Electron Configuration of Krypton
Have you ever wondered what makes krypton so unique among the elements? Understanding the electron configuration of krypton unlocks the secrets behind its chemical behavior and stability.
By diving into how its electrons are arranged, you’ll gain a clear picture of why krypton is a noble gas and why it rarely reacts with other substances. You’ll discover the step-by-step electron configuration of krypton, learn the principles that govern its arrangement, and see how this knowledge applies to real-world chemistry.
Stick with us, and you’ll find that mastering krypton’s electron configuration is simpler than you think—and essential for anyone interested in chemistry.
Krypton Basics
Krypton is a noble gas element found in the periodic table. It is colorless, odorless, and tasteless. Krypton is chemically inert, meaning it rarely reacts with other elements.
This element has the atomic number 36. That means it has 36 protons in its nucleus. In a neutral atom, it also has 36 electrons orbiting the nucleus.
Krypton is part of group 18, known as the noble gases. These gases have full outer electron shells, making them very stable. This stability explains krypton's low reactivity.
Atomic Number And Symbol
The atomic number of krypton is 36. Its chemical symbol is Kr. This symbol represents krypton in the periodic table and chemical formulas.
Physical Properties
Krypton is a gas at room temperature. It has a density higher than air. Krypton emits a bright greenish-yellow light when electrified, useful in lighting.
Chemical Behavior
Krypton rarely forms compounds due to its full electron shell. It is very stable and does not easily gain or lose electrons. This makes krypton a noble gas.
Atomic Number And Electrons
The atomic number of an element tells how many protons it has in its nucleus. For a neutral atom, the number of electrons equals the atomic number. Electrons orbit the nucleus in shells and subshells. Their arrangement defines the element’s chemical properties.
Krypton is a noble gas with a full outer shell of electrons. This stable electron arrangement makes it mostly unreactive. Knowing the atomic number and electron count helps us write its electron configuration correctly.
Atomic Number Of Krypton
Krypton has an atomic number of 36. This means it contains 36 protons in its nucleus. Since krypton is neutral, it also has 36 electrons orbiting the nucleus.
Number Of Electrons In Krypton
The 36 electrons in krypton fill several energy levels. Electrons fill orbitals in a specific order. This order follows the principles of quantum mechanics.
Significance Of Electron Count
The total electron count defines krypton’s chemical behavior. Its full outer shell makes it very stable. It rarely forms chemical bonds with other elements.
Electron Shells Of Krypton
Krypton is a noble gas with a full set of electron shells. Its electron shells are arranged in a way that makes it stable and unreactive. Each shell holds a specific number of electrons. These electrons fill the shells in a fixed order. This order follows clear rules based on energy levels.
Studying the electron shells of krypton helps us understand its chemical behavior. The arrangement explains why krypton rarely forms compounds. It also shows how electrons are distributed around the nucleus.
Electron Shell Distribution Of Krypton
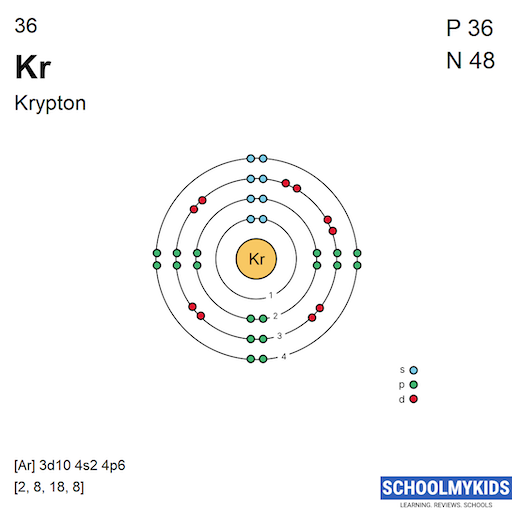
Krypton has 36 electrons. These electrons are divided into four main shells. The first shell holds 2 electrons. The second shell contains 8 electrons. The third shell carries 18 electrons. The fourth shell has 8 electrons. This distribution follows the 2n2 rule, where n is the shell number.
Energy Levels And Subshells
Each shell has subshells with different energy levels. Krypton’s electrons fill the subshells in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p. The 3d subshell fills after the 4s shell. This filling order is important for the electron configuration. It ensures the lowest energy state for the atom.
The electron configuration of krypton is written as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶. This notation shows how electrons fill each subshell. The superscripts indicate the number of electrons in each subshell. This full configuration reflects krypton’s stable electron shell arrangement.

Credit: www.webelements.com
Electron Configuration Notation
Electron configuration notation shows how electrons arrange around an atom's nucleus. It uses numbers, letters, and superscripts to describe electron placement in energy levels and orbitals.
This notation helps us understand an element's chemical behavior and position in the periodic table. For krypton, electron configuration reveals its full outer shell, explaining its stability.
Basics Of Electron Configuration Notation
Electron configuration uses numbers to show energy levels. Letters s, p, d, and f indicate orbital types. Superscripts tell how many electrons occupy each orbital.
For example, "1s²" means two electrons are in the first energy level's s orbital. This simple code organizes complex atomic structure clearly.
Electron Configuration Of Krypton
Krypton has 36 electrons. Its configuration fills orbitals up to the fourth energy level. The full notation is: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶.
This shows krypton has a complete outer shell with eight electrons in the 4p and 4s orbitals. This full shell makes krypton a noble gas.
Using Noble Gas Shortcut Notation
To simplify, we use the noble gas before krypton in brackets. For krypton, this is [Ar], representing argon's 18 electrons.
The shorthand notation for krypton is: [Ar] 3d¹⁰ 4s² 4p⁶. This saves space and keeps the notation clear.
Using The Aufbau Principle
The Aufbau Principle helps us understand how electrons fill atomic orbitals. It states that electrons occupy the lowest energy orbitals first before moving to higher ones. This rule is essential for writing electron configurations, including that of krypton.
Following this principle ensures the correct order of filling orbitals. Electrons fill orbitals starting from 1s, then 2s, 2p, and so on. This order explains krypton's stable electron arrangement.
What Is The Aufbau Principle?
The Aufbau Principle means "building up" in German. It guides how electrons fill orbitals in an atom. Electrons fill orbitals from lowest to highest energy. This method helps predict the electron configuration of elements.
Applying The Aufbau Principle To Krypton
Krypton has 36 electrons. Using the Aufbau Principle, electrons fill orbitals in this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p. Each orbital fills completely before electrons enter the next one.
This order leads to krypton's full electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶. This makes krypton stable and chemically inert.
Why The Aufbau Principle Matters
The principle helps explain the chemical behavior of elements. For krypton, full orbitals mean low reactivity. It also aids students and chemists in predicting electron arrangements easily.
Pauli Exclusion Principle Role
The Pauli Exclusion Principle plays a vital role in the electron configuration of krypton. It states that no two electrons in an atom can have the same set of four quantum numbers. This rule guides how electrons fill the orbitals of krypton, ensuring each electron occupies a unique state.
Because of this principle, electrons in krypton's orbitals pair up with opposite spins. This pairing affects the arrangement of electrons in the 4s and 4p orbitals. The principle limits the number of electrons that can occupy each orbital, shaping the atom's structure.
Understanding Electron Spin And Orbital Filling
The Pauli Exclusion Principle relies on electron spin, which can be either up or down. Each orbital can hold two electrons, but their spins must differ. This spin difference allows two electrons to share an orbital without violating the principle.
In krypton, the 4p orbitals fill with paired electrons having opposite spins. This balanced filling stabilizes the atom and explains krypton's noble gas behavior.
Impact On Krypton's Stable Electron Configuration
Krypton has 36 electrons that follow the Pauli Exclusion Principle strictly. Its electron configuration ends at 4p6, meaning the outer shell is full. This full outer shell results from the principle's rule on electron pairing and unique quantum states.
This stability makes krypton chemically inert, with little tendency to react. The principle ensures electrons fill orbitals in a way that leads to this stable, low-energy state.
Relation To Other Quantum Rules
The Pauli Exclusion Principle works alongside the Aufbau Principle and Hund’s Rule. While Aufbau directs the order of orbital filling, and Hund’s Rule governs electron distribution within orbitals, Pauli’s principle restricts electron spins.
These three rules together describe krypton's electron arrangement clearly. They explain why electrons occupy certain orbitals and how their spins organize to maintain atomic stability.
Hund’s Rule Application
Hund’s Rule plays a key role in understanding krypton’s electron arrangement. It guides how electrons fill orbitals within the same energy level. The rule states that electrons fill empty orbitals singly before pairing up. This reduces electron repulsion and stabilizes the atom.
In krypton, Hund’s Rule helps explain the distribution of electrons in the 4p sublevel. It ensures that electrons occupy separate orbitals first. This leads to a more stable and lower-energy electron configuration.
Electron Distribution In Krypton’s 4p Sublevel
Krypton has six electrons in its 4p sublevel. According to Hund’s Rule, these electrons spread out across three 4p orbitals. Each orbital gets one electron before any pairing occurs. This arrangement minimizes repulsion and balances the atom’s energy.
Impact Of Hund’s Rule On Krypton’s Stability
By following Hund’s Rule, krypton achieves a stable electron configuration. The electrons are arranged to lower repulsive forces inside the atom. This stability contributes to krypton’s chemical inertness and noble gas characteristics.
Visualizing Hund’s Rule With Orbital Diagrams
Orbital diagrams show how electrons fill orbitals following Hund’s Rule. For krypton, each 4p orbital is first singly occupied. Then, electrons pair up if more electrons remain. This visual tool helps learners see electron placement clearly.
Credit: www.schoolmykids.com
Orbital Diagram For Krypton
Krypton’s electron configuration shows how its 36 electrons fill the atomic orbitals. The orbital diagram arranges these electrons by energy levels and sublevels clearly. This structure explains krypton’s stability as a noble gas.
Introduction To The Orbital Diagram For Krypton
The orbital diagram for krypton shows how its electrons are arranged in different orbitals. Each orbital can hold up to two electrons with opposite spins. Krypton has 36 electrons that fill its orbitals in a specific order.
This diagram helps visualize electron distribution. It follows the Aufbau principle, which states electrons fill the lowest energy orbitals first. The diagram also respects Hund’s rule and the Pauli exclusion principle.
Filling Order Of Orbitals In Krypton
Krypton’s electrons fill orbitals in this sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p. The 1s orbital fills first with two electrons. Then, 2s fills with two electrons, followed by six in 2p orbitals.
Next, 3s and 3p orbitals fill with two and six electrons, respectively. The 4s orbital fills before the 3d orbitals due to energy levels. After 3d is filled with ten electrons, 4p fills last with six electrons.
Orbital Diagram Representation
In the orbital diagram, each box represents one orbital. Arrows inside boxes show electrons and their spins. A pair of arrows with opposite directions means two electrons share that orbital.
For krypton, the diagram shows fully filled subshells. The 1s, 2s, 2p, 3s, 3p, 4s, 3d, and 4p orbitals each have all possible electrons. This full filling explains krypton’s chemical stability.
Significance Of Krypton’s Orbital Diagram
Krypton’s orbital diagram reveals why it is a noble gas. All its outer orbitals are full, making it very stable and unreactive. The full 4p orbital means krypton does not easily gain or lose electrons.
This stability is why krypton rarely forms compounds. Its electron arrangement creates a balanced, low-energy state. The orbital diagram visually explains these important chemical properties.
Valence Electrons And Stability
Krypton is a noble gas with a full outer shell of electrons. This full shell makes krypton very stable and less likely to react with other elements. The number of valence electrons plays a key role in this stability. Valence electrons are the electrons in the outermost shell of an atom. They determine how an atom interacts with others.
Krypton has eight valence electrons. This complete set fills its outermost energy level. Atoms with full valence shells are often called "inert." They do not easily lose, gain, or share electrons. This is why krypton rarely forms chemical bonds.
Electron Configuration Of Krypton
Krypton’s electron configuration is written as [Ar] 3d10 4s2 4p6. This shows all the electrons in its shells. The last part, 4s2 4p6, represents the eight valence electrons. These electrons fill the fourth energy level completely, leading to a stable structure.
Role Of Valence Electrons In Stability
Valence electrons control chemical reactivity. With a full valence shell, krypton has low reactivity. It does not need to gain or lose electrons. This lack of need creates chemical stability. Krypton’s stability is why it is found in group 18 of the periodic table, known as noble gases.
Why Krypton Rarely Forms Compounds
Krypton’s stable electron configuration makes it less likely to bond. Other elements form compounds to reach full valence shells. Krypton already has a full shell. This means it rarely forms compounds under normal conditions. It stays mostly in its elemental form.
Krypton’s Chemical Inertness
Krypton is a noble gas known for its chemical inertness. Its electron configuration plays a key role in this property. The stable arrangement of electrons prevents it from reacting easily with other elements. This section explains why krypton remains mostly unreactive under normal conditions.
Krypton’s Full Electron Configuration
Krypton has 36 electrons. These electrons fill energy levels in a specific order. Its configuration is written as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶. This means the outermost shell is full. The full outer shell is very stable.
How Electron Configuration Causes Inertness
A full outer shell means krypton does not need to gain or lose electrons. It has no tendency to form bonds with other atoms. This makes krypton chemically inert. The energy required to change its electron setup is very high.
Comparison With Other Noble Gases
Like helium and neon, krypton’s outer shell is complete. All noble gases share this trait. This full shell blocks chemical reactions. Krypton’s electron configuration is a perfect example of why noble gases rarely form compounds.

Credit: study.com
Frequently Asked Questions
How To Write Electron Configuration For Krypton?
Write krypton's electron configuration as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶. It matches atomic number 36. Use Aufbau principle to fill orbitals sequentially.
What Electron Configuration Is 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 represents the element calcium (Ca) with atomic number 20.
What Element Has The Following Electron Configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d7?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d7 belongs to cobalt (Co). It has atomic number 27.
Is Electron Configuration 2 8 8 Or 2 8 18?
Electron configuration is 2 8 8 for elements up to argon. For heavier elements like calcium, it becomes 2 8 18. The correct configuration depends on the element's atomic number.
Conclusion
Krypton's electron configuration shows its full outer shell stability. This arrangement makes krypton a noble gas with low reactivity. Understanding this helps explain why it rarely forms compounds. Its electron setup follows clear rules like the Aufbau principle. Knowing krypton's configuration supports learning about other elements.
This knowledge is useful in chemistry and science studies. Simple and steady, krypton's electrons tell a story of balance.



No comments