Electron Configuration of Arsenic
Are you curious about how arsenic’s electrons are arranged and why that matters? Understanding the electron configuration of arsenic can unlock insights into its chemical behavior, reactivity, and role in everyday materials.
Whether you’re a student tackling chemistry or just someone wanting to grasp the basics, this guide will break down arsenic’s electron setup in a clear, simple way. Keep reading, and you’ll discover how those tiny particles shape everything about this fascinating element—and how you can use that knowledge to make sense of the world around you.
Credit: enthu.com
Basic Electron Layout
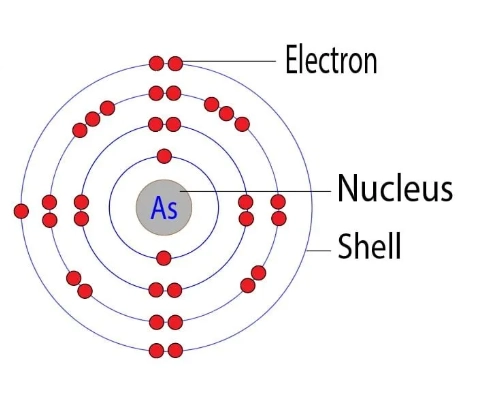
The basic electron layout reveals how electrons fill the orbitals around arsenic’s nucleus. It helps us understand arsenic’s chemical behavior and bonding traits. Each electron occupies a specific energy level and sublevel in an orderly pattern.
Arsenic has 33 electrons. These electrons fill orbitals from the lowest energy level up. The arrangement follows the Aufbau principle, ensuring stability in the atom’s structure.
Electron Shells And Energy Levels
Electrons in arsenic are arranged in shells numbered 1 to 4. The first shell holds 2 electrons. The second shell contains 8 electrons. The third shell fills with 18 electrons. The fourth shell has 5 electrons. Each shell corresponds to increasing energy levels.
Subshells And Orbital Types
Within each shell, electrons occupy subshells labeled s, p, d, and f. Arsenic’s electrons fill s and p subshells mostly. The 3d subshell is also filled before the 4p starts filling. This order follows electron energy rules and orbital capacity limits.
Arsenic’s Ground State Configuration
The ground state electron configuration of arsenic is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p³. This layout shows how electrons fill from the innermost shell outward. The outermost electrons in 4p orbitals contribute to chemical reactions and bonding.

Credit: winter.group.shef.ac.uk
Orbital Filling Order
Arsenic's electrons fill orbitals following a specific order based on energy levels. This order helps explain its electron configuration clearly. Electrons occupy orbitals starting from lower to higher energy states.
Understanding Orbital Filling Order
Orbital filling order explains how electrons fill atomic orbitals. Electrons occupy orbitals from lowest to highest energy. This order follows specific rules to keep atoms stable. The arrangement affects the chemical properties of elements, including arsenic.
Each orbital can hold a limited number of electrons. The order depends on energy levels and sublevels. Knowing this helps predict electron configuration accurately.
Energy Levels And Sublevels
Electrons fill orbitals by increasing energy levels. Each level contains sublevels named s, p, d, and f. The s sublevel holds 2 electrons, p holds 6, d holds 10, and f holds 14. The filling order is not strictly sequential by principal quantum number.
Applying The Aufbau Principle
The Aufbau principle guides electron filling order. Electrons enter the lowest energy orbital available first. This principle ensures stability in the electron arrangement. For arsenic, electrons fill orbitals up to the 4p sublevel.
Electron Configuration Of Arsenic
Arsenic has 33 electrons to place in orbitals. Following the order, electrons fill 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p orbitals. The final configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p³. This reflects the orbital filling order clearly.
Electron Configuration Notation
Electron configuration notation shows how electrons arrange around an atom's nucleus. It uses numbers and letters to represent electron positions in energy levels and orbitals.
This notation helps understand an element's chemical behavior and properties. For arsenic, it reveals the distribution of electrons that influence bonding and reactivity.
Basic Structure Of Electron Configuration Notation
The notation includes numbers, letters, and superscripts. Numbers indicate energy levels, letters stand for orbital types, and superscripts show electron counts.
For example, "1s²" means two electrons in the first energy level's s orbital. This format repeats for each orbital filled by electrons.
Electron Configuration Of Arsenic
Arsenic has 33 electrons. Its electron configuration is written as: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p³.
This shows electrons fill lower energy orbitals first, then move to higher ones. The 4p³ indicates three electrons in the 4p orbital, important for arsenic's chemistry.
Significance Of Electron Configuration Notation
This notation simplifies understanding electron arrangement. It helps predict chemical bonds and element behavior in reactions.
Using this notation, scientists and students quickly grasp arsenic’s electronic structure and its role in compounds.
Arsenic Valence Electrons
Valence electrons are the outermost electrons of an atom. They play a key role in chemical bonding and reactions. Arsenic has five valence electrons. These electrons determine its chemical behavior and bonding patterns.
Understanding arsenic’s valence electrons helps explain its position in the periodic table. Arsenic belongs to group 15, also called the nitrogen group. Elements in this group share the same number of valence electrons.
Electron Configuration Of Arsenic
The full electron configuration of arsenic is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p³. The electrons in the 4s and 4p orbitals are the valence electrons. These are the electrons available for bonding.
Number Of Valence Electrons
Arsenic has five valence electrons. Two electrons are in the 4s orbital. Three electrons are in the 4p orbital. These five electrons influence arsenic’s chemical properties and reactivity.
Importance Of Valence Electrons In Bonding
Valence electrons form bonds with other atoms. Arsenic can share or gain electrons to complete its outer shell. This leads to different types of chemical compounds. Its five valence electrons allow it to form three covalent bonds easily.
Electron Shell Distribution
The electron shell distribution shows how electrons are arranged around an atom's nucleus. It helps us understand chemical behavior and bonding. Each shell can hold a specific number of electrons. Electrons fill shells starting from the closest to the nucleus.
Arsenic has 33 electrons. These electrons spread across several shells. The distribution follows strict rules based on energy levels and quantum mechanics.
First Electron Shell (k-shell)
The first shell is the closest to the nucleus. It can hold up to 2 electrons. For arsenic, this shell is fully occupied with 2 electrons. These electrons are in the 1s orbital, which is the lowest energy level.
Second Electron Shell (l-shell)
The second shell holds up to 8 electrons. Arsenic’s L-shell is also full. It contains electrons in the 2s and 2p orbitals. This shell protects the nucleus and supports outer electrons.
Third Electron Shell (m-shell)
The third shell can hold up to 18 electrons. Arsenic fills 18 electrons here. These electrons occupy the 3s, 3p, and 3d orbitals. This shell is important for chemical reactivity.
Fourth Electron Shell (n-shell)
The fourth shell contains the remaining electrons for arsenic. It holds 5 electrons in the 4s and 4p orbitals. These are the valence electrons that define arsenic’s chemical properties.
Ground State Configuration
Arsenic’s ground state electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p³. This shows how electrons fill energy levels around the nucleus. Understanding this helps explain arsenic’s chemical behavior.
Understanding The Ground State Configuration
The ground state configuration shows how electrons fill the lowest energy levels. It reveals the natural arrangement of electrons in an atom. For arsenic, this arrangement affects its chemical behavior and bonding.
Knowing the ground state helps predict how arsenic interacts with other elements. It also explains its position in the periodic table and its physical properties.
Electron Shells And Subshells In Arsenic
Arsenic has 33 electrons. These electrons fill shells and subshells in order of increasing energy. The shells are labeled as 1, 2, 3, and so on. Each shell contains subshells named s, p, d, and f.
The filling order follows specific rules that minimize the atom’s energy. Arsenic’s electrons fill up to the 4p subshell, showing its place in the periodic table.
Detailed Electron Configuration Of Arsenic
The full ground state electron configuration of arsenic is:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p³
This means arsenic’s electrons fill inner shells fully. The outermost electrons are in the 4p subshell with three electrons. These outer electrons are key for chemical reactions.
Importance Of The 4p³ Configuration
The 4p³ electrons give arsenic its unique properties. These electrons are less tightly held and can form bonds. This configuration explains arsenic’s reactivity and its behavior as a metalloid.
The half-filled p subshell (with three electrons) offers stability to arsenic’s atom. This stability impacts its electrical and chemical characteristics.
Excited State Possibilities
Exploring excited state possibilities reveals how arsenic’s electrons shift to higher energy levels. These changes affect its chemical behavior and bonding. Understanding this helps explain arsenic’s unique properties in reactions.
Understanding Excited State Possibilities
Arsenic's electrons can move from their normal places to higher energy spots.
This shift creates excited states, different from the usual ground state.
Studying these states helps us learn about arsenic's chemical behavior and reactions.
Electron Promotion In Arsenic
In excited states, electrons jump from filled orbitals to empty ones.
For arsenic, an electron may leave the 4p orbital and move to a higher 4d orbital.
This change requires energy but alters arsenic's bonding and properties.
Energy Levels And Stability
Excited states are less stable than the ground state because of higher energy.
Electrons tend to return quickly to their original orbitals, releasing energy.
The time spent in excited states affects arsenic’s reactivity and spectroscopy signals.
Impact On Chemical Reactions
Excited electrons can form different bonds than in the ground state.
These states may enable arsenic to react with other elements uniquely.
Understanding excited states helps predict arsenic's role in complex compounds.

Credit: www.youtube.com
Relation To Periodic Table
Arsenic’s electron configuration reflects its position in the periodic table’s p-block. It helps explain arsenic’s chemical behavior and bonding patterns. Electrons fill orbitals in order, matching arsenic’s atomic number 33.
Position Of Arsenic In The Periodic Table
Arsenic is element number 33 on the periodic table. It belongs to group 15, also called the nitrogen group. This group contains elements with five valence electrons. Arsenic sits in period 4, which means it has four energy levels of electrons.
Electron Configuration And Group Trends
Arsenic’s electron configuration shows its place in group 15 clearly. The configuration ends with 4p³, meaning three electrons fill the p orbital in the fourth energy level. This pattern matches other group 15 elements like nitrogen and phosphorus. They all have five electrons in their outer shell, giving them similar chemical properties.
Relation To Period And Energy Levels
The period number indicates how many electron shells arsenic has. Arsenic has electrons in shells 1 through 4. Its electron configuration starts from 1s and goes up to 4p. This explains arsenic’s size and energy compared to elements above and below it in the table.
Metalloid Characteristics Linked To Electron Configuration
Arsenic is a metalloid due to its electron arrangement. The mix of filled and half-filled orbitals gives arsenic unique properties. It can behave like a metal or a non-metal. This dual behavior is connected to its position in the periodic table and its electron structure.
Impact On Chemical Bonding
The electron configuration of arsenic directly affects how it bonds with other elements. Arsenic has five valence electrons. These electrons are available to form bonds. This configuration shapes arsenic’s chemical behavior and bonding type.
Arsenic's bonding pattern is mainly influenced by its valence shell. The arrangement allows arsenic to form three covalent bonds easily. It can also form bonds by sharing or gaining electrons. This versatility makes arsenic important in many chemical compounds.
Valence Electrons And Bond Formation
Arsenic's valence electrons are in the 4s and 4p orbitals. It has the configuration 4s² 4p³. These five electrons help arsenic to reach a stable octet. Arsenic usually shares three electrons to form three covalent bonds. This is common in compounds like arsenic trioxide and arsenic acid.
Covalent Bonding Characteristics
Arsenic often forms covalent bonds by sharing electrons. It tends to bond with elements like hydrogen, oxygen, and sulfur. The shared electrons create stable molecules with arsenic at the center. The covalent bonds are strong and directional, influencing the shape of molecules.
Role In Metallic And Ionic Bonding
Arsenic can also participate in metallic bonding in alloys. In these, electrons move freely around atoms. Arsenic does not typically form ionic bonds because it does not easily lose or gain electrons completely. Instead, it prefers sharing electrons to form covalent bonds.
Effect On Molecular Geometry
The electron configuration affects arsenic's molecular geometry. With three bonding pairs and one lone pair, arsenic compounds often have a trigonal pyramidal shape. This shape influences physical and chemical properties, like polarity and reactivity.
Comparison With Neighboring Elements
Comparing the electron configuration of arsenic with its neighboring elements reveals interesting patterns. These patterns help explain differences in chemical behavior and properties. Arsenic sits in group 15 of the periodic table, with elements like phosphorus before it and selenium after it.
Each neighboring element adds or removes electrons in specific orbitals. This change affects their reactivity and bonding characteristics. Understanding these differences helps in grasping arsenic’s role in chemistry and materials science.
Electron Configuration Of Phosphorus
Phosphorus has the electron configuration 1s² 2s² 2p⁶ 3s² 3p³. It ends with three electrons in the 3p sublevel. This is similar to arsenic but in the third shell instead of the fourth. Phosphorus tends to form three bonds, like arsenic, due to these three valence electrons.
Electron Configuration Of Arsenic
Arsenic’s electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p³. It has three electrons in the 4p sublevel, like phosphorus but in a higher energy level. This causes arsenic to have slightly different chemical properties and to be less reactive than phosphorus.
Electron Configuration Of Selenium
Selenium has the electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁴. It has four electrons in the 4p sublevel, one more than arsenic. This extra electron changes its behavior, making selenium more likely to gain electrons and form negative ions, unlike arsenic which often shares electrons.
Frequently Asked Questions
How To Write The Electron Configuration Of Arsenic?
Write arsenic’s electron configuration as 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p³. This shows all electrons in order of increasing energy levels.
Which Element Has The Electron Configuration Of 1s2 2s2 2p6 3s2 3p6 4s2 3d6?
The element with electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d6 is Iron (Fe). It has 26 electrons.
What Element Is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 6 5s 2 4d 6?
The element with electron configuration 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d⁶ is Ruthenium (Ru).
What Is The Electron Configuration Of Kr 5s24d105p2?
The electron configuration of krypton (Kr) is 1s² 2s² 2p⁶ 3s² 3p⁶ 3d¹⁰ 4s² 4p⁶. The given configuration 5s² 4d¹⁰ 5p² corresponds to tin (Sn), not Kr.
Conclusion
Understanding the electron configuration of arsenic helps explain its chemical behavior. Arsenic has 33 electrons arranged in specific energy levels. Its valence electrons play a key role in bonding and reactions. Knowing this setup aids in predicting arsenic’s properties and uses.
This knowledge is useful for students and science enthusiasts alike. Simple steps make learning electron configurations easier and more fun. Keep exploring to grasp how atoms like arsenic work in nature.



No comments