light efficiently. Researchers use this feature to create advanced optical materials.
Frequently Asked Questions
How To Write Electron Configuration For Silver?
Write silver's electron configuration as [Kr] 4d^10 5s^1. It fills the 4d subshell fully and has one electron in 5s. This unique arrangement provides silver's chemical properties.
What Element Has The Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d10?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d10 is Silver (Ag). It has a unique 5s1 4d10 arrangement.
What Is The Electron Configuration Of 1s2 2s2 2p6 3s2 3p6?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ corresponds to the element Argon (Ar). It shows a complete filling of the first three shells. This configuration represents a stable, noble gas atom with 18 electrons.
Is Silver Always 2+?
Silver commonly shows a +1 oxidation state but can also exhibit a +2 state in rare compounds. It is not always 2+.
Conclusion
Silver’s electron configuration shows its unique atomic structure. It has one electron in the 5s orbital and ten in the 4d orbitals. This arrangement makes silver stable and explains its chemical behavior. Understanding this helps in studying silver’s properties and uses.
Remember, the electron configuration is key to predicting how silver interacts with other elements. Simple and clear knowledge like this aids learning chemistry effectively. Keep exploring atomic structures to see how elements behave in nature.



No comments