Electron Configuration of Ruthenium
If you’ve ever wondered what makes ruthenium unique among the transition metals, understanding its electron configuration is the key. Knowing how its electrons are arranged can reveal why ruthenium behaves the way it does in chemical reactions and why it’s valuable in industries like electronics and catalysis.
You’ll discover the exact electron setup of ruthenium, learn why it differs from what you might expect, and see how this knowledge can deepen your grasp of chemistry. Ready to unlock the secrets behind ruthenium’s electrons? Let’s dive in and explore!
Ruthenium Basics
Ruthenium is a rare metal found in the platinum group. It has many uses in electronics and industry. Understanding its electron structure helps explain its properties. This section introduces the basic facts about ruthenium.
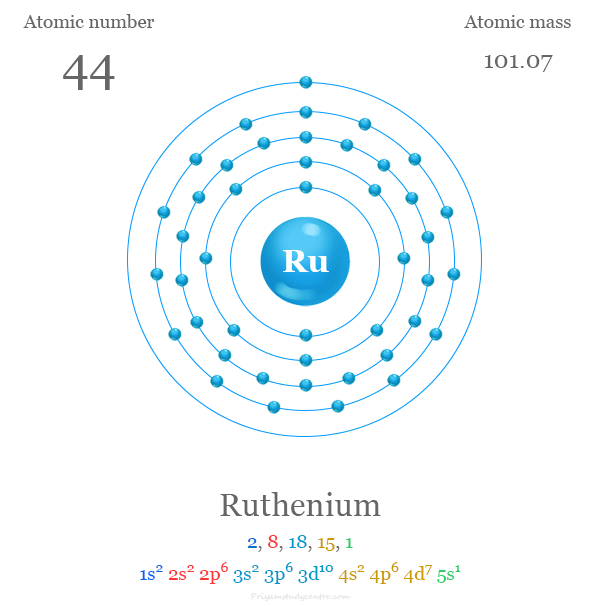
Its atomic number is 44. This means ruthenium has 44 protons and 44 electrons. These electrons are arranged in layers around the nucleus. The arrangement affects how ruthenium reacts with other elements.
Atomic Number And Symbol
The atomic number of ruthenium is 44. Its chemical symbol is Ru. This symbol comes from its Latin name, “Ruthenium.” The element is part of the transition metals group.
Position In The Periodic Table
Ruthenium is in period 5 and group 8. It sits among other platinum group metals. Its position shows it has properties between iron and osmium. This helps predict how it behaves in chemical reactions.
Physical Characteristics
Ruthenium is a hard, silvery-white metal. It has a high melting point and good corrosion resistance. These traits make it useful in harsh environments. Its physical strength supports industrial uses.
Atomic Structure
The atomic structure of ruthenium defines its chemical behavior and physical properties. It is a transition metal found in the periodic table with the symbol Ru and atomic number 44. Understanding its atomic structure helps explain its role in catalysts and electronics.
Ruthenium has a unique arrangement of electrons around its nucleus. These electrons occupy different energy levels or shells. Each shell contains sublevels that hold electrons in specific orbitals.
Electron Shells And Energy Levels
Ruthenium's electrons are organized into shells labeled by numbers. Each shell corresponds to a principal energy level. The first shell holds 2 electrons, and the number of electrons increases in outer shells. Ruthenium’s electrons fill up to the fifth shell.
Subshells And Orbitals
Within each shell, electrons occupy subshells named s, p, d, and f. These subshells have different shapes and capacities. Ruthenium’s outer electrons mainly fill the 4d and 5s subshells. The d subshell can hold up to 10 electrons, while s can hold 2.
Electron Configuration Of Ruthenium
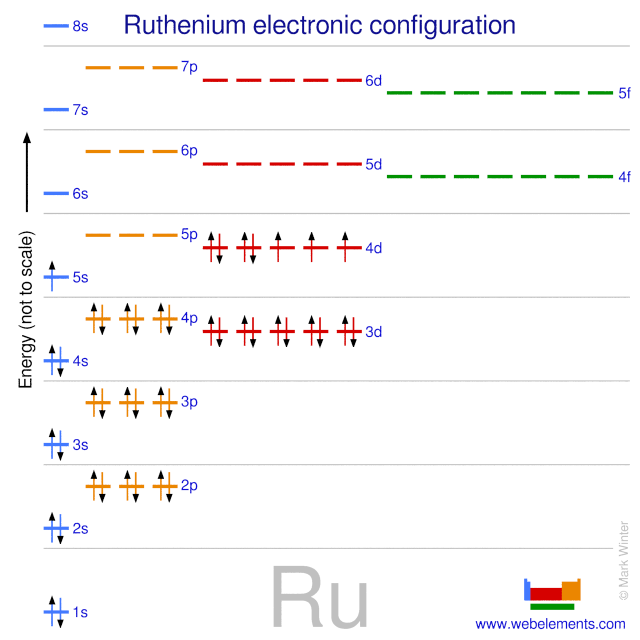
The electron configuration of ruthenium is written as [Kr] 4d7 5s1. This notation shows it has one electron in the 5s subshell and seven in the 4d subshell. This arrangement affects its chemical bonding and reactivity.
Ruthenium’s electron configuration differs from the expected pattern. It has one electron moved from 5s to 4d to gain stability. This subtle change makes it more chemically stable and reactive in certain conditions.
Electron Shells
Ruthenium’s electron configuration shows how electrons fill its shells and orbitals. It has a unique arrangement with electrons in the 4d and 5s shells. This pattern helps explain ruthenium’s chemical behavior and properties.
The electron shells of ruthenium reveal how its electrons are arranged around the nucleus. These shells hold electrons in specific energy levels. Each shell contains subshells with different shapes and capacities.
Understanding ruthenium’s electron shells helps explain its chemical behavior. The arrangement shows how ruthenium shares or transfers electrons in reactions. It also influences its magnetic and electrical properties.
Principal Energy Levels Of Ruthenium
Ruthenium has electrons filling several principal energy levels. These levels are labeled 1 to 5, with increasing distance from the nucleus. The higher the level, the more energy its electrons have.
In ruthenium, the first few shells are fully filled. The outer shells contain electrons that participate in bonding. This balance affects ruthenium’s stability and reactivity.
Distribution Of Electrons In Subshells
Electrons in ruthenium occupy s, p, and d subshells within each shell. The s subshell holds up to 2 electrons, p up to 6, and d up to 10. Ruthenium’s unique electron configuration arises from its d subshell.
The 4d subshell in ruthenium is partially filled, which impacts its chemical properties. This partial filling allows ruthenium to form various oxidation states. It also gives ruthenium its catalytic abilities.
Electron Configuration Summary
Ruthenium’s electron configuration is [Kr] 4d7 5s1. This means it has one electron in the 5s shell and seven in the 4d shell. This arrangement differs from the expected pattern and reflects stability in its electron shells.
The unusual distribution helps ruthenium maintain balance between energy and stability. This shell structure is key to understanding ruthenium’s behavior in compounds and materials.
Orbital Filling Order
The orbital filling order explains how electrons occupy atomic orbitals in an atom. Electrons fill orbitals in a specific sequence to minimize energy. This order shapes the chemical behavior of elements like ruthenium.
Ruthenium, with atomic number 44, follows the general principles of orbital filling. Its electrons fill lower energy orbitals before moving to higher ones. This order affects ruthenium’s properties and bonding patterns.
Understanding The Aufbau Principle
The Aufbau principle guides the order of orbital filling. Electrons enter orbitals starting from the lowest energy level. This rule helps predict the electron arrangement for ruthenium and other elements.
Role Of Pauli Exclusion Principle
The Pauli exclusion principle states that each orbital holds a maximum of two electrons. These electrons must have opposite spins. This rule limits how ruthenium’s electrons occupy orbitals.
Hund's Rule And Electron Distribution
Hund's rule says electrons fill degenerate orbitals singly first. They pair up only after each orbital has one electron. This rule explains ruthenium’s electron placement in d orbitals.
Filling Order Specific To Ruthenium
Ruthenium’s electron configuration is unique due to d-orbital stability. Its configuration is often written as [Kr] 4d7 5s1. The 5s orbital fills before the 4d orbitals but can lose one electron to increase d orbital stability.
Ground State Configuration
Ruthenium’s ground state electron configuration is [Kr] 4d7 5s1. This arrangement shows one electron in the 5s orbital and seven in the 4d orbitals. Such a setup helps explain its chemical behavior and properties.
What Is Ground State Configuration?
The ground state configuration shows the arrangement of electrons in an atom. It represents the lowest energy state. Electrons fill orbitals starting from the lowest energy level. This pattern follows the Aufbau principle.
For ruthenium, understanding this arrangement helps explain its chemical behavior. The ground state tells us how electrons occupy shells and subshells. This setup impacts its bonding and reactivity.
Ruthenium’s Electron Arrangement
Ruthenium has the atomic number 44. This means it has 44 electrons. These electrons are arranged to minimize energy. The order fills the 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, and 4d orbitals.
The ground state configuration of ruthenium is [Kr] 4d7 5s1. This means seven electrons are in the 4d subshell and one in the 5s subshell. This arrangement is slightly different from the expected 4d6 5s2.
Why Does Ruthenium Have This Configuration?
This configuration occurs due to electron stability. Half-filled and fully filled subshells have extra stability. Moving one electron from 5s to 4d creates a more stable state. The 4d subshell gains stability with seven electrons.
Such electron shifts are common in transition metals. They show the subtle balance between energy levels. Understanding this helps predict ruthenium’s chemical properties.
Credit: winter.group.shef.ac.uk
Exceptions In Configuration
Electron configurations usually follow clear rules, but some elements show exceptions. Ruthenium is one such element. Its electron arrangement differs from the expected pattern. These exceptions arise from the stability of half-filled or fully filled orbitals.
Understanding these exceptions helps explain ruthenium’s chemical behavior. It also shows how electron interactions influence atomic structure. Let’s explore the unique aspects of ruthenium’s electron configuration.
Expected Electron Configuration Of Ruthenium
Ruthenium has an atomic number of 44. The expected configuration fills the 5s orbital before the 4d orbitals. This would be written as 5s² 4d⁶. This follows the general order of filling orbitals based on energy levels.
Actual Electron Configuration
Ruthenium’s real electron configuration is 5s¹ 4d⁷. It has one electron less in the 5s orbital and one more in the 4d orbitals. This shift provides extra stability to the atom. The 4d orbitals become more stable when partially filled.
Reason For The Exception
Electrons repel each other and try to minimize energy. Moving one electron from the 5s to the 4d orbital lowers the total energy. Half-filled or nearly half-filled d orbitals are more stable. This makes ruthenium more chemically stable with this configuration.
Impact On Chemical Properties
This unusual configuration affects ruthenium’s reactivity. It influences bonding and magnetic properties. The electron arrangement helps explain ruthenium’s role in catalysts and alloys. It also affects how ruthenium interacts with other elements.
Valence Electrons Role
Valence electrons determine ruthenium’s chemical behavior and bonding. Its electron configuration shows one electron in the 5s orbital and seven in the 4d orbitals. These outer electrons influence ruthenium’s reactivity and properties.
Understanding Ruthenium’s Valence Electrons
Valence electrons are the outermost electrons of an atom. They play a key role in chemical reactions and bonding. Ruthenium’s valence electrons determine its ability to form compounds and interact with other elements.
Ruthenium has an electron configuration of [Kr] 4d7 5s1. This means it has eight valence electrons in the 4d and 5s orbitals. These electrons are crucial for its chemical properties.
How Valence Electrons Influence Ruthenium’s Properties
The valence electrons control how ruthenium bonds with other atoms. They affect its oxidation states and reactivity. Ruthenium can lose or share these electrons to form stable compounds.
Its ability to switch between different oxidation states makes ruthenium useful in catalysts and industrial applications. The valence electrons allow it to adapt and participate in various chemical processes.
Valence Electrons And Ruthenium’s Electron Configuration
The unique arrangement of ruthenium’s valence electrons impacts its stability. The presence of one electron in the 5s orbital and seven in the 4d orbital gives it special chemical behavior.
This electron configuration is slightly different from other transition metals. It helps ruthenium achieve a balance between energy levels, making it versatile in forming bonds.

Credit: www.youtube.com
Comparison With Rhodium
Ruthenium and rhodium are neighbors in the periodic table. Both belong to the same group of transition metals. Their electron configurations share similarities but have key differences.
These differences influence their chemical behavior and physical properties. Understanding their electron arrangements helps explain their unique characteristics.
Electron Configuration Of Ruthenium
Ruthenium has the electron configuration [Kr] 4d7 5s1. It fills the 4d orbitals with seven electrons and has one electron in the 5s orbital. This arrangement is slightly unusual compared to the expected filling order.
Electron Configuration Of Rhodium
Rhodium’s electron configuration is [Kr] 4d8 5s1. It has eight electrons in the 4d orbitals and one in the 5s orbital. This differs from ruthenium by one more electron in the d subshell.
Impact On Chemical Properties
The difference in d electrons affects their bonding abilities. Ruthenium often shows multiple oxidation states. Rhodium is more stable in fewer oxidation states. These patterns influence their use in catalysts and alloys.
Physical And Catalytic Differences
Ruthenium is harder and more resistant to corrosion than rhodium. Rhodium has a higher melting point. Both metals serve as catalysts but excel in different chemical reactions. Their electron structures shape these traits.
Chemical Properties Link
The electron configuration of ruthenium shapes its chemical behavior. This arrangement of electrons influences how ruthenium bonds with other elements. Understanding this link helps explain ruthenium’s reactivity and stability.
Ruthenium’s unique electron setup affects its oxidation states and catalytic abilities. These chemical properties determine its applications in industry and research.
Electron Configuration Overview
Ruthenium has the electron configuration [Kr] 4d7 5s1. This means seven electrons fill the 4d sublevel and one electron occupies the 5s sublevel. This unusual distribution affects its chemical traits.
Impact On Oxidation States
The electron arrangement allows ruthenium to have multiple oxidation states. Common states include +2, +3, +4, and +8. This variety arises from the ease of losing or sharing electrons in d and s orbitals.
Influence On Catalytic Behavior
Ruthenium’s electron configuration promotes catalytic activity in chemical reactions. The partially filled d orbitals enable bonding with reactants. This helps speed up reactions without being consumed.
Bonding Characteristics
Ruthenium forms strong bonds with many elements. The electron distribution supports both covalent and metallic bonding. This flexibility enhances ruthenium’s use in alloys and compounds.
Applications Of Ruthenium
Ruthenium is a rare metal with many practical uses. Its unique electron configuration gives it special properties. These properties make ruthenium valuable in different fields.
Ruthenium finds applications in electronics, catalysis, and jewelry. Each use takes advantage of its strength, durability, and chemical stability.
Electronic Devices
Ruthenium is used in electronic components. It improves the performance of computer chips and resistors. Thin films of ruthenium help in making durable circuits. These films resist corrosion and wear well over time.
Catalysis In Chemical Reactions
Ruthenium acts as a catalyst in many chemical processes. It speeds up reactions without being used up. Industries use ruthenium catalysts to produce ammonia and other chemicals. It also helps in hydrogenation and oxidation reactions.
Jewelry And Decorative Uses
Ruthenium is added to platinum and other metals in jewelry. This addition increases hardness and scratch resistance. It gives a dark, attractive finish to rings and watches. Its resistance to tarnish keeps jewelry looking new longer.
Renewable Energy
Ruthenium plays a role in renewable energy technologies. It is part of catalysts in fuel cells. Fuel cells convert hydrogen into electricity efficiently. Ruthenium helps improve the durability and efficiency of these cells.
Credit: www.priyamstudycentre.com
Frequently Asked Questions
What Element Has An Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 is Rubidium (Rb), atomic number 37.
Which Element Is This 1s22s22p63s23p64s23d104p5?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5 represents the element Bromine (Br).
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration can be 2, 8, 8 or 2, 8, 18 depending on the element and its energy levels.
What Is The Element Configuration For 1s 2 2s 2 2p 6 3s 2 3p 4?
The electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴ corresponds to the element sulfur (S). It has 16 electrons.
Conclusion
Understanding ruthenium’s electron configuration helps explain its chemical behavior. Its unique arrangement of electrons influences bonding and reactivity. Knowing this configuration aids in studying transition metals and their properties. This knowledge supports learning about the periodic table and element trends.
Ruthenium’s configuration shows how electrons fill orbitals in complex ways. Such details make chemistry clearer and more interesting. Keep exploring elements and their electron patterns for better science understanding.



No comments