Electron Configuration of Indium
When you dive into the world of chemistry, understanding the electron configuration of an element like indium can unlock many secrets about its behavior and properties. Are you curious about how indium’s electrons are arranged and why this matters for its chemical reactions?
By grasping its electron configuration, you’ll gain insights into its place in the periodic table, its valence electrons, and how it interacts with other elements. This knowledge is not only essential for students and professionals but also for anyone wanting a clearer picture of how atoms shape the world around you.
Keep reading to discover the step-by-step breakdown of indium’s electron configuration and why it’s important for your studies or curiosity.
Atomic Structure Of Indium
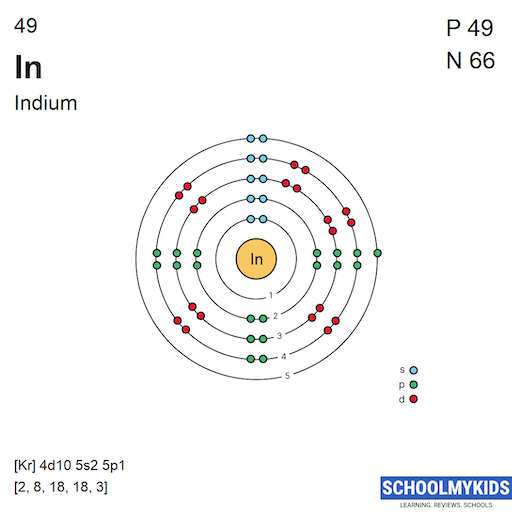
Indium’s electron configuration is [Kr] 4d10 5s2 5p1, showing its 49 electrons in shells. This arrangement explains its chemical behavior and bonding. Understanding electron placement helps reveal indium’s atomic structure clearly.
Understanding The Atomic Structure Of Indium
Indium is a soft, silvery metal found in the periodic table. Its atomic structure defines how it interacts with other elements. The structure consists of protons, neutrons, and electrons arranged in energy levels. These arrangements influence indium’s chemical and physical properties.
The atomic number of indium is 49, meaning it has 49 protons in its nucleus. To balance this, it also has 49 electrons orbiting the nucleus. These electrons fill specific orbitals following strict rules. This electron arrangement is called the electron configuration.
Electron Configuration Of Indium
Electrons occupy orbitals in a way that lowers the atom's energy. For indium, the electron configuration is written as: [Kr] 4d10 5s2 5p1. This notation shows the electrons beyond the noble gas krypton (Kr).
The 4d orbital holds 10 electrons, fully filling that shell. The 5s orbital contains 2 electrons, and the 5p orbital has 1 electron. This single electron in the 5p orbital plays a key role in indium’s reactivity.
Importance Of Valence Electrons In Indium
Valence electrons are the outermost electrons that atoms use to bond. Indium has three valence electrons: two in the 5s orbital and one in the 5p orbital. These electrons are involved in chemical reactions and bonding.
Because of its valence electrons, indium often forms +3 oxidation states. This means it can lose three electrons to form positive ions. Understanding valence electrons helps explain indium’s behavior in compounds.
Electron Shells And Subshells
Electron shells and subshells explain how electrons arrange around an atom's nucleus. These shells form layers at different energy levels. Each shell contains one or more subshells, where electrons fill specific orbitals. Understanding this helps explain an element’s chemical behavior and bonding.
Indium, with atomic number 49, has electrons spread across several shells and subshells. The arrangement follows a clear pattern defined by quantum rules. This pattern shows which electrons are in the outermost shell, influencing Indium’s reactivity.
Electron Shells In Indium
Indium has five electron shells. Each shell corresponds to a principal energy level, numbered 1 to 5. The first shell holds 2 electrons, while the outer shells hold more. The fifth shell contains the outermost electrons that determine chemical properties.
Subshells And Their Types
Subshells are groups within shells. They have different shapes and energies. The main types are s, p, d, and f subshells. Each type holds a specific number of electrons: s holds 2, p holds 6, d holds 10, and f holds 14.
Indium’s Electron Subshell Filling
Electrons fill subshells in order of increasing energy. For Indium, the filling sequence ends in the 5p subshell. It has electrons in the 1s, 2s, 2p, 3s, 3p, 3d, 4s, 4p, 4d, 5s, and 5p subshells. The final configuration reveals its position in the periodic table.
Ground State Configuration
The ground state configuration of an element shows how electrons fill its atomic orbitals. This arrangement defines the element’s chemical properties and stability. Indium, a post-transition metal, has a unique electron setup that influences its behavior in compounds and reactions.
Understanding indium’s ground state configuration helps in grasping its role in electronics and materials science. The electrons arrange themselves to minimize energy, following specific rules that determine the order of filling.
Basic Principles Of Electron Configuration
Electrons fill orbitals starting from the lowest energy level. The order follows the Aufbau principle. Each orbital can hold a limited number of electrons. Indium has 49 electrons that occupy orbitals in a specific sequence.
Electron Configuration Of Indium
Indium’s ground state electron configuration is: [Kr] 4d10 5s2 5p1. This means it has a full 4d subshell and two electrons in the 5s orbital. The last electron goes into the 5p orbital.
Significance Of The Configuration
This configuration explains indium’s +3 oxidation state in compounds. The outermost electrons in the 5s and 5p orbitals are involved in bonding. The filled 4d shell remains stable and less reactive.
Noble Gas Notation
Noble gas notation simplifies electron configurations by using the nearest noble gas as a starting point. This method saves time and makes it easier to read and write electron arrangements. It shows only the electrons added after the noble gas core.
For indium, which has 49 electrons, the noble gas that comes before it is krypton. Krypton has 36 electrons, so its electron configuration represents the core electrons of indium. The remaining electrons are then added outside the krypton core.
What Is Noble Gas Notation?
Noble gas notation uses the symbol of a noble gas in brackets to represent core electrons. It helps avoid writing the full configuration from the start. This method highlights only the valence and outer electrons.
Noble Gas Notation For Indium
Indium’s full electron configuration is long and complex. Using noble gas notation, it starts with [Kr]. This stands for the 36 electrons in krypton’s configuration.
After [Kr], indium’s electrons fill the 4d, 5s, and 5p orbitals. The notation looks like this: [Kr] 4d10 5s2 5p1. This shows the extra 13 electrons beyond krypton.
Why Use Noble Gas Notation?
This notation makes reading and writing easier. It reduces errors and keeps focus on important electrons. Chemists use it to quickly understand an element’s electron setup and chemical properties.
Valence Electrons In Indium
Understanding the valence electrons in indium is key to grasping its chemical behavior. Valence electrons are the electrons in the outermost shell of an atom. They play a major role in bonding and reactions.
Indium has three valence electrons. These electrons reside in the fifth energy level. They influence how indium interacts with other elements.
Electron Configuration Of Indium
Indium’s full electron configuration is [Kr] 4d10 5s2 5p1. This shows all the electrons arranged in their shells and subshells. The last three electrons (5s2 5p1) are the valence electrons.
Role Of Valence Electrons In Chemical Properties
The three valence electrons help indium form bonds with other atoms. They can be shared or lost in chemical reactions. This makes indium a good conductor and useful in electronics.
Valence Electrons And Indium’s Group
Indium belongs to group 13 in the periodic table. Elements in this group have three valence electrons. This similarity explains why indium shares some properties with other group 13 elements.
Credit: www.schoolmykids.com
Electron Configuration Of Indium Ions
Indium ions form when indium atoms lose electrons. These ions have different electron configurations than neutral atoms. Understanding these changes helps explain indium's chemical behavior and bonding.
Indium commonly forms +1 and +3 ions. Each ion loses electrons from specific orbitals. This loss affects the electron arrangement around the nucleus.
Electron Configuration Of Indium (in⁺) Ion
The In⁺ ion loses one electron from the outermost shell. This electron is removed from the 5p orbital. The configuration becomes [Kr] 4d10 5s2 5p0. This means the 5p orbital is empty in In⁺.
Electron Configuration Of Indium (in³⁺) Ion
The In³⁺ ion loses three electrons. It loses two from the 5p and one from the 5s orbital. The new configuration is [Kr] 4d10. This shows a stable, full d-subshell after ionization.
Effect Of Ionization On Indium’s Electron Configuration
Losing electrons changes the electron cloud size. It increases the effective nuclear charge on remaining electrons. This makes In³⁺ smaller and more positively charged than In or In⁺.
These changes influence indium’s reactivity and bonding. Ions interact differently with other atoms and molecules.
Role In Chemical Bonding
The electron configuration of indium plays a key role in its chemical bonding. Understanding how its electrons are arranged helps explain how indium interacts with other elements. This knowledge is important for predicting the types of bonds indium can form.
Indium’s valence electrons, those in the outermost shell, are most involved in bonding. These electrons determine indium’s chemical properties and reactivity. The configuration influences how indium shares or transfers electrons to form compounds.
Electron Configuration Of Indium
Indium’s electron configuration is [Kr] 4d10 5s2 5p1. This means it has a full 4d subshell, two electrons in the 5s, and one electron in the 5p orbital. These outer electrons are available for bonding in chemical reactions.
Valence Electrons And Bond Formation
Indium has three valence electrons: two 5s and one 5p electron. These electrons can be lost or shared during bonding. Indium often forms +3 oxidation state by losing all three valence electrons. This creates ionic or covalent bonds depending on the partner element.
Types Of Bonds Formed By Indium
Indium can form metallic, ionic, and covalent bonds. In metallic bonds, indium atoms share electrons freely in a lattice. In ionic bonds, indium donates electrons to nonmetals. Covalent bonding involves sharing electrons, especially with other metals or metalloids.
Impact On Chemical Reactivity
Indium’s electron configuration makes it moderately reactive. It reacts easily with halogens and oxygen. Its ability to lose three electrons allows it to create stable compounds. The electron arrangement also influences its use in alloys and semiconductors.

Credit: www.youtube.com
Comparison With Neighboring Elements
Indium’s electron configuration shares similarities with its neighbors, tin and cadmium. It has a unique arrangement in the 5s and 4d orbitals. These differences affect its chemical behavior and placement in the periodic table.
Electron Configuration Of Indium
Indium has an atomic number of 49. Its electron configuration is unique compared to its neighbors. It fills the 5s and 4d orbitals before placing electrons in 5p. This pattern affects its chemical behavior and properties.
Comparison With Tin (sn)
Tin has an atomic number of 50, just after indium. Its electron configuration ends in 5p², one more electron than indium's 5p¹. This extra electron changes tin's reactivity and bonding characteristics. Tin tends to form +2 and +4 oxidation states, while indium commonly forms +3.
Comparison With Cadmium (cd)
Cadmium, atomic number 48, lies just before indium. Its electron configuration ends with 4d¹⁰ 5s², completely filling the d and s orbitals. Unlike indium, cadmium does not have electrons in the 5p orbital. This difference makes cadmium less reactive and more metallic.
Impact Of Electron Configuration Differences
Indium's single electron in the 5p orbital gives it unique chemical traits. Neighboring elements either have fully filled or more populated 5p orbitals. This influences their conductivity, reactivity, and bonding. Indium's configuration balances metallic and non-metallic properties.
Electron Configuration And Indium Properties
Indium is a soft, silvery metal with unique chemical traits. Its electron configuration reveals key insights into its behavior. Understanding how electrons arrange in indium helps explain its reactivity and uses.
Electrons fill orbitals in a specific order based on energy levels. This order influences how indium bonds and interacts with other elements. The electron configuration also relates to its position in the periodic table.
Basic Electron Configuration Of Indium
Indium has an atomic number of 49, meaning it has 49 electrons. These electrons fill orbitals starting from the lowest energy. The full electron configuration is: 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶ 4d¹⁰ 5s² 5p¹.
This shows indium has filled 4d and 5s subshells, with one electron in the 5p orbital. The outermost electrons mainly define its chemical properties.
Shorthand Electron Configuration
Using the noble gas notation, indium’s electron configuration is written as [Kr] 4d¹⁰ 5s² 5p¹. This is shorter and easier to read. Here, [Kr] represents the electron configuration of krypton, the preceding noble gas.
This notation highlights the valence electrons in the 5s and 5p orbitals, crucial for bonding and reactions.
Relation Between Electron Configuration And Indium’s Properties
The single electron in the 5p orbital makes indium moderately reactive. It tends to lose this electron and the two 5s electrons, forming a +3 oxidation state. This is common in many of its compounds.
Indium’s electron arrangement leads to metallic conductivity and softness. The filled 4d subshell adds some stability to the atom. Its properties are useful in electronics and alloys.
Applications Influenced By Electron Arrangement
The electron configuration of indium shapes many of its uses in technology and industry. Indium's unique arrangement of electrons allows it to interact with other elements and materials in special ways. This electron structure affects its electrical, optical, and chemical properties. These properties make indium valuable in various applications.
Understanding how indium’s electrons are arranged helps explain why it works well in these areas. The way electrons fill the 5s and 5p orbitals influences its behavior. This section explores key applications where electron configuration plays a crucial role.
Electronics and SemiconductorsIndium’s electron structure gives it excellent electrical conductivity. It is widely used in semiconductors and electronic devices. Its ability to form thin, transparent layers of indium tin oxide (ITO) is important. ITO layers serve as conductive coatings in touchscreens and flat-panel displays. The electron arrangement enables efficient electron flow, enhancing device performance.
Alloys and SoldersIndium’s unique electron configuration helps it bond with other metals easily. This property makes it ideal for creating special alloys and low-melting-point solders. These solders are used in delicate electronics where heat must be controlled. The specific electron arrangement contributes to indium’s softness and malleability, allowing it to form strong, flexible bonds.
Solar Panels and PhotovoltaicsIndium compounds, like indium gallium arsenide, have useful electron properties for solar cells. Their electron arrangement helps absorb sunlight efficiently. This improves solar panel efficiency by converting more light into electricity. Electron configuration thus directly impacts the energy capture and conversion process.
Optoelectronics and LEDsThe way electrons fill indium’s orbitals allows it to emit light at different wavelengths. This makes indium important in light-emitting diodes (LEDs) and lasers. Indium-based materials are used to produce bright, efficient light sources. Their electron structure supports stable and tunable light output.

Credit: www.chegg.com
Frequently Asked Questions
What Element Has An Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 corresponds to the element calcium.
How To Write The Electron Configuration Of Indium?
Write indium’s electron configuration by filling orbitals in order: [Kr] 4d¹⁰ 5s² 5p¹. This represents 49 electrons.
What Element Is 1s2 2s2 2p6 3s2 3p6 3d7 4s2?
The electron configuration 1s2 2s2 2p6 3s2 3p6 3d7 4s2 corresponds to the element cobalt (Co).
Which Atom Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d7?
The electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d7 belongs to the element Cobalt (Co).
Conclusion
Understanding the electron configuration of indium helps explain its chemical behavior. Indium has electrons arranged in a specific order within its shells and subshells. This arrangement influences its bonding and reactivity in many compounds. Knowing this pattern aids students and chemists in predicting indium’s properties.
It also connects to its position in the periodic table. Simple and clear, the electron configuration offers a foundation for further study in chemistry. Exploring elements like indium builds a stronger grasp of atomic structure and element trends.



No comments