Electron Configuration of Bromine
Are you curious about how bromine’s electrons are arranged and why that matters? Understanding the electron configuration of bromine can unlock a clearer picture of its chemical behavior, reactivity, and role in everyday compounds.
Whether you’re a student tackling chemistry or simply fascinated by the elements around you, knowing where bromine’s electrons live will give you an edge. You’ll discover step-by-step how to write bromine’s electron configuration, what it reveals about its properties, and why those outer electrons are so important.
Ready to dive in and make sense of bromine’s atomic structure? Let’s get started!
Credit: www.schoolmykids.com
Bromine Basics
Bromine is a chemical element with the symbol Br. It belongs to the halogen group. Bromine is unique because it is a liquid at room temperature. It has a reddish-brown color and a strong smell.
This element plays an important role in many chemical reactions. It is used in flame retardants, photography, and medicines. Understanding bromine's electron configuration helps explain its behavior and properties.
Atomic Number And Symbol
Bromine's atomic number is 35. This means it has 35 protons in its nucleus. Its chemical symbol is Br. The atomic number tells how many electrons the atom has when neutral.
Position In The Periodic Table
Bromine is located in period 4 and group 17 of the periodic table. Group 17 elements are called halogens. These elements share similar properties like high reactivity and seven valence electrons.
Valence Electrons And Reactivity
Bromine has seven valence electrons. These electrons are in the outermost shell. They determine how bromine reacts with other elements. Bromine tends to gain one electron to complete its outer shell.
Atomic Number And Electrons
The atomic number of bromine reveals the number of electrons it has. This number is essential for understanding its electron configuration. Bromine’s atomic number is 35. This means it contains 35 electrons orbiting its nucleus.
Electrons fill energy levels or shells around the nucleus. The arrangement of these electrons defines the chemical behavior of bromine. Electrons fill shells in a specific order based on their energy.
Understanding Atomic Number
Atomic number is the count of protons in an atom’s nucleus. For bromine, this number is 35. Since atoms are neutral, the number of electrons equals the atomic number. Thus, bromine has 35 electrons.
Counting Electrons In Bromine
Electrons are arranged in shells around the nucleus. Each shell holds a limited number of electrons. The first shell can hold 2, the second 8, the third 18, and so on. Bromine’s 35 electrons fill these shells step-by-step.
Importance Of Electron Arrangement
The way electrons fill shells affects bromine’s properties. Electrons fill lower energy shells first. Outer shells determine how bromine reacts with other elements. Knowing the electron count helps predict these reactions.
Periodic Table Position
Bromine holds a unique spot in the periodic table. It is part of the halogen group. This group sits in Group 17, known for reactive nonmetals.
Bromine’s atomic number is 35. This means it has 35 protons and electrons in its neutral state. It is placed in Period 4, which shows it has four energy levels of electrons.
Its position influences its electron configuration and chemical behavior. Being a halogen, bromine has seven valence electrons. These electrons determine how bromine bonds with other elements.
Group And Period
Bromine belongs to Group 17. This group contains fluorine, chlorine, iodine, and astatine. All these elements share similar properties.
It is in Period 4. Period number shows the highest energy level that bromine’s electrons occupy. For bromine, this is the fourth shell.
Block In The Periodic Table
Bromine is in the p-block. The p-block includes groups 13 to 18. Elements here have their outermost electrons in p orbitals.
This block placement affects bromine’s electron configuration. It ends in the 4p orbital, which holds five of its valence electrons.
Relationship To Neighboring Elements
Bromine is between selenium and krypton. Selenium is a nonmetal in Group 16. Krypton is a noble gas in Group 18.
This position shows bromine’s intermediate reactivity. It is more reactive than krypton but less than selenium’s neighbor, arsenic.
Shells And Subshells
Understanding the electron configuration of bromine starts with its shells and subshells. Electrons orbit the nucleus in layers called shells. Each shell holds smaller groups called subshells. These subshells have different shapes and energy levels. They are labeled as s, p, d, and f. The arrangement of electrons in these shells and subshells defines bromine’s chemical behavior.
What Are Electron Shells?
Electron shells are energy levels around an atom's nucleus. They are numbered starting from 1, closest to the nucleus. Each shell can hold a limited number of electrons. The first shell holds up to 2 electrons. The second shell holds up to 8 electrons. The third shell can hold up to 18 electrons. The fourth shell holds electrons as well, but it is not always full.
Understanding Subshells In Bromine
Subshells are parts of each shell. They have different shapes named s, p, d, and f. Each subshell holds a specific number of electrons. The s subshell holds 2 electrons, the p holds 6, the d holds 10, and the f holds 14. Bromine’s electrons fill these subshells in order, starting with the lowest energy level.
Bromine’s Electron Distribution In Shells And Subshells
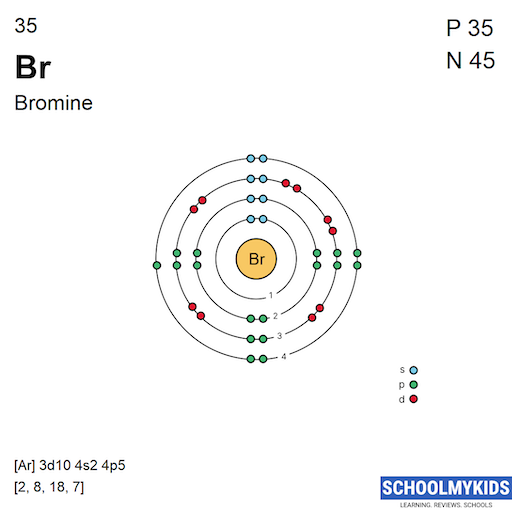
Bromine has 35 electrons. These electrons fill shells and subshells step by step. The first shell has 2 electrons in the 1s subshell. The second shell has 8 electrons split between 2s and 2p subshells. The third shell has 18 electrons filling 3s, 3p, and 3d subshells. The fourth shell has 7 electrons in 4s and 4p subshells. This pattern explains bromine's position on the periodic table and its chemical traits.
Filling Order Of Orbitals
The filling order of orbitals explains how electrons are arranged in an atom. Electrons fill orbitals starting from the lowest energy level to higher levels. This order follows specific rules that keep atoms stable. Understanding this helps to know the electron configuration of bromine.
Bromine has 35 electrons. These electrons fill the orbitals step by step. The order follows the Aufbau principle, which means "building up." Each orbital can hold a limited number of electrons. The filling order is important to write the correct electron configuration for bromine.
What Is The Aufbau Principle?
The Aufbau principle says electrons fill orbitals starting with the lowest energy. Electrons fill the 1s orbital first, then 2s, 2p, and so on. This rule helps predict where electrons are in an atom. For bromine, electrons fill orbitals up to the 4p level.
The Order Of Orbital Filling
Orbitals fill in a specific sequence: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p. This order is based on energy levels. Lower energy orbitals fill before higher energy ones. For bromine, electrons fill orbitals up to 4p with five electrons in that sublevel.
Electron Configuration Of Bromine
Bromine’s electron configuration is written as 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁵. This shows how electrons fill each orbital. The 4p orbital has five electrons, which explains bromine’s chemical behavior. The filling order helps understand these arrangements clearly.

Credit: homework.study.com
Full Electron Configuration
Bromine’s full electron configuration shows how its 35 electrons fill each energy level and orbital. Starting from 1s to 4p, electrons arrange to follow the rules of atomic structure. This configuration helps explain bromine’s chemical behavior and properties.
The full electron configuration of an element shows the exact arrangement of electrons in its atomic orbitals. It explains how electrons fill different energy levels and sublevels around the nucleus. This arrangement affects the element's chemical behavior and properties.
Bromine has 35 electrons. These electrons occupy orbitals in a specific order based on energy levels. The full configuration lists all occupied orbitals from the lowest to the highest energy.
Electron Arrangement In Bromine
Bromine’s electrons fill orbitals starting with the 1s orbital. Then they move through 2s, 2p, 3s, and so on. The process follows the Aufbau principle, which means electrons fill lower energy orbitals first.
The orbitals fill in this order: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p. Each orbital can hold a specific number of electrons. For example, s orbitals hold 2, p orbitals hold 6, and d orbitals hold 10 electrons.
Complete Electron Configuration Of Bromine
The full electron configuration of bromine is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁵
This shows all electrons placed in their correct orbitals. The superscripts indicate the number of electrons in each orbital.
The 4p⁵ part means that bromine has five electrons in the 4p sublevel. This is important for understanding bromine’s chemical reactivity.
Noble Gas Shorthand Notation
The noble gas shorthand notation simplifies writing electron configurations. It uses the nearest noble gas to represent core electrons. This method saves time and space in chemistry writing.
For bromine, the noble gas shorthand starts with argon. Argon’s electron configuration covers the first 18 electrons. Then, the remaining electrons fill the outer shells of bromine.
What Is Noble Gas Shorthand Notation?
Noble gas shorthand notation replaces inner electron shells with a noble gas symbol. It shows only the electrons beyond the noble gas core. This way, it makes complex electron configurations easier to read.
Noble Gas Shorthand For Bromine
Bromine’s atomic number is 35, meaning it has 35 electrons. The nearest noble gas before bromine is argon, with 18 electrons. Its symbol is [Ar].
After argon, bromine’s electrons fill the 4s, 3d, and 4p orbitals. The shorthand configuration is:
Benefits Of Using Noble Gas Shorthand
This notation reduces long electron configurations to shorter forms. It helps students and chemists focus on valence electrons. Valence electrons determine chemical behavior and bonding.
Shorthand notation also minimizes errors in writing or reading configurations. It is a standard method in chemistry education and research.
Valence Electrons Role
The valence electrons of bromine play a crucial role in its chemical behavior. These electrons are located in the outermost shell and determine how bromine reacts with other elements. Understanding valence electrons helps explain bromine's bonding and reactivity.
Bromine has seven valence electrons. These electrons fill the 4th energy level in the 4s and 4p orbitals. The presence of seven valence electrons makes bromine highly reactive, as it seeks to complete its outer shell.
What Are Valence Electrons?
Valence electrons are the electrons in the highest energy level of an atom. They are responsible for forming chemical bonds. In bromine, these electrons are found in the 4s and 4p orbitals.
Valence Electron Configuration Of Bromine
Bromine's electron configuration ends with 4s² 4p⁵. This means it has two electrons in the 4s orbital and five in the 4p orbitals. These seven valence electrons influence bromine’s properties.
How Valence Electrons Affect Bromine's Reactivity
Bromine tends to gain one electron to complete its outer shell of eight electrons. This makes bromine a strong oxidizing agent. The valence electrons are key to this behavior.
Valence Electrons And Chemical Bonding
Bromine forms bonds by sharing or gaining electrons. The seven valence electrons allow bromine to form covalent or ionic bonds. This explains bromine's role in many chemical compounds.
Bromide Ion Configuration
Bromide ion forms when bromine gains an extra electron. This changes its electron arrangement. Understanding this helps explain its chemical behavior and reactivity.
The extra electron fills the outermost shell. This makes the ion more stable than neutral bromine. The electron configuration shows how electrons are spread in energy levels and orbitals.
What Is The Bromide Ion?
Bromide ion has a negative charge. It forms from bromine atom by gaining one electron. This extra electron completes the outer shell. This makes the ion more stable and less reactive.
Electron Configuration Of Bromide Ion
The neutral bromine atom has 35 electrons. Its electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁵. Adding one electron changes it to 4p⁶ for bromide ion.
So, the bromide ion configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶. This full outer shell is similar to the noble gas krypton. It explains bromide’s stable and less reactive nature.
Why Does Bromide Gain An Electron?
Bromine wants to complete its outer shell. Gaining one electron fills the 4p orbital. This creates a stable electron configuration. The full 4p orbital lowers energy and increases stability.
Impact On Chemical Properties
The bromide ion acts as a stable anion in compounds. It often pairs with positive ions. This ion is important in many chemical reactions. Its full outer shell helps it resist further change.

Credit: winter.group.shef.ac.uk
Applications In Chemistry
The electron configuration of bromine plays a key role in chemistry. It helps explain how bromine atoms interact with other elements. Understanding this configuration allows chemists to predict bromine's behavior in chemical reactions and compounds.
Bromine’s electron arrangement determines its bonding and reactivity. This knowledge is essential in organic and inorganic chemistry. It also supports the design of new materials and chemical processes involving bromine.
Formation Of Bromine Compounds
Bromine’s electron configuration ends in 4p5. This means bromine has seven electrons in its outer shell. It tends to gain one electron to complete its octet. This makes bromine a strong oxidizing agent in forming compounds.
Bromine commonly forms ionic bonds by accepting an electron. It also forms covalent bonds by sharing electrons. These bonds create important compounds like hydrogen bromide and brominated hydrocarbons.
Role In Redox Reactions
Bromine’s ability to gain or lose electrons makes it active in redox reactions. It can act as an oxidizing agent by accepting electrons. This property is useful in chemical synthesis and industrial processes.
Its electron configuration allows bromine to participate in electron transfer easily. This helps in reactions such as halogen displacement and organic halogenation. These reactions are vital in producing many chemicals.
Use In Analytical Chemistry
Bromine’s electron configuration influences its color and reactivity. These traits help identify bromine in mixtures. Analytical methods like titration use bromine's reactivity for precise measurements.
Its distinct electron arrangement also aids in spectroscopy. Scientists use this to detect bromine in different samples. This is important for environmental and pharmaceutical analysis.
Frequently Asked Questions
How Do You Write The Electron Configuration For Bromine?
Write bromine’s electron configuration as 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁵. This represents its 35 electrons arranged by energy levels and orbitals.
What Element Has The Electron Configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5 is bromine (Br). It has 35 electrons.
Is Electron Shell 2 8 8 Or 2 8 18?
The electron shell configuration can be 2 8 8 or 2 8 18 depending on the element. Elements with atomic numbers up to 18 have 2 8 8. Heavier elements fill the third shell up to 18 electrons, so 2 8 18 applies for those beyond argon.
What Is The Electronic Configuration Of Bromine 2,8,18,7?
The electronic configuration of bromine is 2, 8, 18, 7. It shows electrons in each shell from inner to outer. This arrangement reflects bromine's 35 total electrons accurately.
Conclusion
Understanding bromine’s electron configuration helps explain its chemical behavior. It has 35 electrons arranged in specific shells and orbitals. The outermost shell has five electrons in the p-orbital, making bromine reactive. This pattern shows why bromine forms certain bonds and compounds.
Knowing this helps in studying chemistry and related sciences. Simple steps and the periodic table make it easy to write bromine’s electron configuration. Keep practicing to become confident in reading electron arrangements for other elements too.



No comments